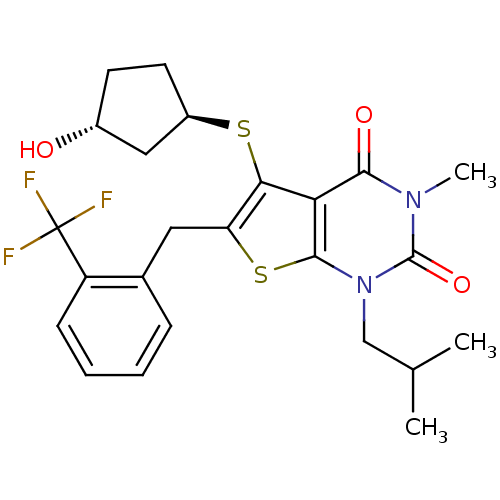
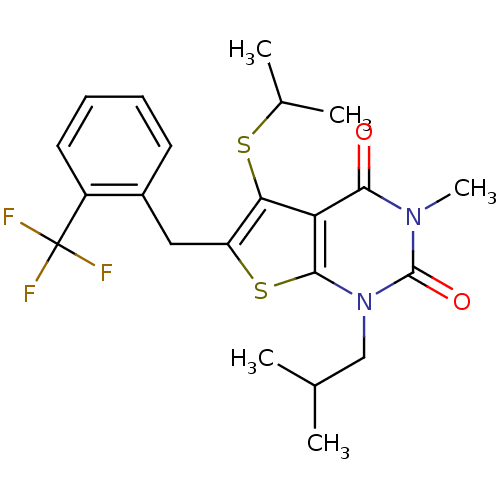
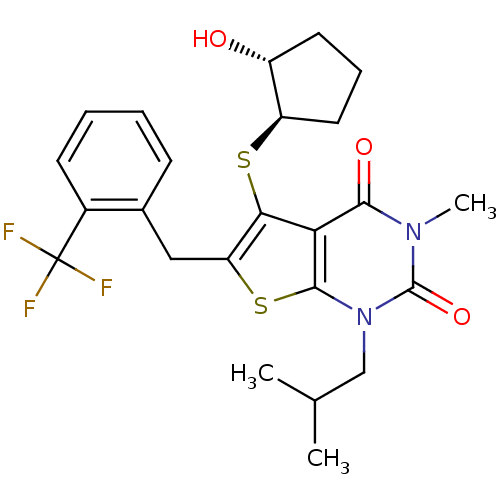
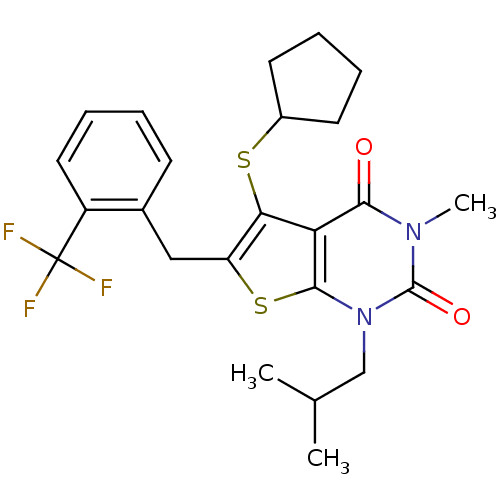
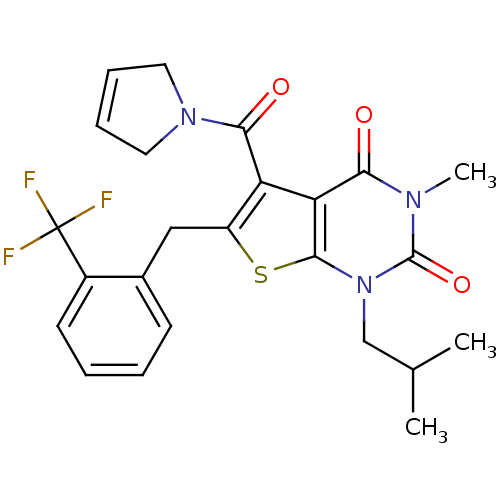
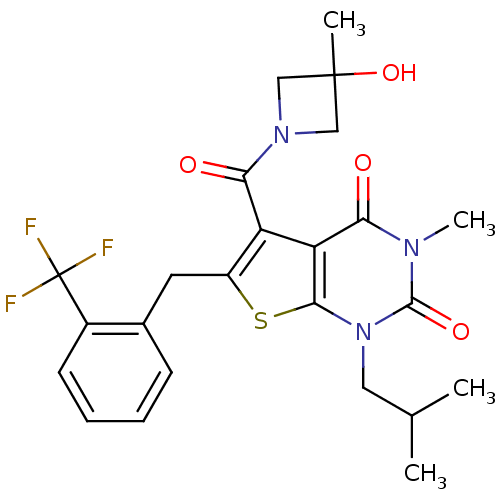
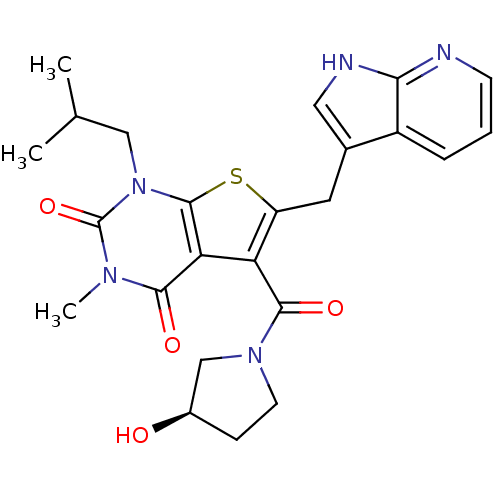
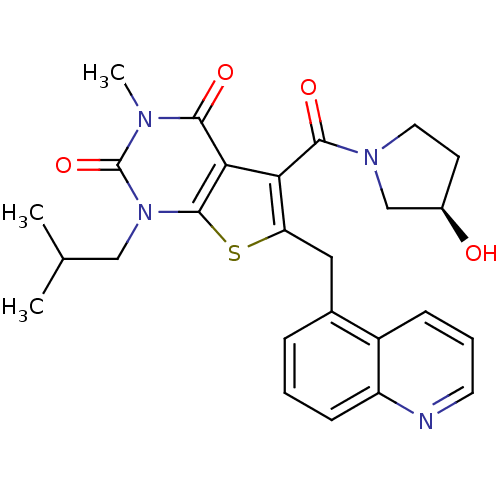
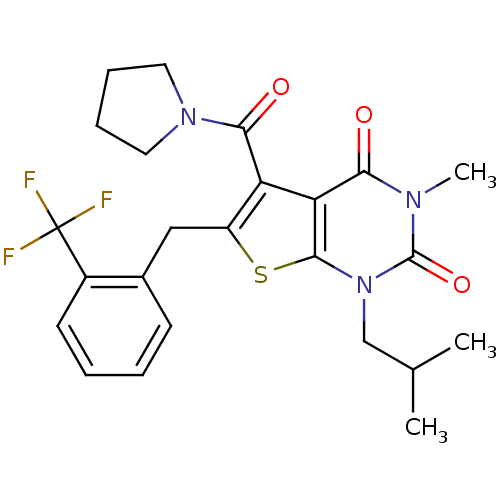
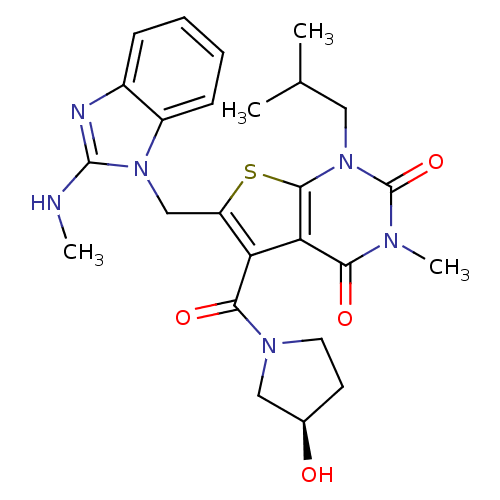
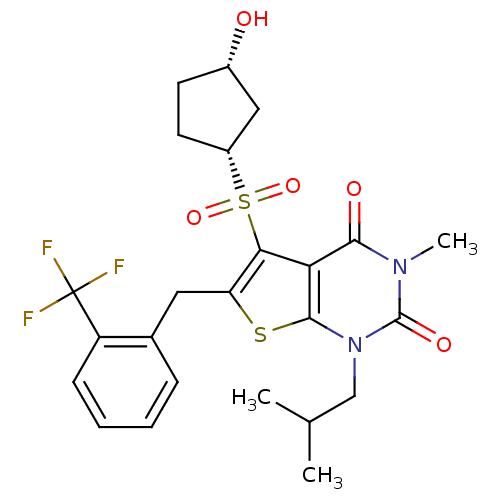
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A(Homo sapiens (Human))
Guilford Pharmaceuticals
Curated by ChEMBL
Guilford Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 0.0100nMAssay Description:Ability to inhibit peptidyl-prolyl isomerase (PPIase, or rotamase) activity of FK506 binding protein 12More data for this Ligand-Target Pair
Affinity DataKi: 0.0955nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 0.100nM ΔG°: -56.5kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 0.275nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 0.280nM ΔG°: -54.0kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 0.290nM ΔG°: -53.9kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 0.302nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 0.330nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 0.330nM ΔG°: -53.6kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 0.331nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 0.350nM ΔG°: -53.4kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 