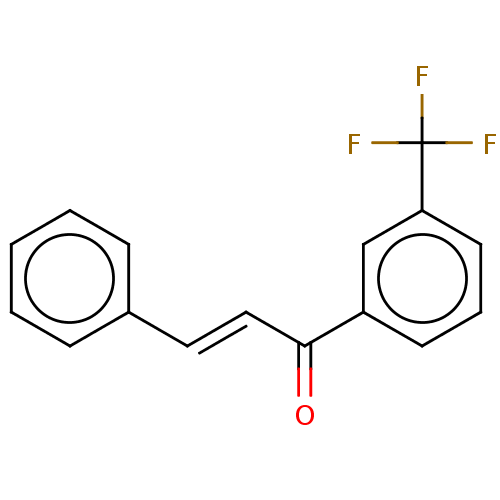
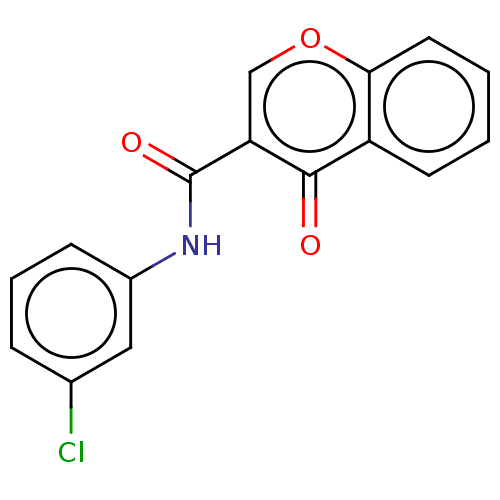
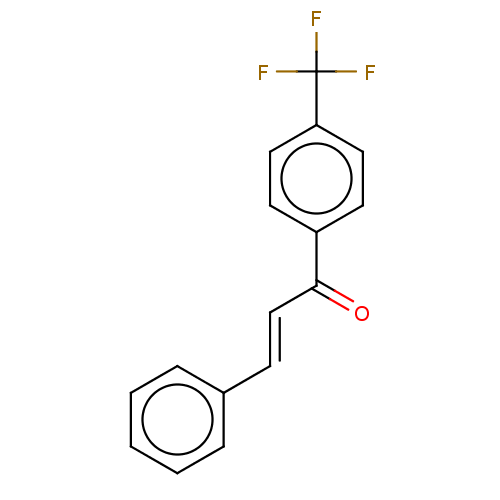
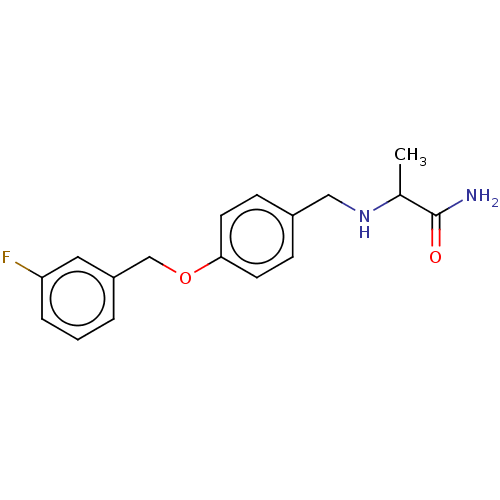
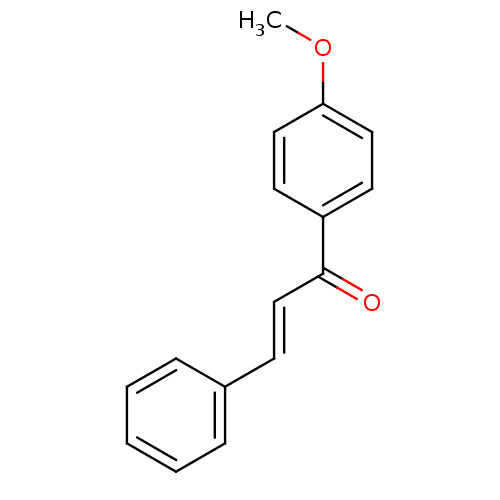
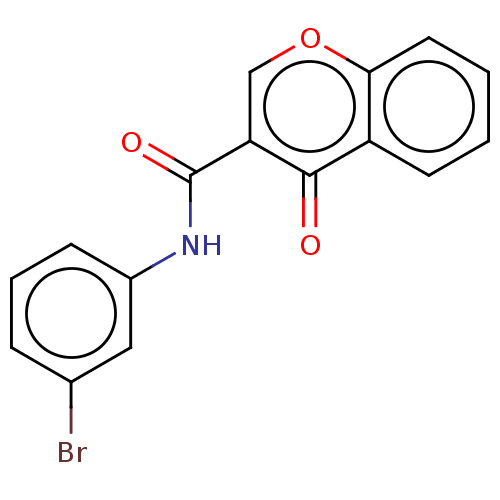
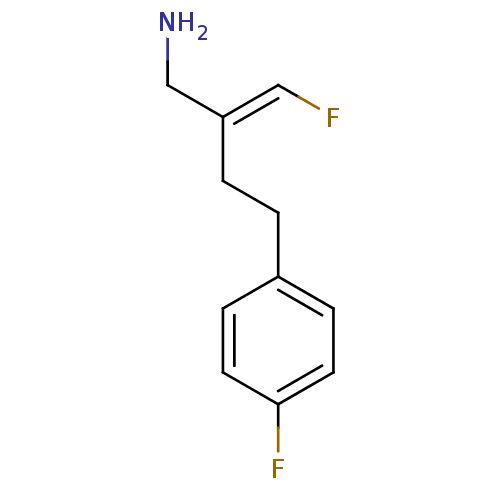
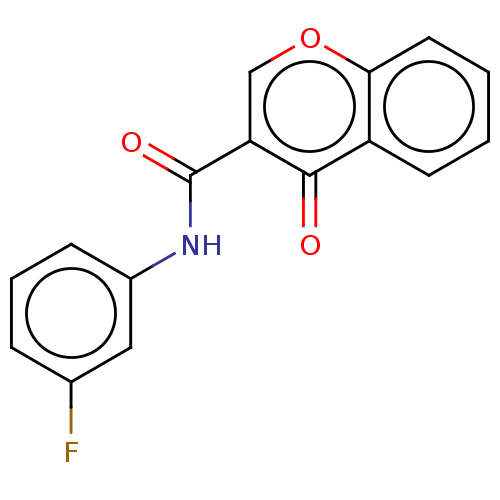
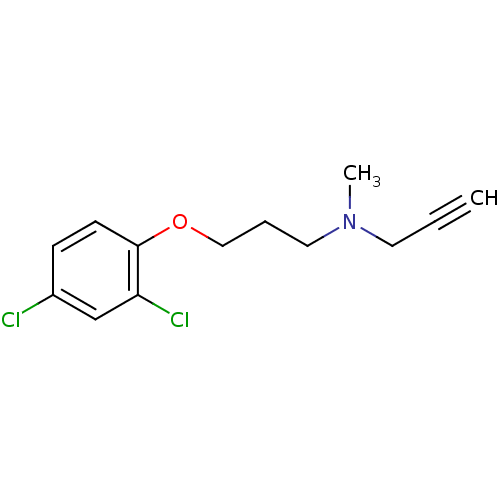
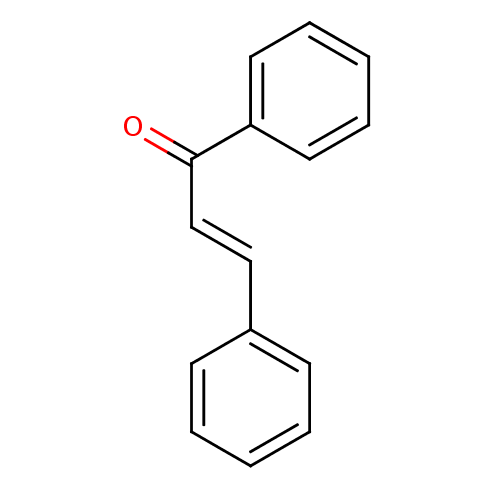
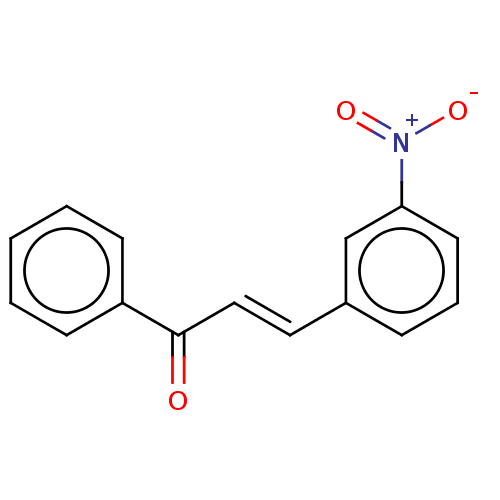
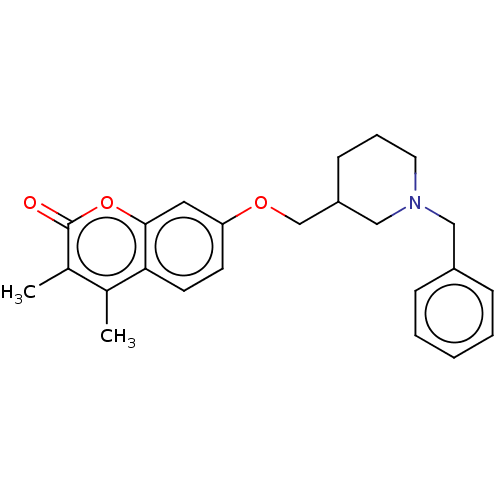
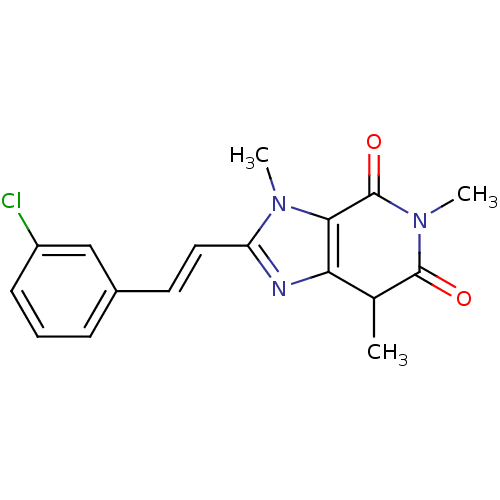
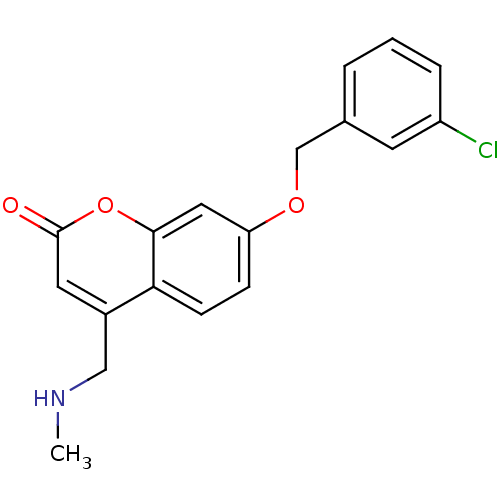
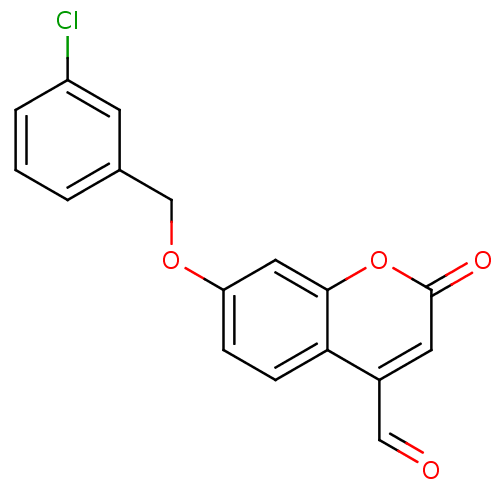
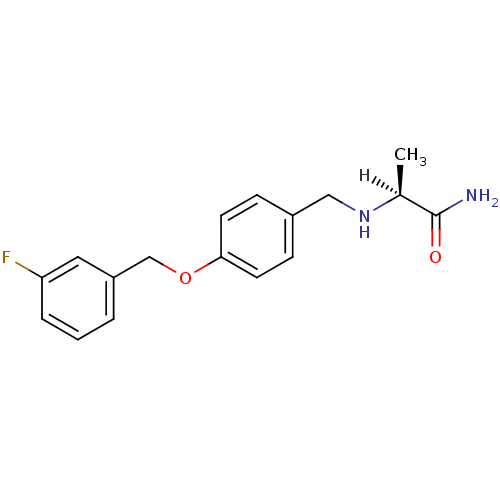
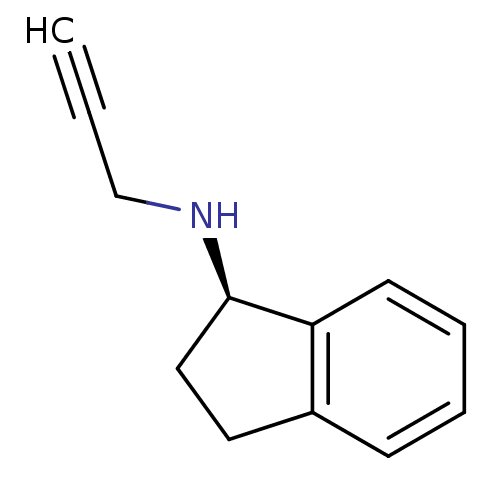
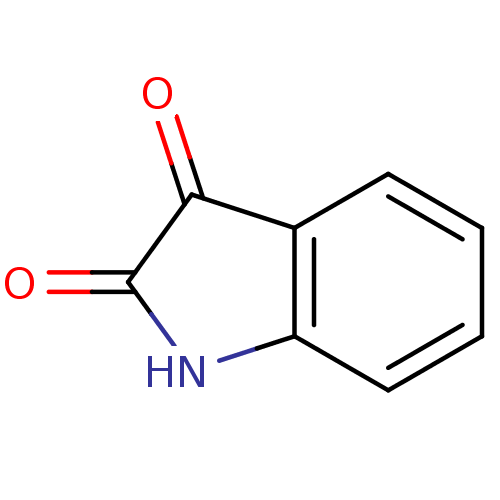
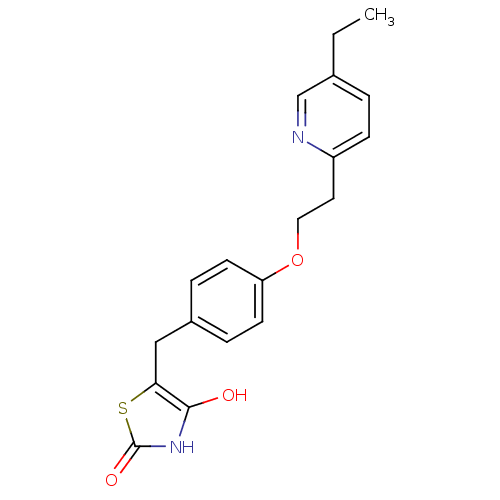
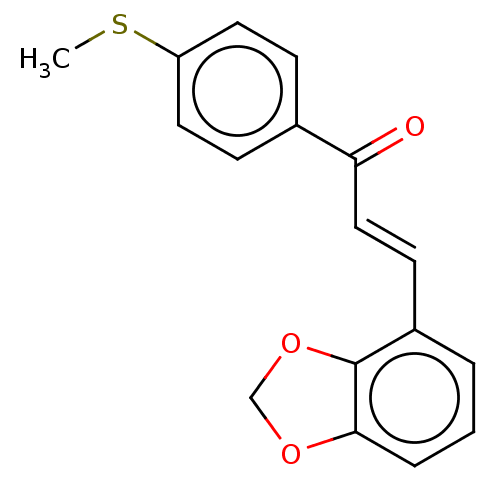
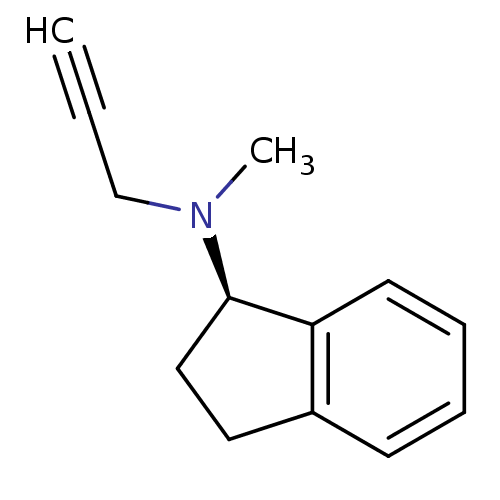
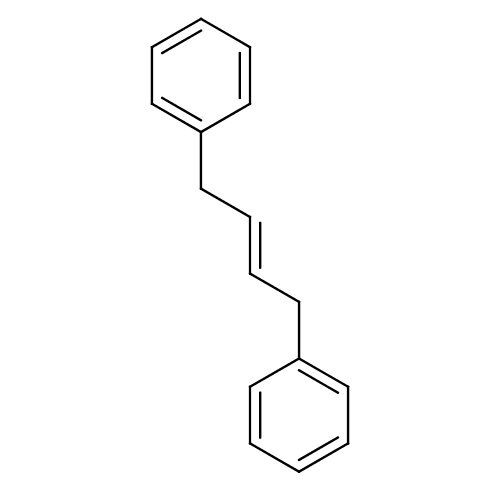
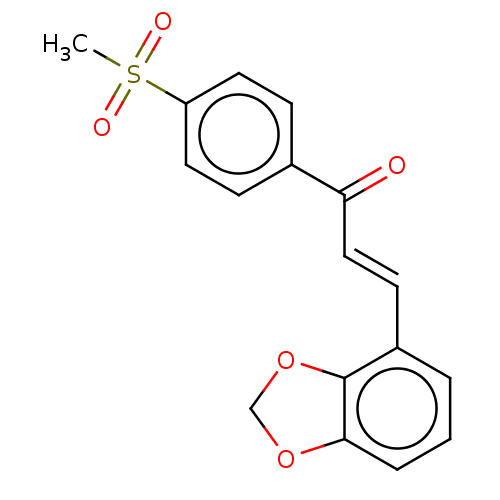
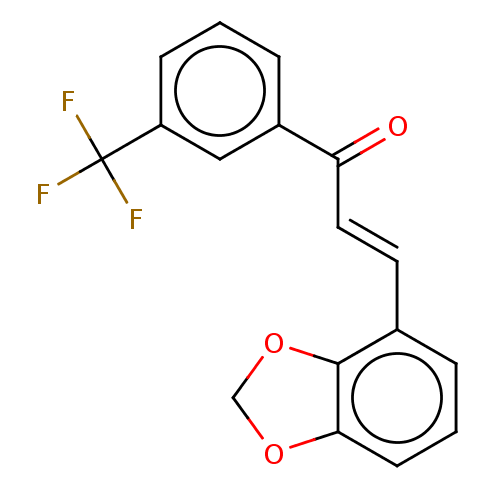
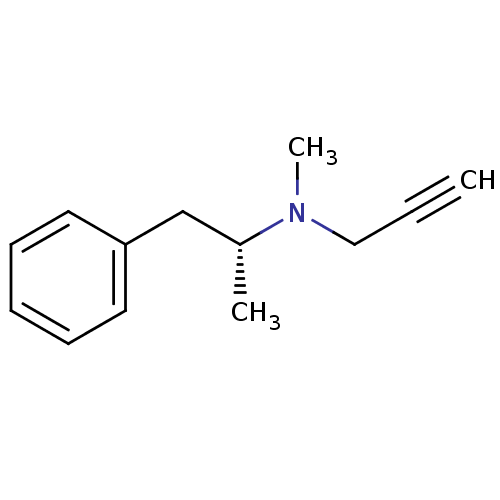
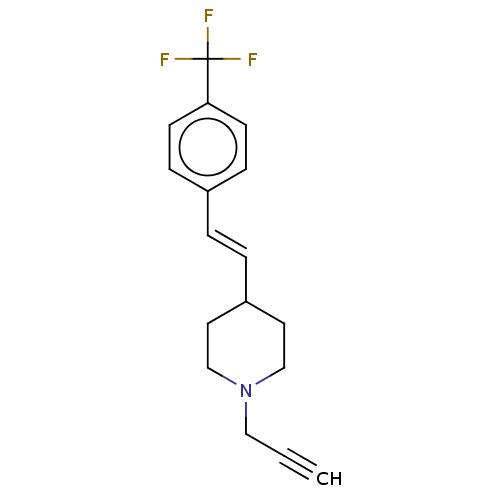
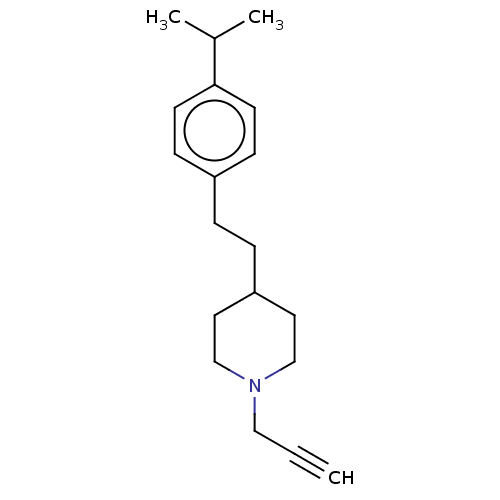
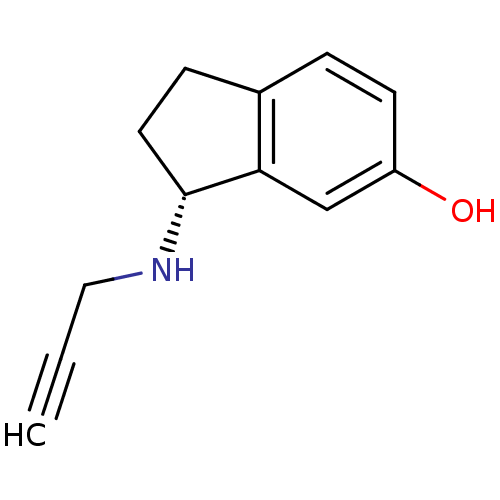
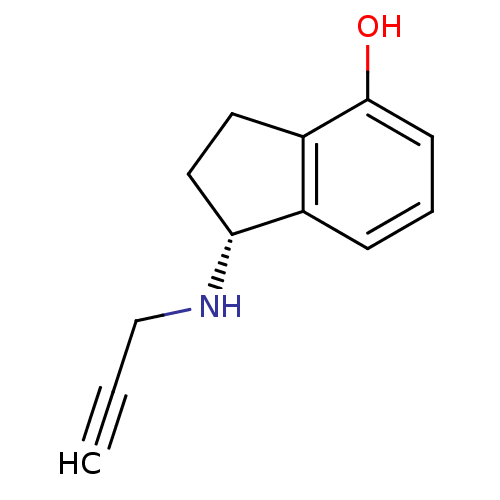
Affinity DataKi: 0.310nMAssay Description:Mixed-type inhibition of recombinant human MAO-B expressed in Pichia pastoris using benzylamine as substrate by fluorimetric horseradish peroxidase-A...More data for this Ligand-Target Pair
Affinity DataKi: 5nMAssay Description:Inhibition of recombinant human MAO-B expressed in baculovirus infected BTI cells assessed as inhibition of H2O2 production using kynuramine as subst...More data for this Ligand-Target Pair
