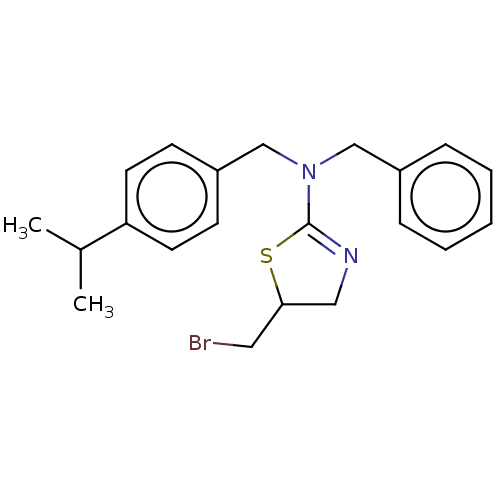
BDBM50500757 CHEMBL3752682
SMILES CC(C)c1ccc(CN(Cc2ccccc2)C2=NCC(CBr)S2)cc1
InChI Key InChIKey=UFBGCJXJQFVUKE-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50500757
Found 3 hits for monomerid = 50500757
TargetLiver carboxylesterase(Pig)
Institute of Physiologically Active Compounds Russian Academy of Sciences
Curated by ChEMBL
Institute of Physiologically Active Compounds Russian Academy of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.55E+3nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetLiver carboxylesterase(Pig)
Institute of Physiologically Active Compounds Russian Academy of Sciences
Curated by ChEMBL
Institute of Physiologically Active Compounds Russian Academy of Sciences
Curated by ChEMBL
Affinity DataKi: 3.54E+3nMAssay Description:Noncompetitive inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate assessed as steady state inhibition constant pre...More data for this Ligand-Target Pair
TargetCholinesterase(Horse)
Institute of Physiologically Active Compounds Russian Academy of Sciences
Curated by ChEMBL
Institute of Physiologically Active Compounds Russian Academy of Sciences
Curated by ChEMBL
Affinity DataIC50: 1.10E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair