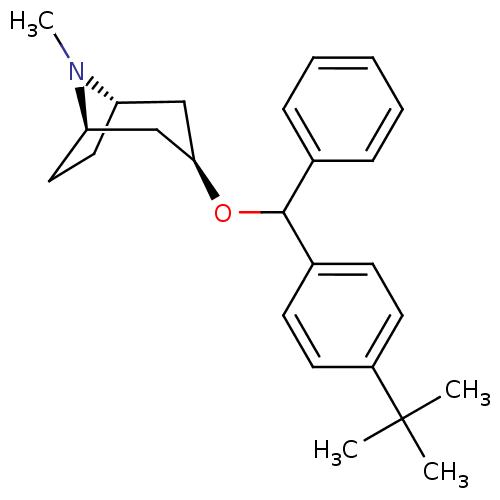
BDBM50453900 CHEMBL3084866
SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)OC(c1ccccc1)c1ccc(cc1)C(C)(C)C)N2C
InChI Key InChIKey=IGNVGOLMZFBAMQ-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50453900
Found 4 hits for monomerid = 50453900
Affinity DataKi: 54nMAssay Description:Affinity for rat M1 acetylcholine receptor using [3H]-AF DX 384 displacement.More data for this Ligand-Target Pair
Affinity DataKi: 148nMAssay Description:Affinity for rat M1 acetylcholine receptor using [3H]pirenzepine displacement.More data for this Ligand-Target Pair
Affinity DataKi: 1.92E+3nMAssay Description:Affinity for rat dopamine transporter using [3H]WIN-35428 displacement.More data for this Ligand-Target Pair
Affinity DataIC50: 4.46E+3nMAssay Description:Inhibition of [3H]dopamine uptake in rat caudate putamen.More data for this Ligand-Target Pair