Affinity DataKi: 7.5nMAssay Description:Non-competitive inhibition of recombinant human MAO-B expressed in Pichia pastoris using benzylamine as substrate by fluorimetric horseradish peroxid...More data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:Inhibition of recombinant human MAO-B expressed in baculovirus infected BTI cells assessed as inhibition of H2O2 production using kynuramine as subst...More data for this Ligand-Target Pair
Affinity DataKi: 17nMAssay Description:Inhibition of recombinant human MAO-B expressed in baculovirus infected BTI cells assessed as inhibition of H2O2 production using kynuramine as subst...More data for this Ligand-Target Pair
Affinity DataKi: 17nMAssay Description:Tight-binding competitive inhibition of recombinant human MAO-B expressed in Pichia pastoris using benzylamine as substrate by fluorimetric horseradi...More data for this Ligand-Target Pair
Affinity DataKi: 22nMAssay Description:Inhibition of recombinant human MAO-B expressed in baculovirus infected BTI cells assessed as inhibition of H2O2 production using kynuramine as subst...More data for this Ligand-Target Pair
Affinity DataKi: 27nMAssay Description:Tight-binding competitive inhibition of recombinant human MAO-B expressed in Pichia pastoris using benzylamine as substrate by fluorimetric horseradi...More data for this Ligand-Target Pair
Affinity DataKi: 28nMAssay Description:Inhibition of human recombinant MAOB expressed in Pichia pastoris by kinetic assayMore data for this Ligand-Target Pair
Affinity DataKi: 31nMAssay Description:Tight-binding competitive inhibition of recombinant human MAO-B expressed in Pichia pastoris using benzylamine as substrate by fluorimetric horseradi...More data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
