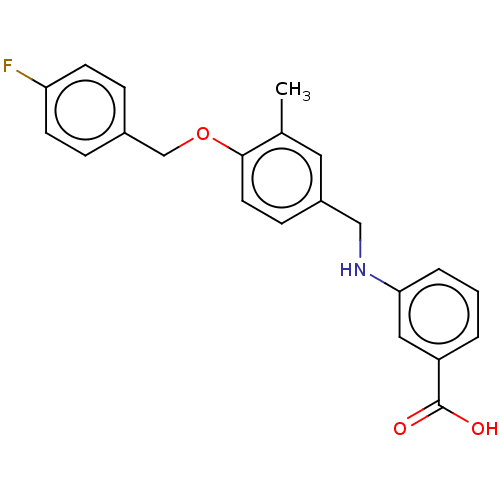
BDBM50511204 CHEMBL4463341
SMILES Cc1cc(CNc2cccc(c2)C(O)=O)ccc1OCc1ccc(F)cc1
InChI Key InChIKey=DDCNGNDFKHLAQY-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50511204
Found 3 hits for monomerid = 50511204
TargetPeroxisome proliferator-activated receptor alpha(Human)
University of Oklahoma
Curated by ChEMBL
University of Oklahoma
Curated by ChEMBL
Affinity DataEC50: 37nMAssay Description:Agonist activity at human PPARalpha expressed in nonhuman mammalian cells assessed as increase in receptor transcriptional activity incubated for 22 ...More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor alpha(Human)
University of Oklahoma
Curated by ChEMBL
University of Oklahoma
Curated by ChEMBL
Affinity DataEC50: 93nMAssay Description:Binding affinity to PPARalpha in human MIO-M1 cells assessed as increase in thermal stability preincubated for 3 hrs followed by exposure to 45.5 deg...More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor alpha(Human)
University of Oklahoma
Curated by ChEMBL
University of Oklahoma
Curated by ChEMBL
Affinity DataKd: 140nMAssay Description:Binding affinity to His-tagged PPARalpha LBD (unknown origin) expressed in Escherichia coli Rosetta (DE3) at 25 degree C by isothermal titration calo...More data for this Ligand-Target Pair