0.355nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 0.355nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 0.420nM ΔG°: -53.0kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 0.680nM ΔG°: -51.8kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 0.708nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 0.790nM ΔG°: -51.4kJ/molepH: 7.4 T: 2°CAssay Description:A radioligand-binding assay was developed using scintillation proximity assay (SPA) technology. The wheat germ agglutinin SPA beads (Amersham) (0.2 m...More data for this Ligand-Target Pair
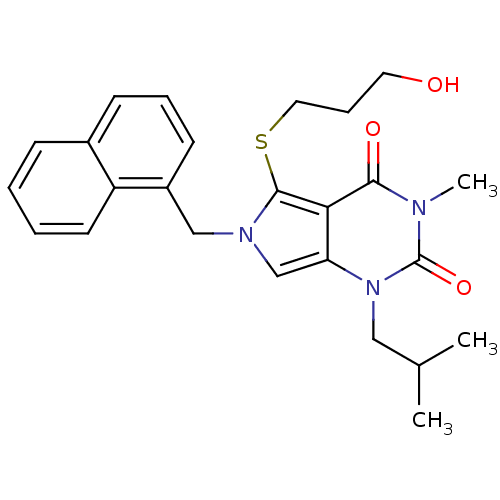
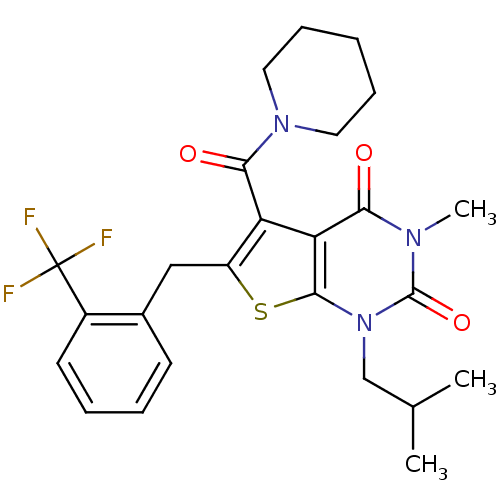
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A(Homo sapiens (Human))
Guilford Pharmaceuticals
Curated by ChEMBL
Guilford Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 1nMAssay Description:Ability to inhibit peptidyl-prolyl isomerase (PPIase, or rotamase) activity of FK506 binding protein 12More data for this Ligand-Target Pair
Affinity DataKi: 1.10nM ΔG°: -50.6kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 1.17nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 2.20nM ΔG°: -48.9kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 2.70nM ΔG°: -48.4kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 3.20nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 3.20nM ΔG°: -48.0kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 3.39nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 3.40nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 