University of Ljubljana
Curated by ChEMBL
University of Ljubljana
Curated by ChEMBL
Affinity DataKi: 40nMAssay Description:Reversible inhibition of recombinant human MAOA expressed in Pichia pastoris using varying levels of kynuramine as substrate measured after 5 mins by...More data for this Ligand-Target Pair
Affinity DataKi: 55nMAssay Description:Tight-binding competitive inhibition of recombinant human MAO-B expressed in Pichia pastoris using benzylamine as substrate by fluorimetric horseradi...More data for this Ligand-Target Pair
Affinity DataKi: 56nMAssay Description:Inhibition of recombinant human MAO-B expressed in baculovirus infected BTI cells assessed as inhibition of H2O2 production using kynuramine as subst...More data for this Ligand-Target Pair
Affinity DataKi: 71nMAssay Description:Inhibition of recombinant human MAO-B expressed in baculovirus infected BTI cells assessed as inhibition of H2O2 production using kynuramine as subst...More data for this Ligand-Target Pair
Affinity DataKi: 93nMAssay Description:Binding affinity to recombinant human MAO-B expressed in Pichia pastoris assessed as inhibition constant using benzylamine as substrate by Michaelis-...More data for this Ligand-Target Pair
Affinity DataKi: 100nM ΔG°: -40.0kJ/molepH: 7.4 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically. Competitive Ki values for both enzymes were determined by measuring initial rates ...More data for this Ligand-Target Pair
Affinity DataKi: 100nM ΔG°: -40.0kJ/molepH: 7.5 T: 2°CAssay Description:MAO B activities were determined spectrophotometrically at 250 nm using benzylamine as substrate. Competitive Ki values were determined by measuring ...More data for this Ligand-Target Pair
