Report error Found 172 Enz. Inhib. hit(s) with Target = 'Granzyme B'
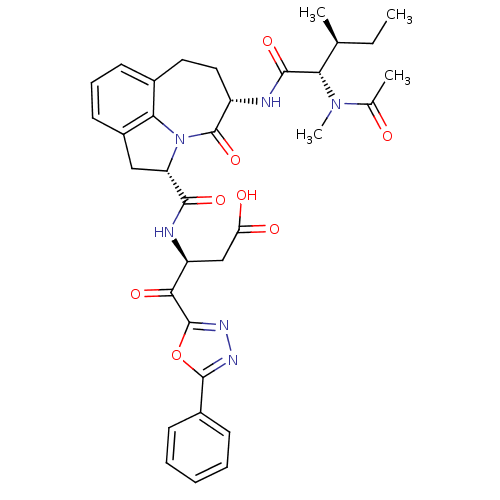
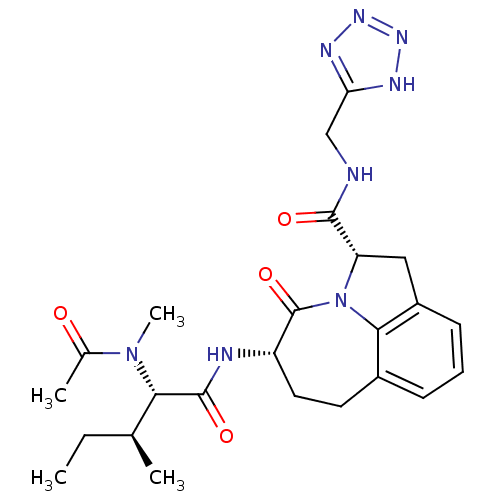
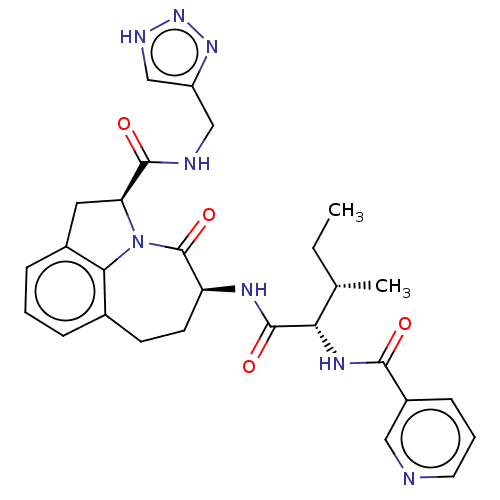
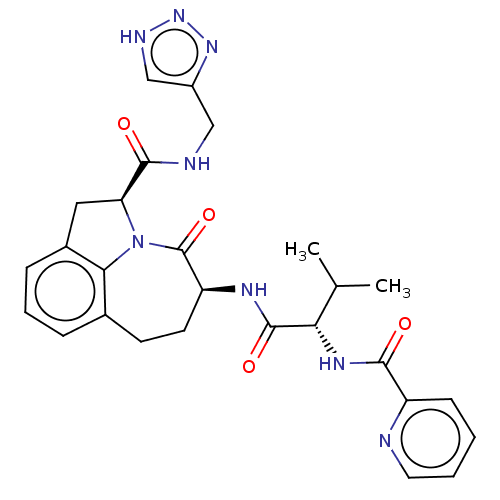
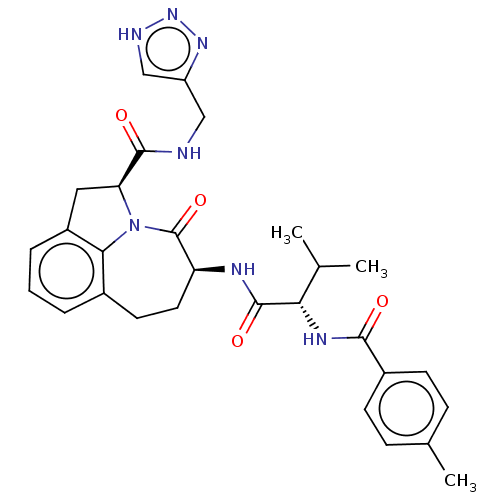
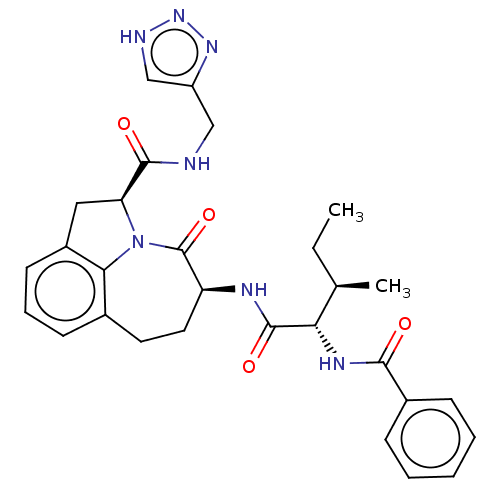
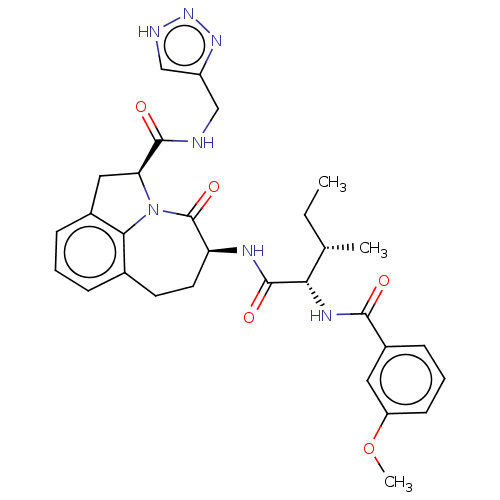
