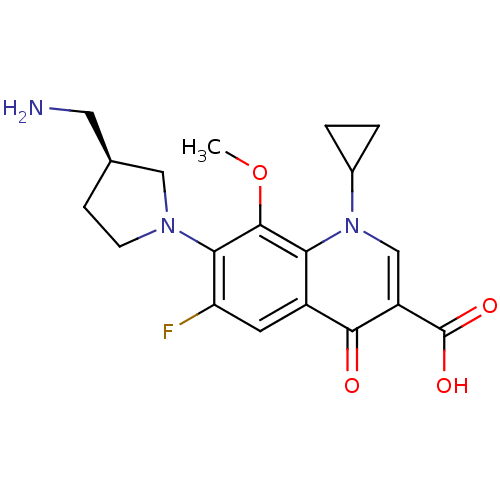
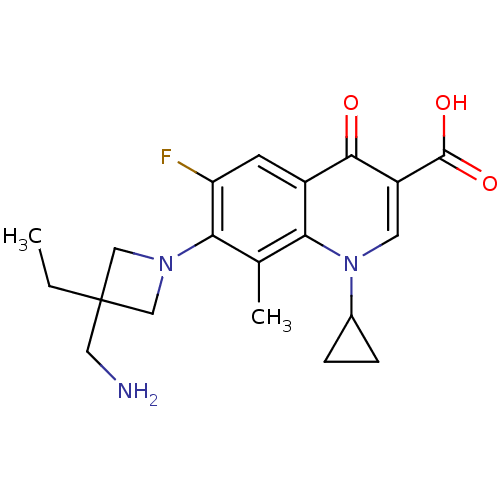
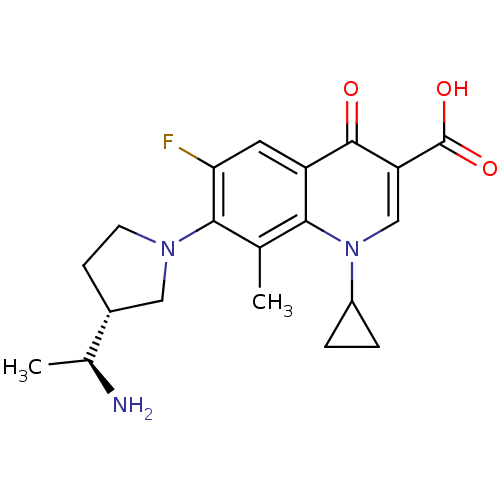
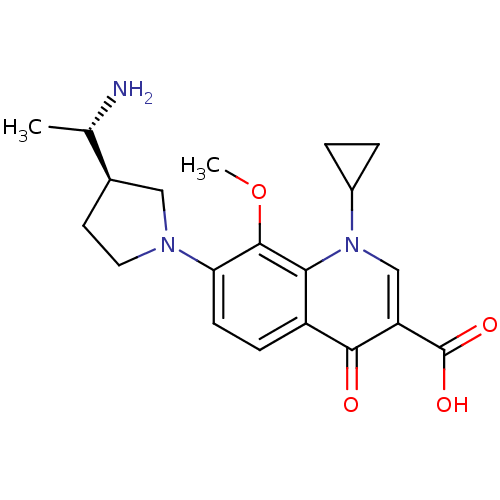
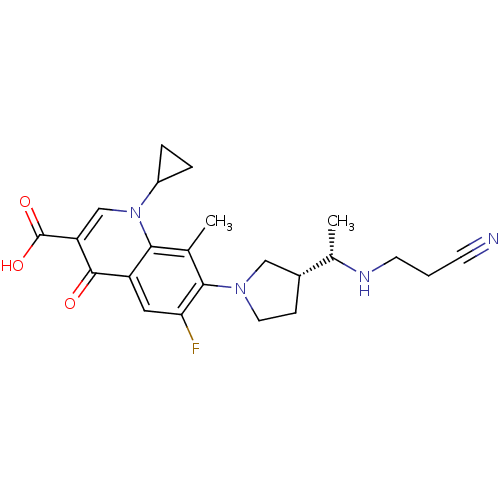
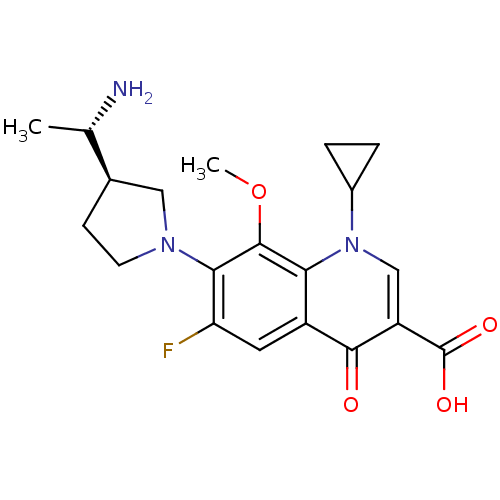
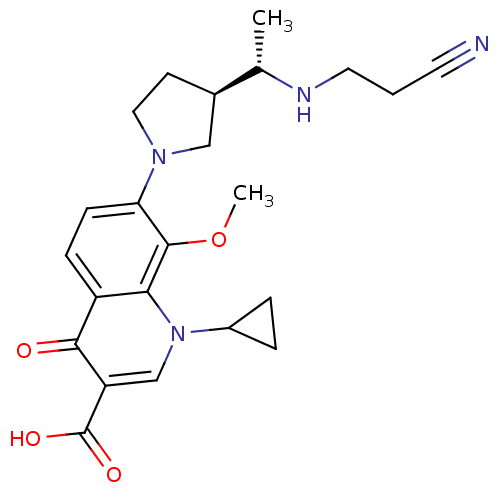
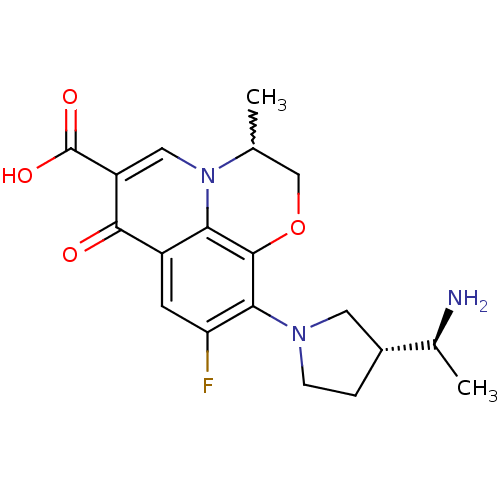
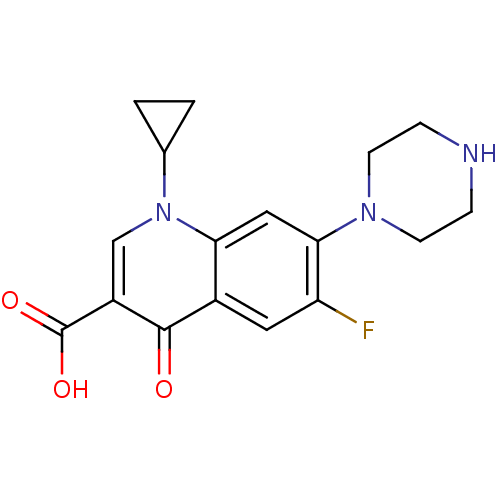
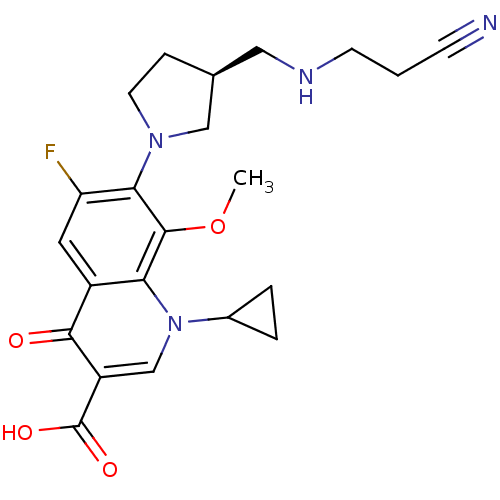
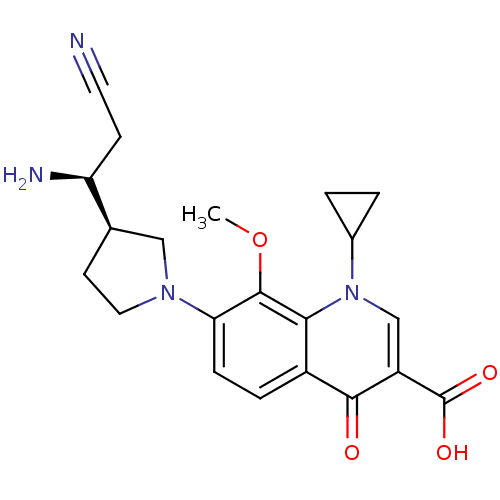
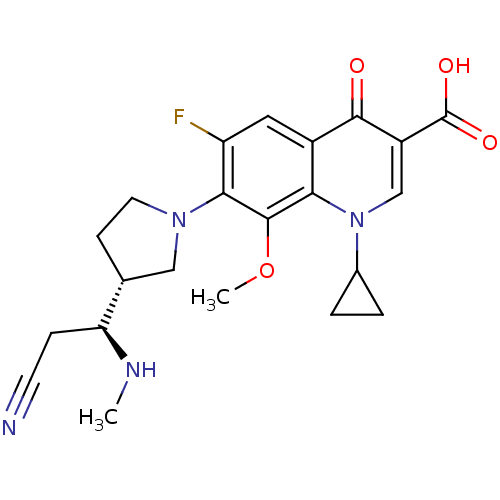