Affinity DataKi: 130nMAssay Description:Binding affinity to recombinant human MAO-B expressed in Pichia pastoris assessed as inhibition constant using benzylamine as substrate by Michaelis-...More data for this Ligand-Target Pair
Affinity DataKi: 190nMAssay Description:Binding affinity to recombinant human MAO-B expressed in Pichia pastoris assessed as inhibition constant using benzylamine as substrate by Michaelis-...More data for this Ligand-Target Pair
Affinity DataKi: 200nM ΔG°: -38.2kJ/molepH: 7.4 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically. Competitive Ki values for both enzymes were determined by measuring initial rates ...More data for this Ligand-Target Pair
Affinity DataKi: 200nM ΔG°: -38.2kJ/molepH: 7.5 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically. Competitive Ki values for both enzymes were determined by measuring initial rates ...More data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
University of Ljubljana
Curated by ChEMBL
University of Ljubljana
Curated by ChEMBL
Affinity DataKi: 220nMAssay Description:Irreversible inhibition of recombinant human MAOA expressed in Pichia pastoris using varying levels of kynuramine as substrate measured after 5 mins ...More data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
University of Ljubljana
Curated by ChEMBL
University of Ljubljana
Curated by ChEMBL
Affinity DataKi: 230nMAssay Description:Reversible inhibition of recombinant human MAOA expressed in Pichia pastoris using varying levels of kynuramine as substrate measured after 5 mins by...More data for this Ligand-Target Pair