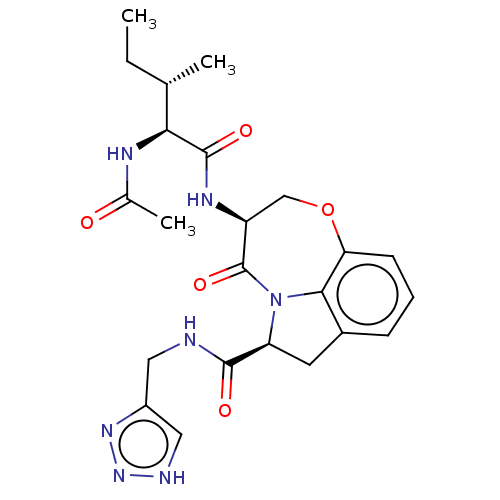
Affinity DataKi: 7nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
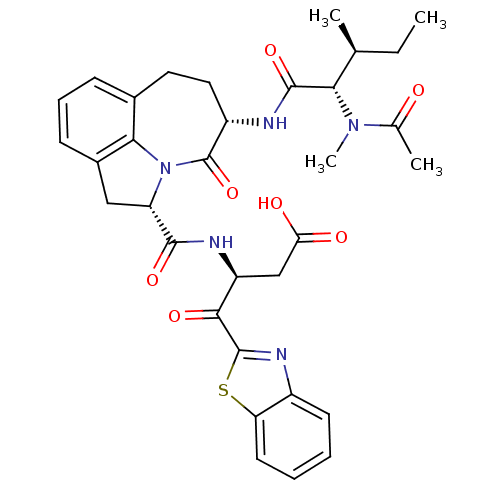
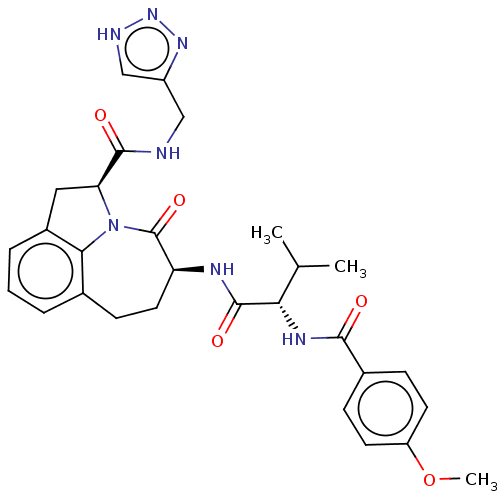
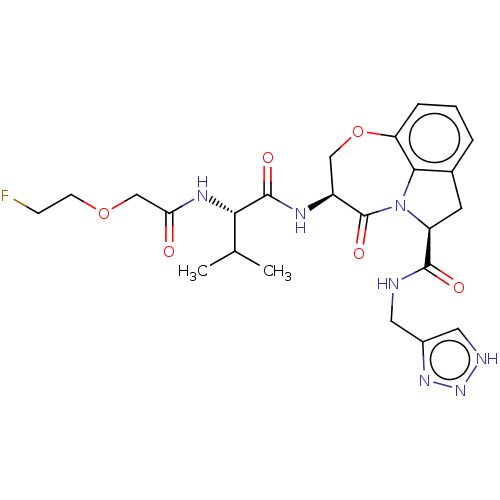
Affinity DataKi: 7nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
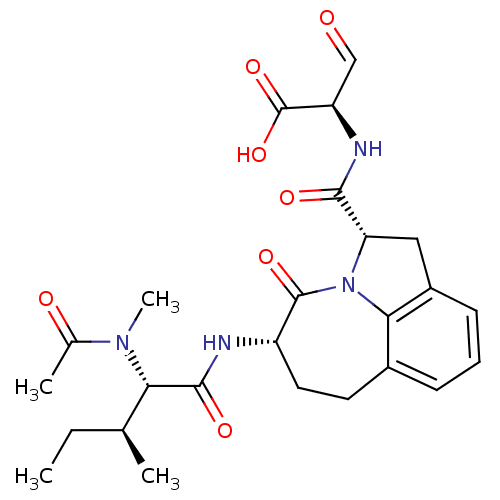
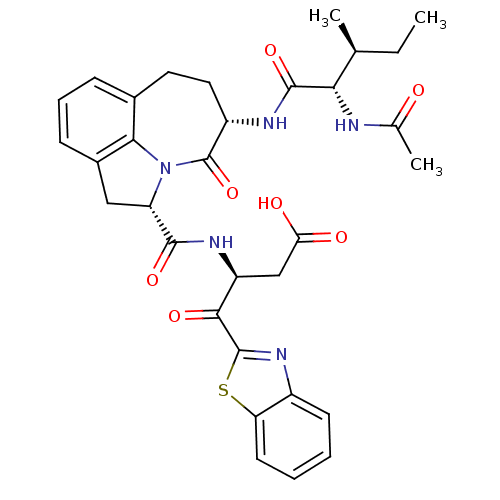