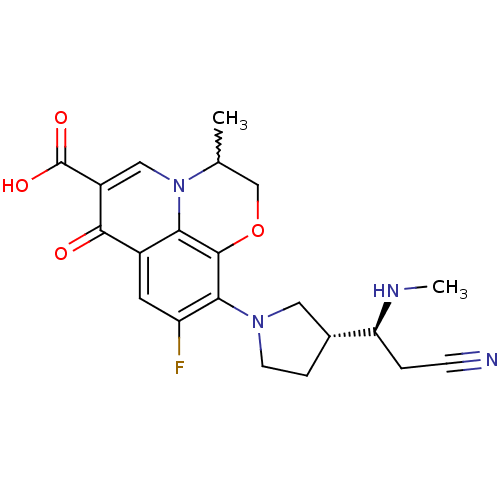
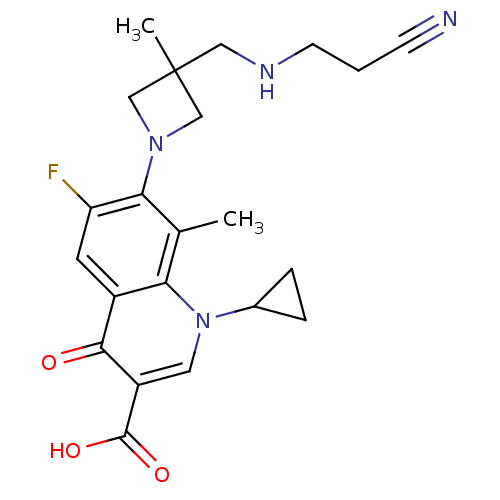
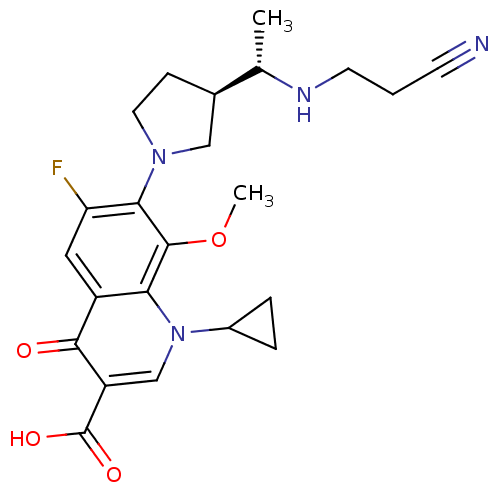
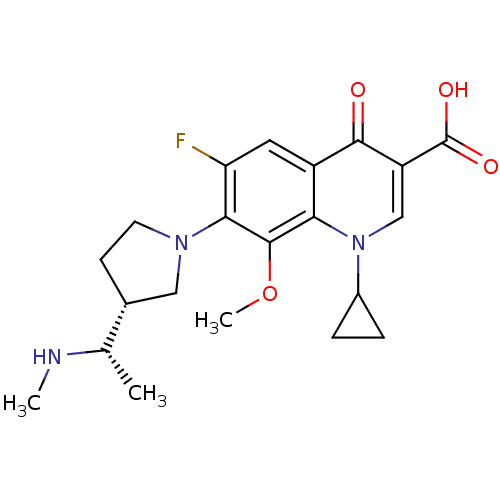
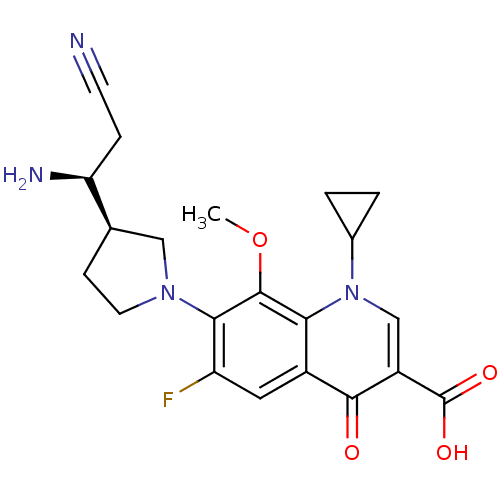
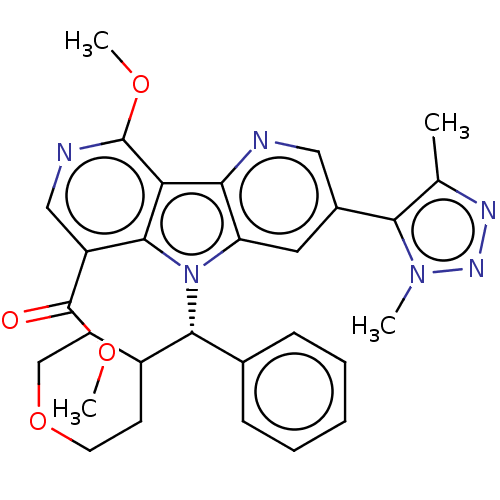
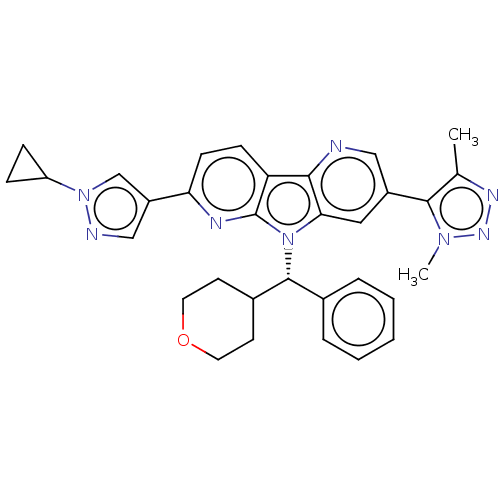
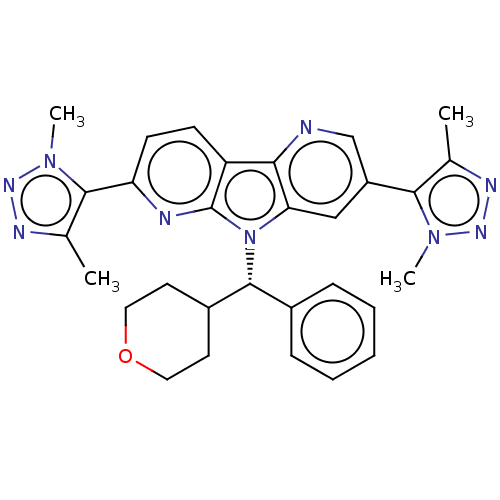
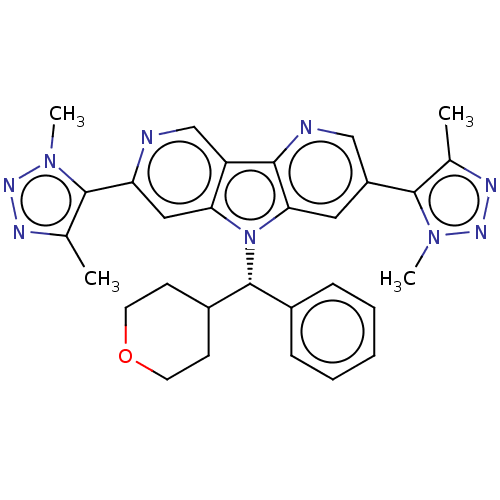
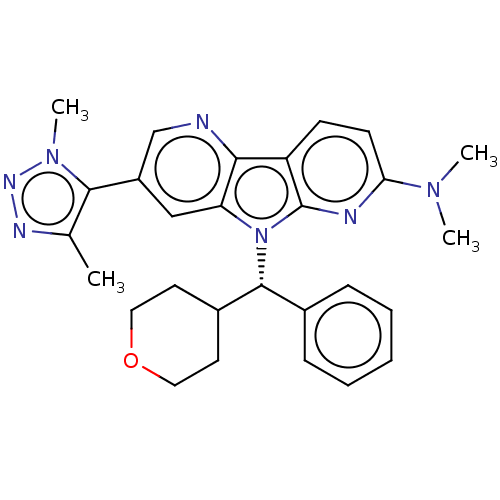
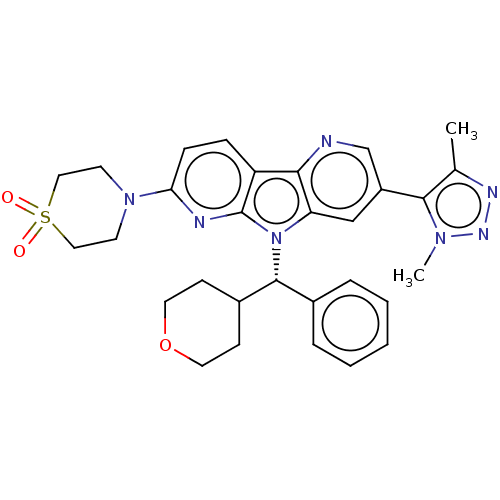
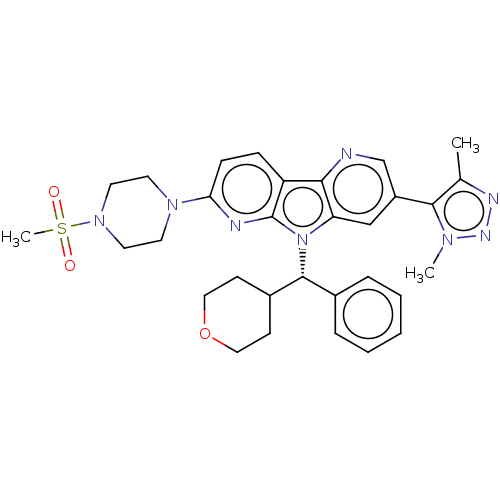