Affinity DataKi: 400nMAssay Description:Inhibition of recombinant human MAO-B expressed in baculovirus infected BTI cells assessed as inhibition of H2O2 production using kynuramine as subst...More data for this Ligand-Target Pair
Affinity DataKi: 400nM ΔG°: -36.5kJ/molepH: 7.5 T: 2°CAssay Description:MAO B activities were determined spectrophotometrically at 250 nm using benzylamine as substrate. Competitive Ki values were determined by measuring ...More data for this Ligand-Target Pair
Affinity DataKi: 450nM ΔG°: -36.2kJ/molepH: 7.5 T: 2°CAssay Description:MAO B activities were determined spectrophotometrically at 250 nm using benzylamine as substrate. Competitive Ki values were determined by measuring ...More data for this Ligand-Target Pair
Affinity DataKi: 500nMAssay Description:MAO A and MAO B activities were determined spectrophotometrically. Competitive Ki values for both enzymes were determined by measuring initial rates ...More data for this Ligand-Target Pair
Affinity DataKi: 500nMAssay Description:MAO A and MAO B activities were determined spectrophotometrically. Competitive Ki values for both enzymes were determined by measuring initial rates ...More data for this Ligand-Target Pair
Affinity DataKi: 500nMAssay Description:Competitive inhibition of human MAOB expressed in Pichia pastorisMore data for this Ligand-Target Pair
Affinity DataKi: 550nMAssay Description:Competitive inhibition of human MAOB overexpressed in Pichia pastoris using MMTP as substrate preincubated for 5 mins followed by substrate addition ...More data for this Ligand-Target Pair
