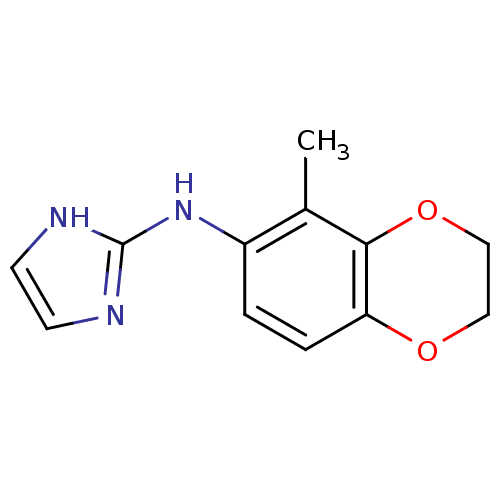
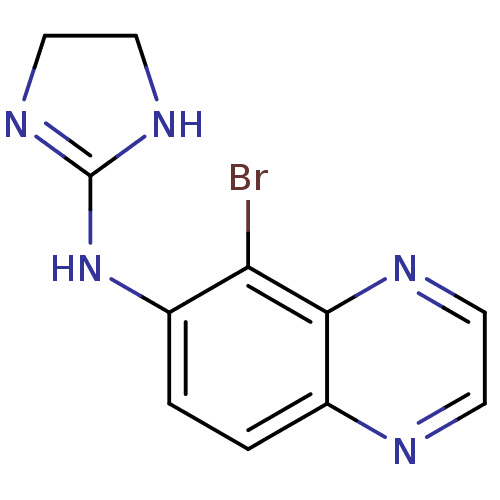
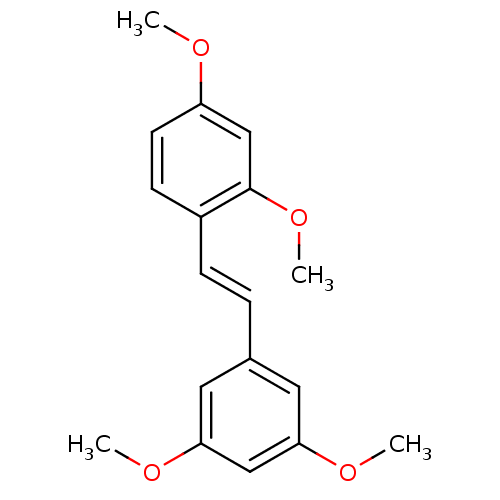
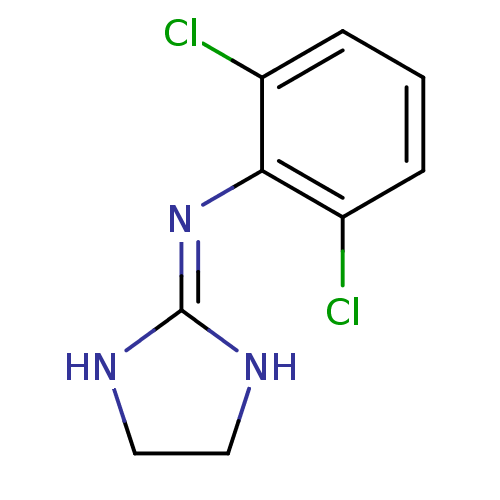
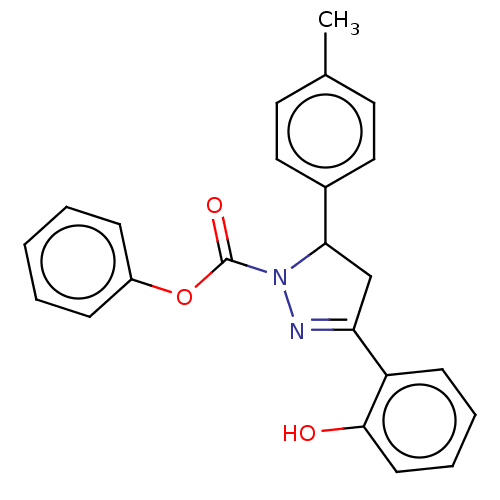
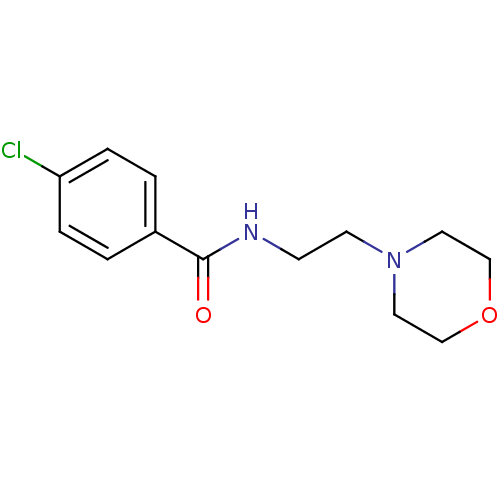
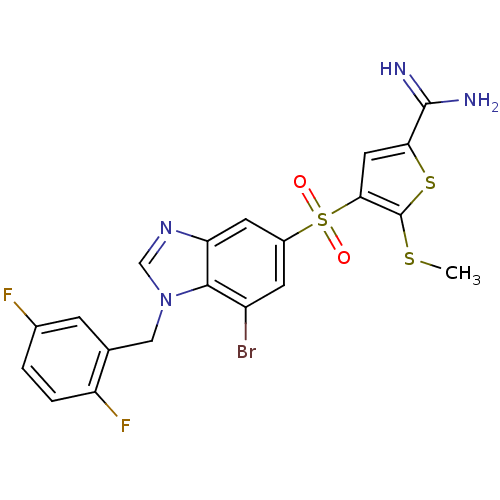
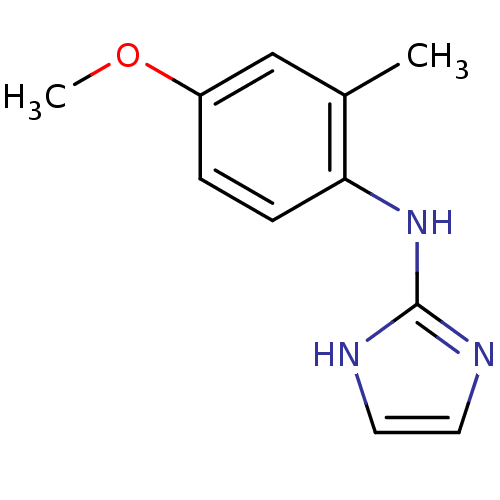
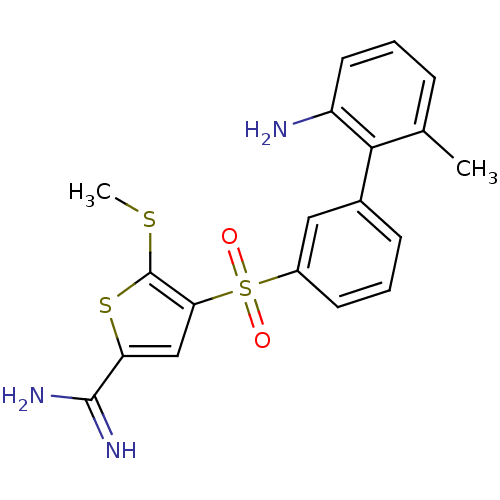
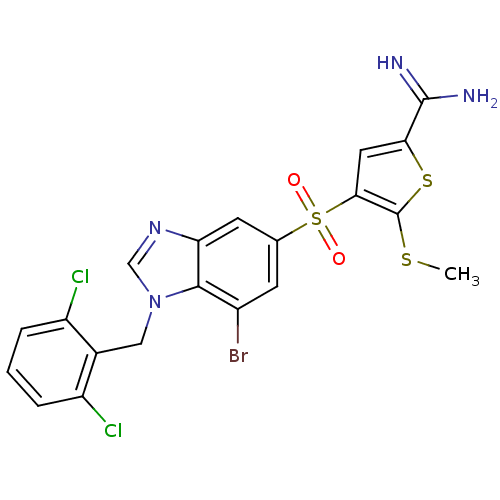
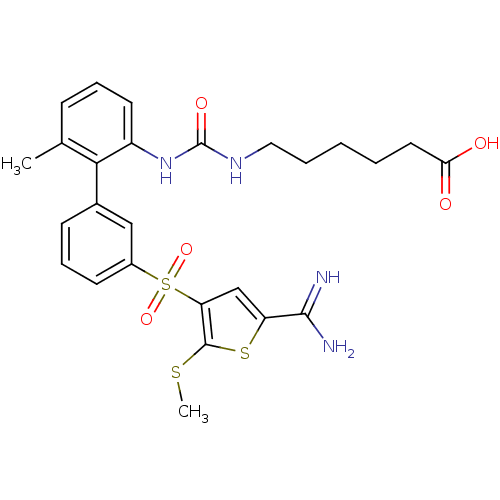
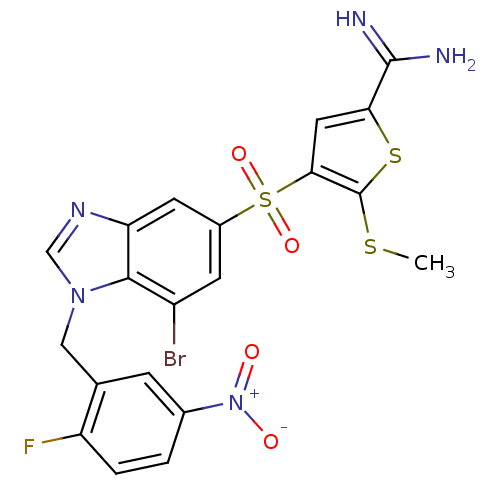
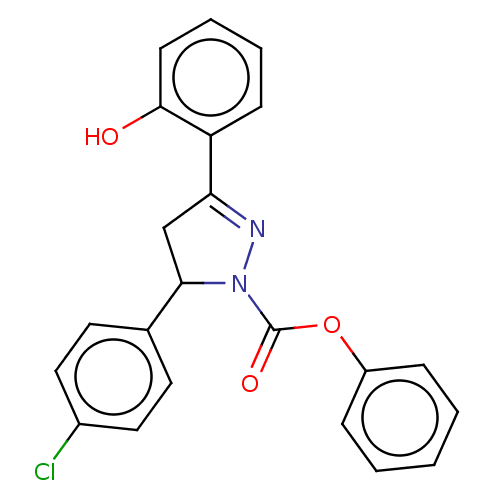
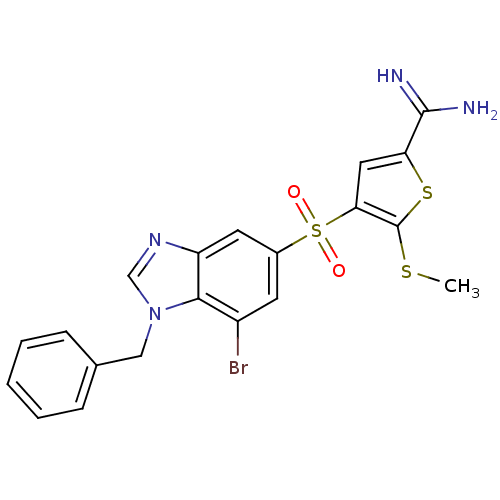
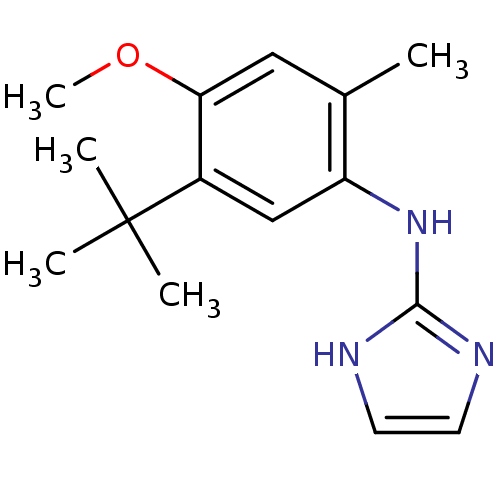
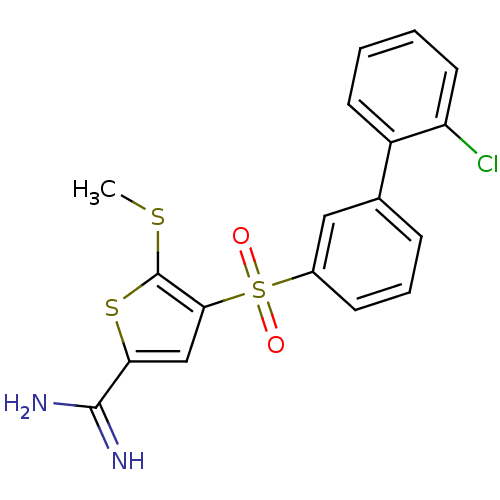
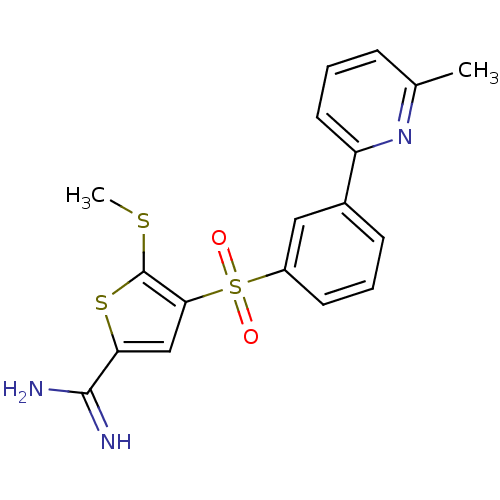
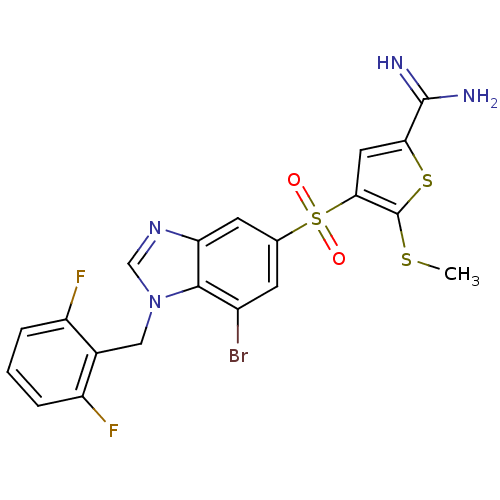
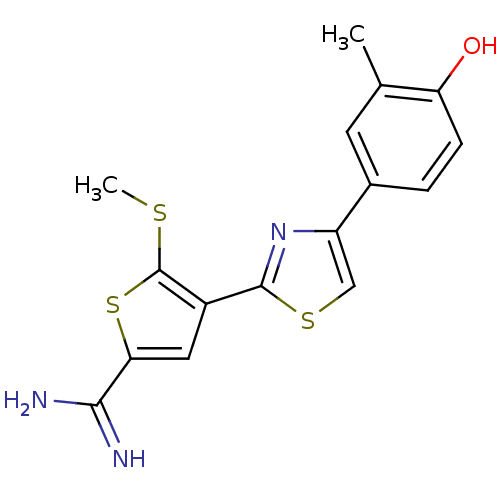
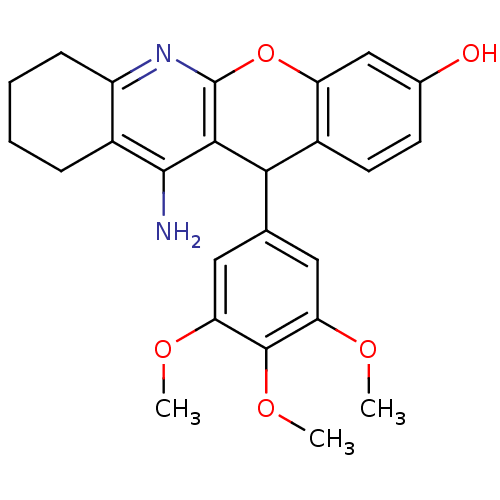
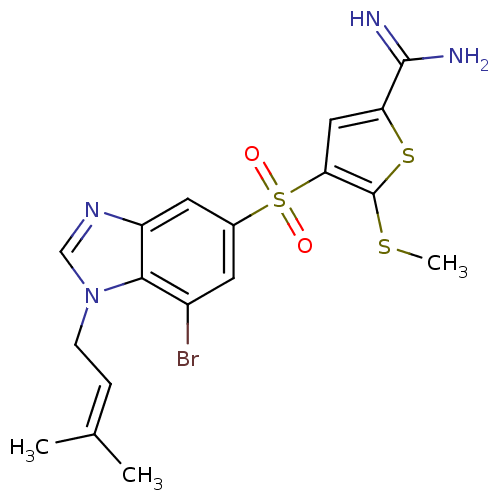
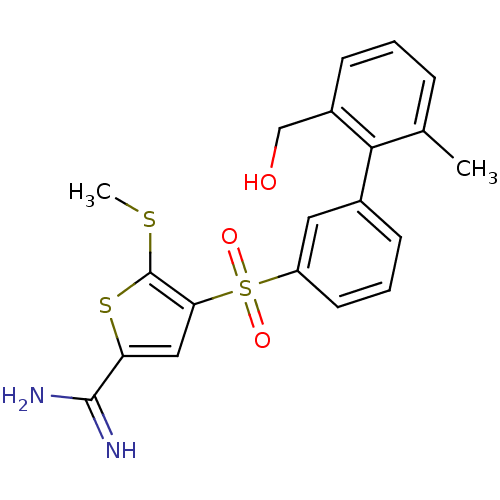
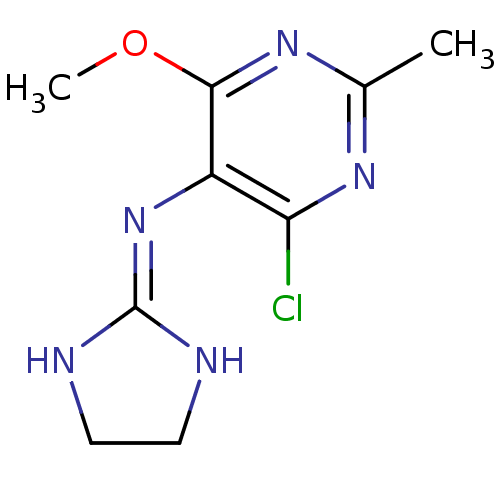
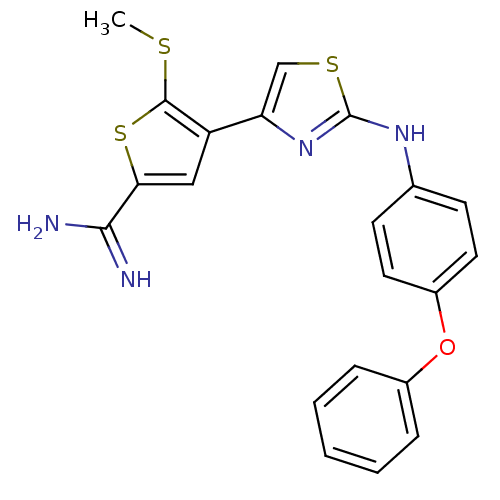
Affinity DataKi: 0.0900nMAssay Description:Inhibition of human GSK3betaMore data for this Ligand-Target Pair
Affinity DataKi: 0.700nMAssay Description:Inhibition of N-terminal GST-tagged human PARP2 (2 to 583 residues) expressed in baculovirus infected Sf9 cells using histone mixture (H2A and H2B) a...More data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Inhibition of N-terminal GST-tagged human full length PARP1 (2 to 1041 residues) expressed in baculovirus infected Sf9 cells using histone mixture (H...More data for this Ligand-Target Pair