3.5nM ΔG°: -47.8kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 3.90nM ΔG°: -47.5kJ/molepH: 7.4 T: 2°CAssay Description:A radioligand-binding assay was developed using scintillation proximity assay (SPA) technology. The wheat germ agglutinin SPA beads (Amersham) (0.2 m...More data for this Ligand-Target Pair
Affinity DataKi: 4.70nM ΔG°: -47.1kJ/molepH: 7.4 T: 2°CAssay Description:A radioligand-binding assay was developed using scintillation proximity assay (SPA) technology. The wheat germ agglutinin SPA beads (Amersham) (0.2 m...More data for this Ligand-Target Pair
Affinity DataKi: 4.80nM ΔG°: -47.0kJ/molepH: 7.4 T: 2°CAssay Description:A radioligand-binding assay was developed using scintillation proximity assay (SPA) technology. The wheat germ agglutinin SPA beads (Amersham) (0.2 m...More data for this Ligand-Target Pair
Affinity DataKi: 4.90nM ΔG°: -47.0kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 5.30nM ΔG°: -46.8kJ/molepH: 7.4 T: 2°CAssay Description:A radioligand-binding assay was developed using scintillation proximity assay (SPA) technology. The wheat germ agglutinin SPA beads (Amersham) (0.2 m...More data for this Ligand-Target Pair
Affinity DataKi: 5.5nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 5.5nM ΔG°: -46.7kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 5.60nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 6nM ΔG°: -46.5kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 6nM ΔG°: -46.5kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