Affinity DataKi: 600nM ΔG°: -35.5kJ/molepH: 7.5 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically at 316 nm and 250 nm using kynuramine and benzylamine as substrates, respectively. ...More data for this Ligand-Target Pair
Affinity DataKi: 700nM ΔG°: -35.1kJ/molepH: 7.5 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically at 316 nm and 250 nm using kynuramine and benzylamine as substrates, respectively. ...More data for this Ligand-Target Pair
Affinity DataKi: 700nM ΔG°: -35.1kJ/molepH: 7.5 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically. Competitive Ki values for both enzymes were determined by measuring initial rates ...More data for this Ligand-Target Pair
Affinity DataKi: 700nM ΔG°: -35.1kJ/molepH: 7.5 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically at 316 nm and 250 nm using kynuramine and benzylamine as substrates, respectively. ...More data for this Ligand-Target Pair
Affinity DataKi: 720nMAssay Description:Competitive inhibition of human MAOB overexpressed in Pichia pastoris using MMTP as substrate preincubated for 5 mins followed by substrate addition ...More data for this Ligand-Target Pair
Affinity DataKi: 780nMAssay Description:Competitive inhibition of human MAOB overexpressed in Pichia pastoris using MMTP as substrate preincubated for 5 mins followed by substrate addition ...More data for this Ligand-Target Pair
Affinity DataKi: 800nM ΔG°: -34.8kJ/molepH: 7.4 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically. Competitive Ki values for both enzymes were determined by measuring initial rates ...More data for this Ligand-Target Pair
Affinity DataKi: 800nM ΔG°: -34.8kJ/molepH: 7.4 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically. Competitive Ki values for both enzymes were determined by measuring initial rates ...More data for this Ligand-Target Pair