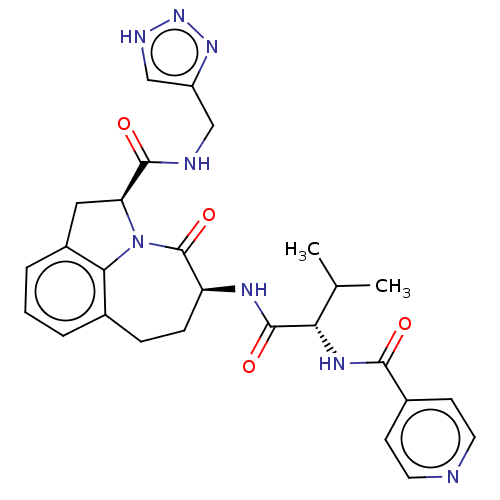
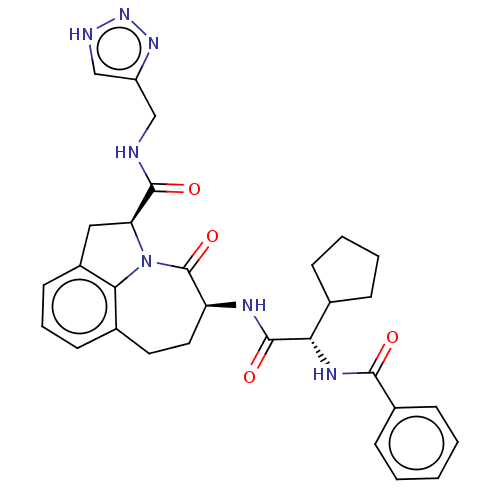
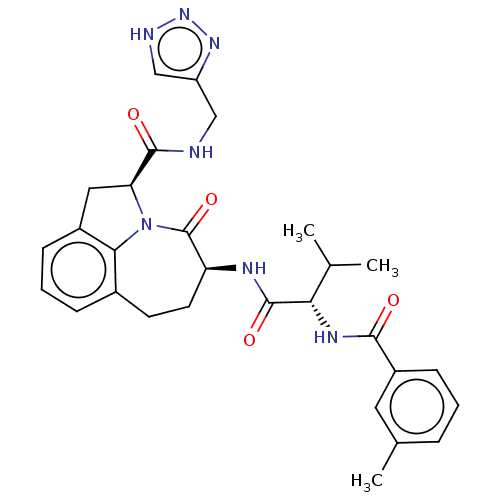
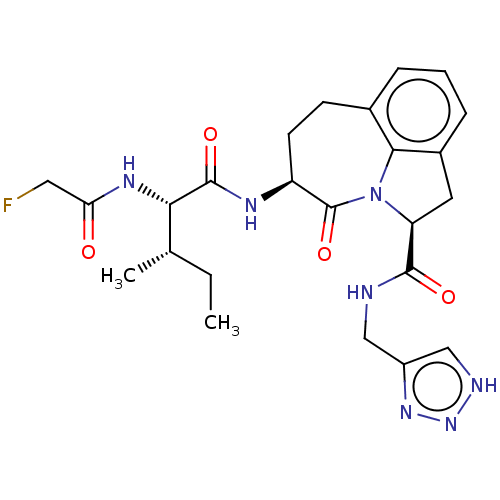
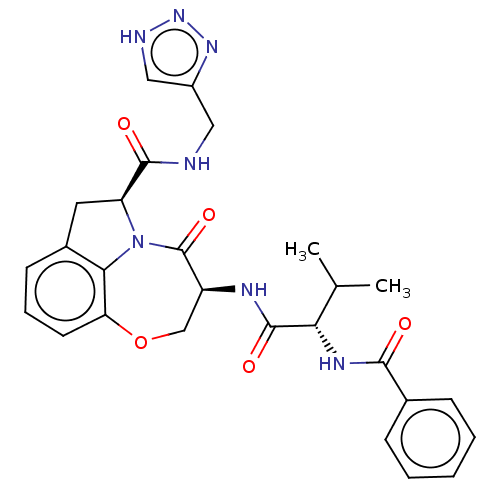
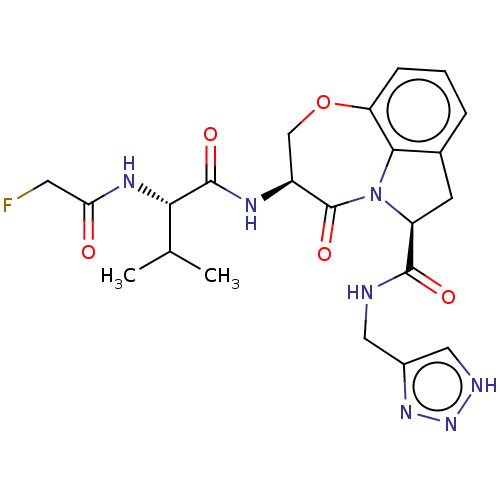
Affinity DataKi: 8nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
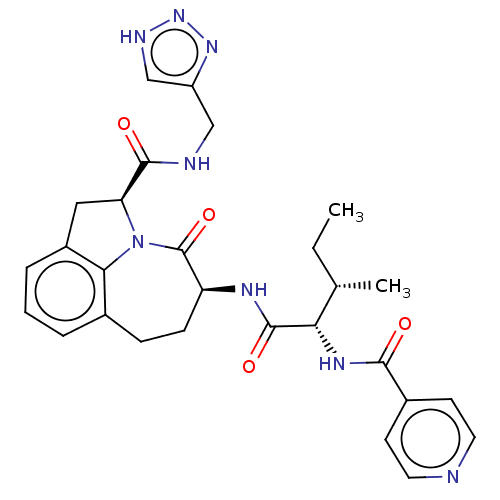