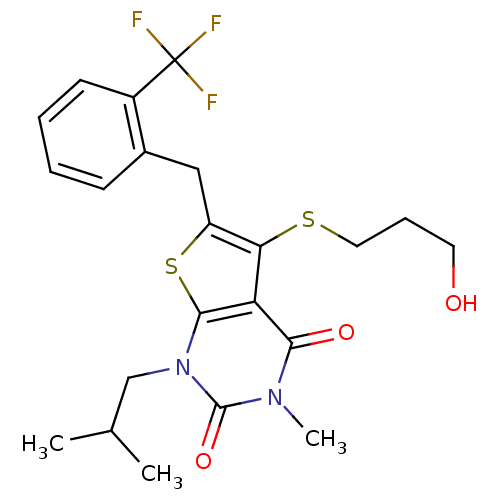
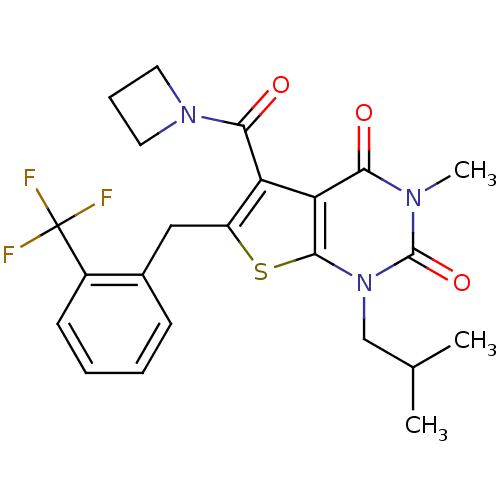
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A(Homo sapiens (Human))
Guilford Pharmaceuticals
Curated by ChEMBL
Guilford Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 6nMAssay Description:Inhibitory activity against FK506 binding protein 12More data for this Ligand-Target Pair
Affinity DataKi: 6.30nMAssay Description:Displacement of [3H]5-[(3-hydroxypropyl)thio]-3-methyl-1-[2-(methyl-t)propyl-2,3-t2]-6-(1-naphthalenylmethyl)-1H-pyrrolo[3,4-d]pyrimidine-2,4(3H,6H)-...More data for this Ligand-Target Pair
Affinity DataKi: 6.5nM ΔG°: -46.3kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
Affinity DataKi: 6.60nM ΔG°: -46.2kJ/molepH: 7.4 T: 2°CAssay Description:A radioligand-binding assay was developed using scintillation proximity assay (SPA) technology. The wheat germ agglutinin SPA beads (Amersham) (0.2 m...More data for this Ligand-Target Pair
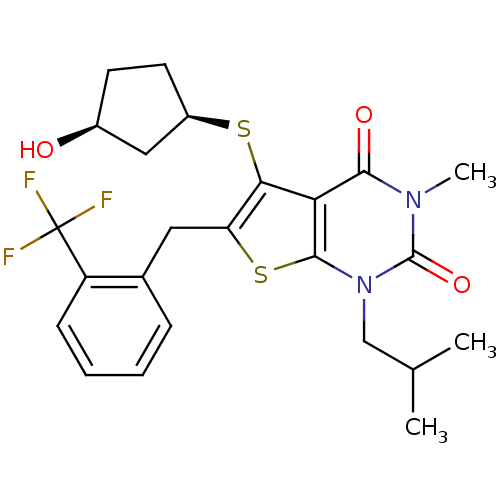
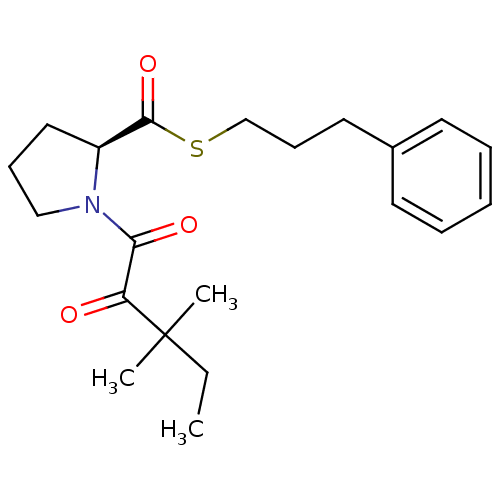
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A(Homo sapiens (Human))
Guilford Pharmaceuticals
Curated by ChEMBL
Guilford Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 7nMAssay Description:Inhibitory activity against FK506 binding protein 12More data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A(Homo sapiens (Human))
Guilford Pharmaceuticals
Curated by ChEMBL