Affinity DataKi: 1.70nMAssay Description:Displacement of rauwolscine from human Alpha-2A adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Displacement of rauwolscine from human Alpha-2A adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 2.70nMAssay Description:Displacement of rauwolscine from human Alpha-2A adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:Inhibition of human CYP1B1 using ethoxyresorufin as substrate preincubated for 3 mins followed by NADPH addition measured after 10 mins by EROD assayMore data for this Ligand-Target Pair
Affinity DataKi: 3.80nMAssay Description:Binding affinity for human Alpha-2A adrenergic receptorMore data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
Birla Institute Of Technology
Curated by ChEMBL
Birla Institute Of Technology
Curated by ChEMBL
Affinity DataKi: 4nMAssay Description:Competitive inhibition of human recombinant MAO-A expressed in baculovirus infected BT1 insect cells using p-tyramine as substrate by Lineweaver-Burk...More data for this Ligand-Target Pair
Affinity DataKi: 4.90nMAssay Description:Displacement of rauwolscine from human Alpha-2A adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Rattus norvegicus (rat))
Institute Of Technology
Curated by ChEMBL
Institute Of Technology
Curated by ChEMBL
Affinity DataKi: 5nMAssay Description:Inhibition of MAO-A in rat liver homogenate by spectrophotometry-based Holt methodMore data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
Birla Institute Of Technology
Curated by ChEMBL
Birla Institute Of Technology
Curated by ChEMBL
Affinity DataKi: 5nMAssay Description:Inhibition of human MAO-A expressed in BTI insect cells using p-tyramine as substrate after 60 minsMore data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
Birla Institute Of Technology
Curated by ChEMBL
Birla Institute Of Technology
Curated by ChEMBL
Affinity DataKi: 5nMAssay Description:Competitive reversible inhibition of human recombinant MAO-A expressed in baculovirus infected BT1 insect cells using p-tyramine as substrate assesse...More data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
Birla Institute Of Technology
Curated by ChEMBL
Birla Institute Of Technology
Curated by ChEMBL
Affinity DataKi: 5nMAssay Description:Competitive reversible inhibition of human recombinant MAO-A expressed in baculovirus infected BT1 insect cells using p-tyramine as substrate assesse...More data for this Ligand-Target Pair
Affinity DataKi: 6nMAssay Description:Displacement of rauwolscine from human Alpha-2A adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 8.30nMAssay Description:Binding affinity for rat Alpha-2B adrenergic receptorMore data for this Ligand-Target Pair
Affinity DataKi: 8.90nMAssay Description:Displacement of [3H]-clonidine from bovine imidazoline receptor I-1More data for this Ligand-Target Pair