Affinity DataKi: 970nMAssay Description:Reversible inhibition of recombinant human MAOB expressed in Pichia pastoris using varying levels of kynuramine as substrate measured after 5 mins by...More data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
University of Ljubljana
Curated by ChEMBL
University of Ljubljana
Curated by ChEMBL
Affinity DataKi: 1.01E+3nMAssay Description:Irreversible inhibition of recombinant human MAOA expressed in Pichia pastoris using varying levels of kynuramine as substrate measured after 5 mins ...More data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
University of Ljubljana
Curated by ChEMBL
University of Ljubljana
Curated by ChEMBL
Affinity DataKi: 1.10E+3nMAssay Description:Inhibition of human recombinant MAOA expressed in Pichia pastoris by kinetic assayMore data for this Ligand-Target Pair
Affinity DataKi: 1.10E+3nM ΔG°: -34.0kJ/molepH: 7.4 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically. Competitive Ki values for both enzymes were determined by measuring initial rates ...More data for this Ligand-Target Pair
Affinity DataKi: 1.15E+3nMAssay Description:Competitive inhibition of human MAOB overexpressed in Pichia pastoris using MMTP as substrate preincubated for 5 mins followed by substrate addition ...More data for this Ligand-Target Pair
Affinity DataKi: 1.25E+3nMAssay Description:Reversible inhibition of recombinant human MAOB expressed in Pichia pastoris using varying levels of kynuramine as substrate measured after 5 mins by...More data for this Ligand-Target Pair