Guilford Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 7.5nMAssay Description:Inhibitory activity against FK506 binding protein 12More data for this Ligand-Target Pair
Affinity DataKi: 7.5nM ΔG°: -42.5kJ/molepH: 8.0 T: 2°CAssay Description:PPIase(Rotamase) activity of FKBP12 was assayed using the peptide N-succinyl Ala-Leu-Pro-Phe p-nitroanilide as substrate. It is based on the observat...More data for this Ligand-Target Pair
Affinity DataKi: 8.70nM ΔG°: -45.5kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A(Homo sapiens (Human))
Guilford Pharmaceuticals
Curated by ChEMBL
Guilford Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 8.70nMAssay Description:Inhibitory activity against FK506 binding protein 12More data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A(Homo sapiens (Human))
Guilford Pharmaceuticals
Curated by ChEMBL
Guilford Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 8.90nMAssay Description:Inhibitory activity against FK506 binding protein 12More data for this Ligand-Target Pair
Affinity DataKi: 9.20nM ΔG°: -45.4kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A(Homo sapiens (Human))
Guilford Pharmaceuticals
Curated by ChEMBL
Guilford Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 11nMAssay Description:Inhibitory activity against FK506 binding protein 12More data for this Ligand-Target Pair
Affinity DataKi: 12nM ΔG°: -44.8kJ/molepH: 7.8 T: 2°CAssay Description:Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ...More data for this Ligand-Target Pair


 3D Structure (crystal)
3D Structure (crystal)