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 10nMAssay Description:Displacement of rauwolscine from human Alpha-2A adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 14nMAssay Description:Inhibition of Complement C1s subcomponentMore data for this Ligand-Target Pair
Affinity DataKi: 17nMAssay Description:Displacement of rauwolscine from rat Alpha-2B adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 19nMAssay Description:Displacement of rauwolscine from human Alpha-2C adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 20nMAssay Description:Inhibition of Complement C1s subcomponentMore data for this Ligand-Target Pair
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 22nMAssay Description:Inhibition of Complement C1s subcomponentMore data for this Ligand-Target Pair
Affinity DataKi: 27nMAssay Description:Displacement of rauwolscine from human Alpha-2C adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 30nMAssay Description:Inhibition of Complement C1s subcomponentMore data for this Ligand-Target Pair
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 30nMAssay Description:Binding affinity for human Alpha-2C adrenergic receptorMore data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
Birla Institute Of Technology
Curated by ChEMBL
Birla Institute Of Technology
Curated by ChEMBL
Affinity DataKi: 30nMAssay Description:Competitive reversible inhibition of human recombinant MAO-A expressed in baculovirus infected BT1 insect cells using p-tyramine as substrate assesse...More data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
Birla Institute Of Technology
Curated by ChEMBL
Birla Institute Of Technology
Curated by ChEMBL
Affinity DataKi: 30nMAssay Description:The activities of recombinant hMAO-A and hMAO-B were determined using p-tyramine as common substrate and calculated as 0.18 +/- 0.01 nmol/mg/min (n =...More data for this Ligand-Target Pair
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 31nMAssay Description:Displacement of rauwolscine from human Alpha-2A adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 39nMAssay Description:Displacement of rauwolscine from human Alpha-2A adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 40nMAssay Description:Inhibition of Complement C1s subcomponentMore data for this Ligand-Target Pair
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 40nMAssay Description:Inhibition of Complement C1s subcomponentMore data for this Ligand-Target Pair
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 42nMAssay Description:Inhibition of Complement C1s subcomponentMore data for this Ligand-Target Pair
Affinity DataKi: 42nMAssay Description:Displacement of [3H]-clonidine from bovine imidazoline receptor I-1More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 44nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 44nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
Affinity DataKi: 44nMAssay Description:Displacement of rauwolscine from human Alpha-2C adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetAcetylcholinesterase(Electrophorus electricus (Electric eel))
Universidad Complutense De Madrid
Curated by ChEMBL
Universidad Complutense De Madrid
Curated by ChEMBL
Affinity DataKi: 47nMAssay Description:Noncompetitive inhibition of electric eel AchE using acetylcholine as substrate preincubated for 15 mins followed by substrate addition measured for ...More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 47nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
TargetComplement C1s subcomponent(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 50nMAssay Description:Inhibition of Complement C1s subcomponentMore data for this Ligand-Target Pair
Affinity DataKi: 52nMAssay Description:Displacement of rauwolscine from rat Alpha-2B adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 56nMAssay Description:Displacement of [3H]-clonidine from bovine imidazoline receptor I-1More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 58nMAssay Description:Inhibition of Human kidney cell urokinaseMore data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 60nMAssay Description:In vitro inhibitory activity against Urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair



 3D Structure (crystal)
3D Structure (crystal)