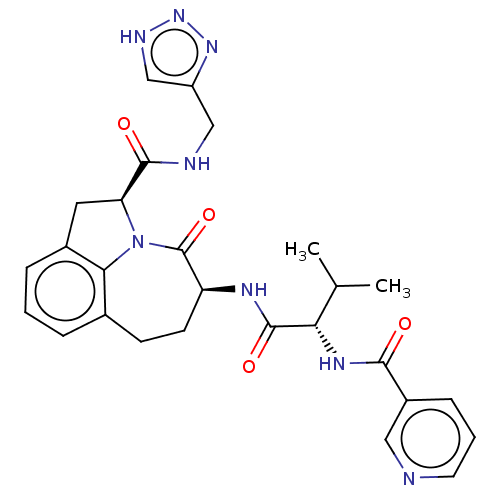
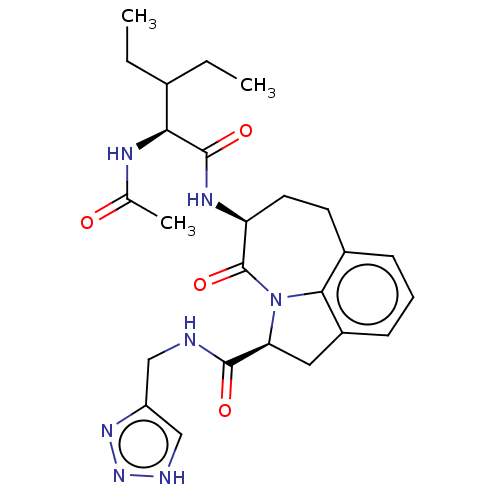
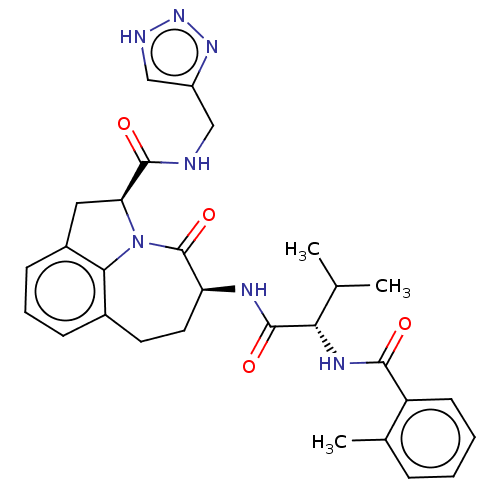
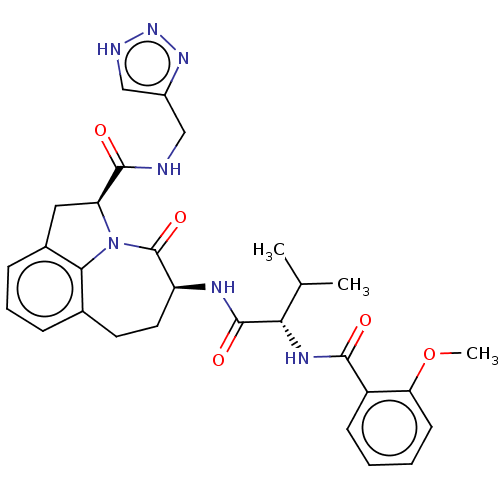
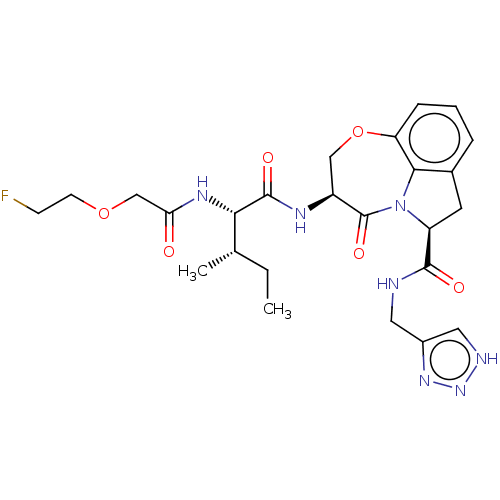
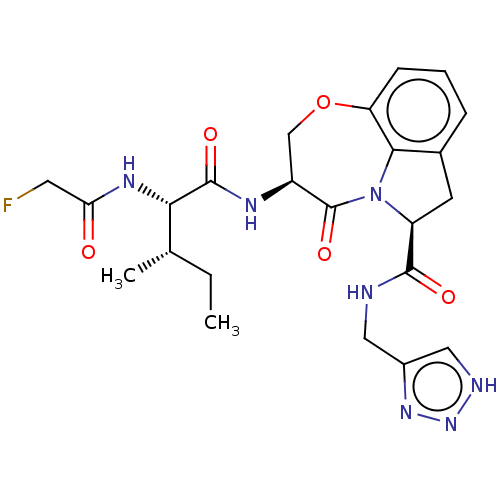
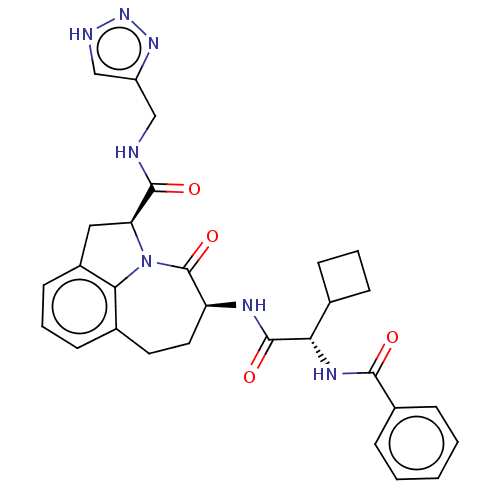
Affinity DataKi: 13nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
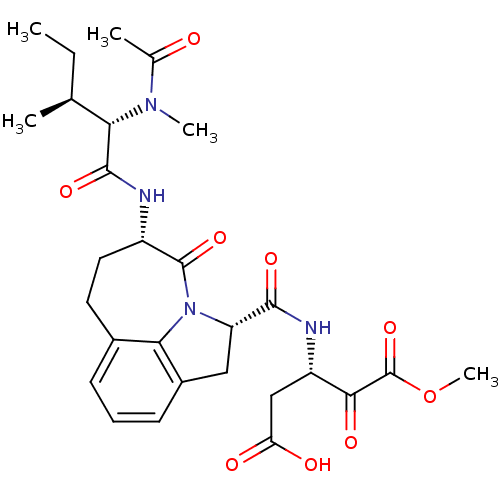