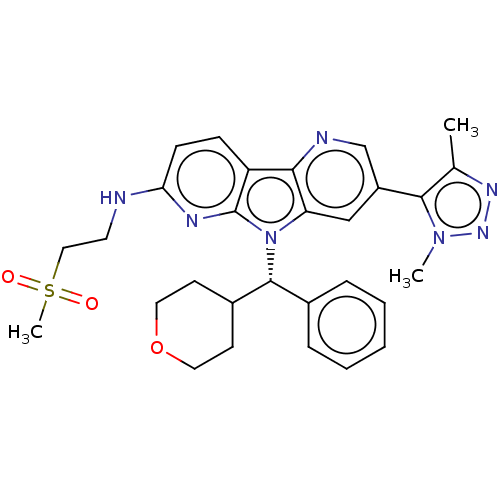
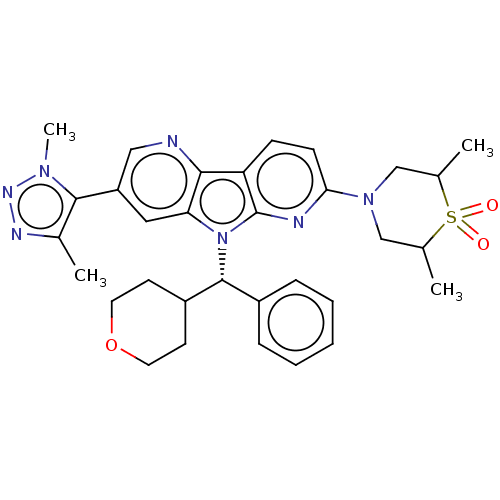
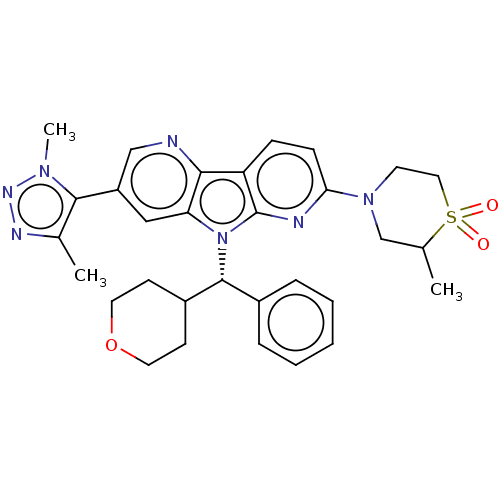
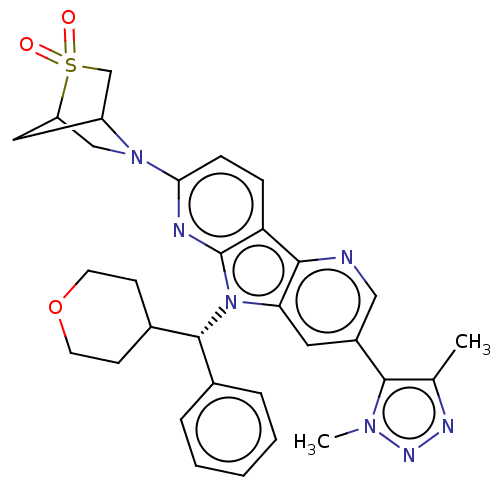
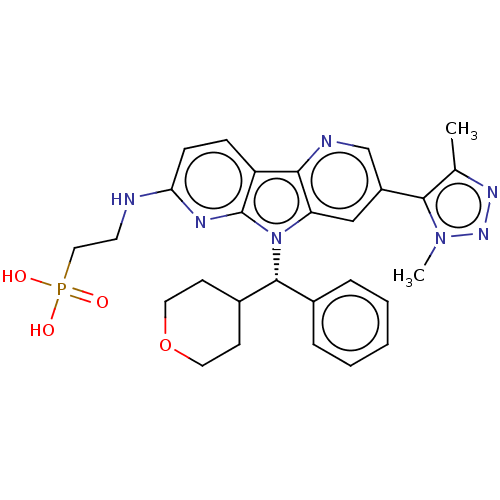
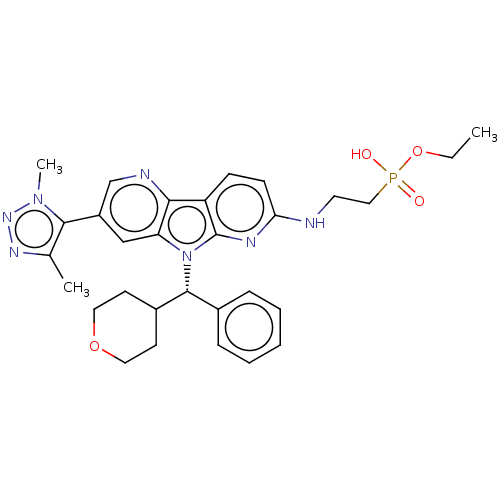
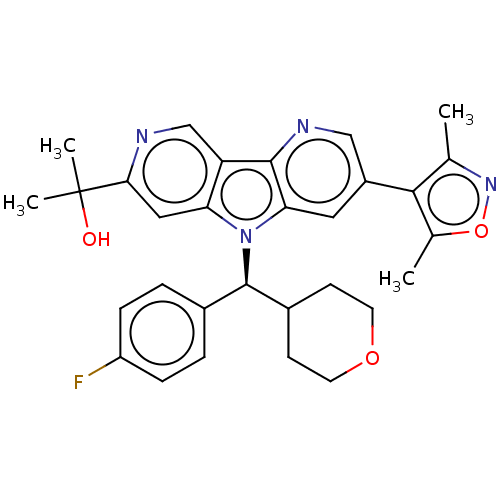
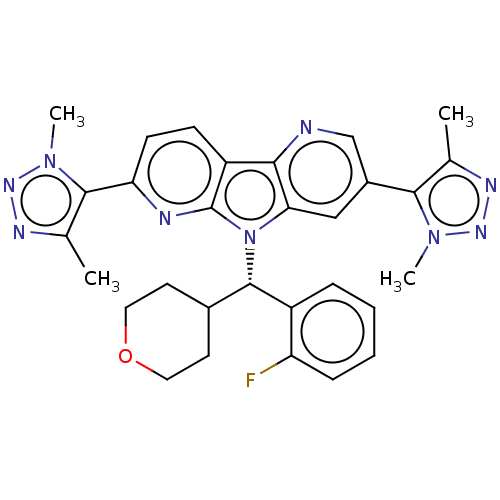
Affinity DataKi: 1.00E+3nMAssay Description:Inhibition of Mycobacterium tuberculosis thymidylate kinaseMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: 2.40E+4nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: 4.50E+4nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: 5.10E+4nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: 6.20E+4nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: 6.40E+4nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: 7.20E+4nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: 8.50E+4nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.00E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.00E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.00E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.00E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: 1.06E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: 1.12E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.50E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.50E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.50E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.50E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.50E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.50E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.50E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.50E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.50E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.50E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: >1.50E+5nMAssay Description:Displacement of [3H]dofetilide from human ERG by fliter binding assayMore data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair
TargetBromodomain-containing protein 4 [1-477]/[333-460]/[44-168]/[44-460](Homo sapiens (Human))
Bristol-Myers Squibb
US Patent
Bristol-Myers Squibb
US Patent
Affinity DataIC50: <1nMAssay Description:The binding of compounds to bromodomain BRD4 (44-168), BRD4 (333-460), and BRD4 (1-477 or 44-460) was assessed using a time resolved fluorescent reso...More data for this Ligand-Target Pair