Affinity DataKi: 1.36E+3nMAssay Description:Reversible inhibition of recombinant human MAOB expressed in Pichia pastoris using varying levels of kynuramine as substrate measured after 5 mins by...More data for this Ligand-Target Pair
Affinity DataKi: 1.38E+3nMAssay Description:Irreversible inhibition of recombinant human MAOB expressed in Pichia pastoris using varying levels of kynuramine as substrate measured after 5 mins ...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+3nM ΔG°: -33.4kJ/molepH: 7.4 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically. Competitive Ki values for both enzymes were determined by measuring initial rates ...More data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
University of Ljubljana
Curated by ChEMBL
University of Ljubljana
Curated by ChEMBL
Affinity DataKi: 1.90E+3nM ΔG°: -32.7kJ/molepH: 7.5 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically at 316 nm and 250 nm using kynuramine and benzylamine as substrates, respectively. ...More data for this Ligand-Target Pair
Affinity DataKi: 1.92E+3nMAssay Description:Reversible inhibition of recombinant human MAOB expressed in Pichia pastoris using varying levels of kynuramine as substrate measured after 5 mins by...More data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
University of Ljubljana
Curated by ChEMBL
University of Ljubljana
Curated by ChEMBL
Affinity DataKi: 2.00E+3nM ΔG°: -32.5kJ/molepH: 7.5 T: 2°CAssay Description:MAO A and MAO B activities were determined spectrophotometrically at 316 nm and 250 nm using kynuramine and benzylamine as substrates, respectively. ...More data for this Ligand-Target Pair

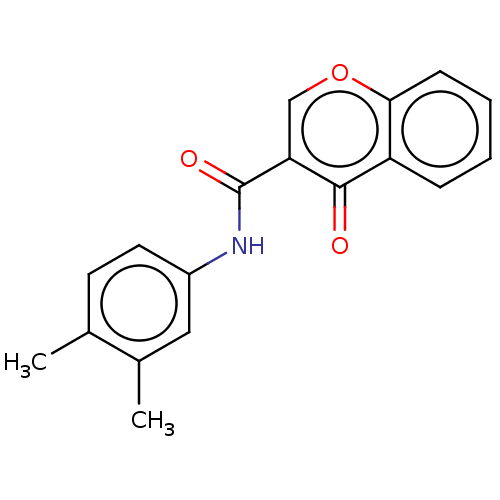
 3D Structure (crystal)
3D Structure (crystal)