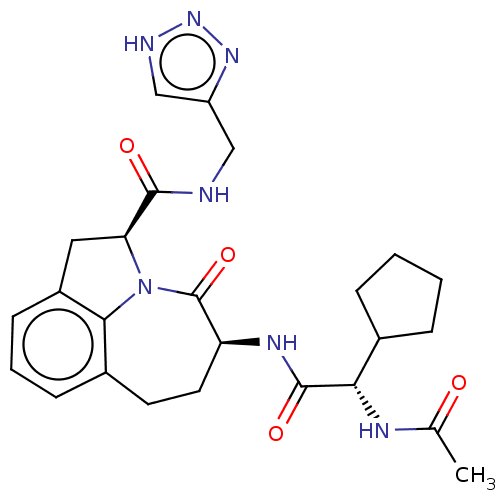
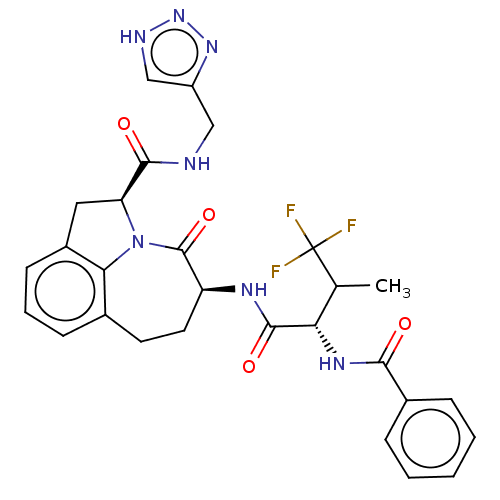
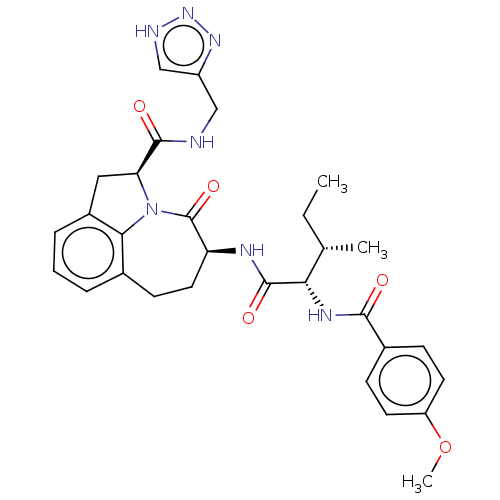
Affinity DataKi: 13nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
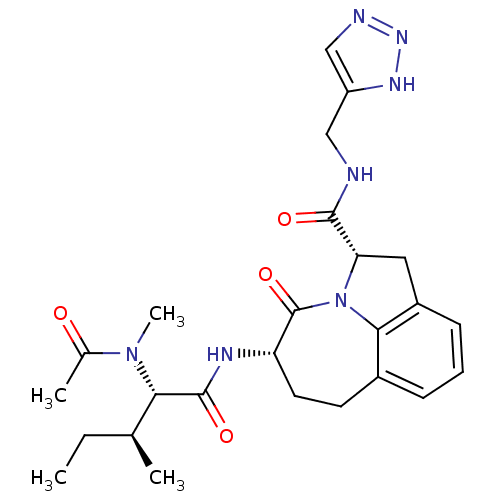
Affinity DataKi: 38nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
Affinity DataKi: 74nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
Affinity DataKi: 75nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
Affinity DataKi: 80nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
Affinity DataKi: 85nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
Affinity DataKi: 85nMAssay Description:Inhibition of granzyme BMore data for this Ligand-Target Pair
Affinity DataKi: 122nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
Affinity DataKi: 133nMAssay Description:Inhibition against human granzyme BMore data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Buffer pH7.4: Prepare 1 (M) KH2PO4 and 1 (M) K2HPO4. Titrate 1(M) K2HPO4 with 1 (M) KH2PO4 to obtain pH 7.40. Dilute this buffer 10 fold in Water (30...More data for this Ligand-Target Pair