Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB (change energy unit to kcal/mol)
 Found 84 hits in this display
Found 84 hits in this display
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
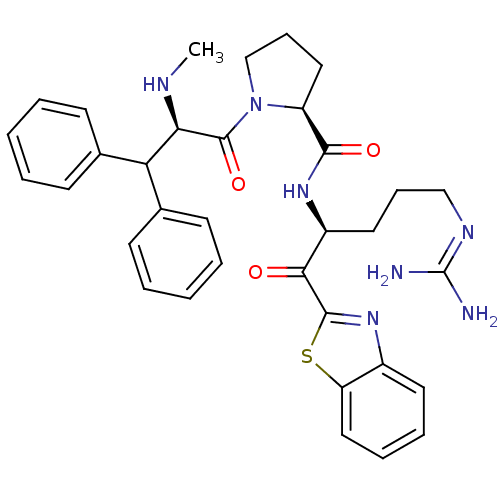 BDBM14073((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
BDBM14073((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14065((2S)-1-[(2R)-2-amino-3-phenylpropanoyl]-N-[1-(1,3-...)
BDBM14065((2S)-1-[(2R)-2-amino-3-phenylpropanoyl]-N-[1-(1,3-...)Ki: 0.00550nM ΔG°: -66.9kJ/mole IC50: 21nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
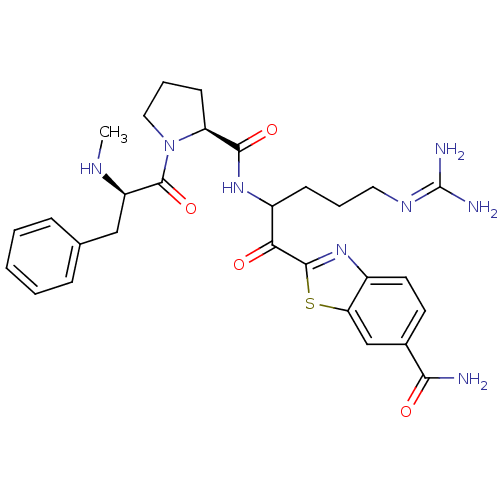 BDBM14127(2-(5-carbamimidamido-2-{[(2S)-1-[(2R)-2-(methylami...)
BDBM14127(2-(5-carbamimidamido-2-{[(2S)-1-[(2R)-2-(methylami...)Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
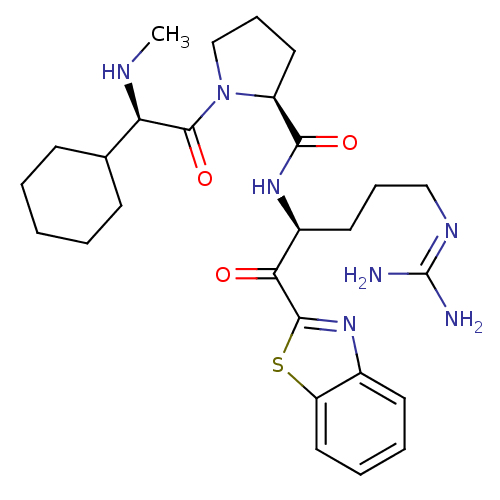 BDBM14076((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
BDBM14076((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)Ki: 0.0180nM ΔG°: -63.8kJ/mole IC50: 5.30nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
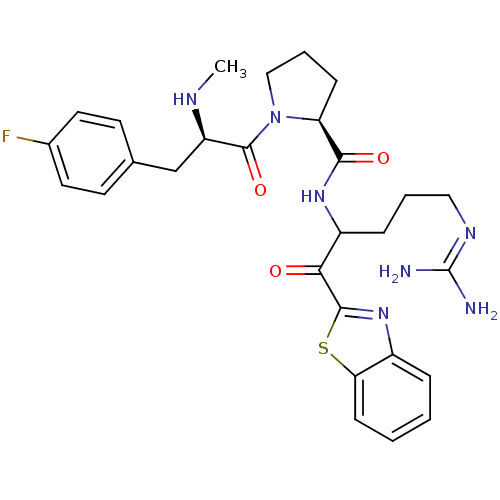 BDBM14066((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14066((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 0.120nM ΔG°: -58.9kJ/mole IC50: 15nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
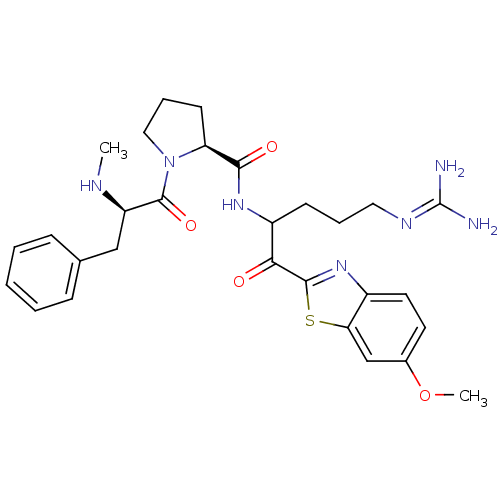 BDBM14123((2S)-N-[5-carbamimidamido-1-(6-methoxy-1,3-benzoth...)
BDBM14123((2S)-N-[5-carbamimidamido-1-(6-methoxy-1,3-benzoth...)Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14125((2S)-N-[5-carbamimidamido-1-(6-fluoro-1,3-benzothi...)
BDBM14125((2S)-N-[5-carbamimidamido-1-(6-fluoro-1,3-benzothi...)Ki: 0.150nM IC50: 7.5nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
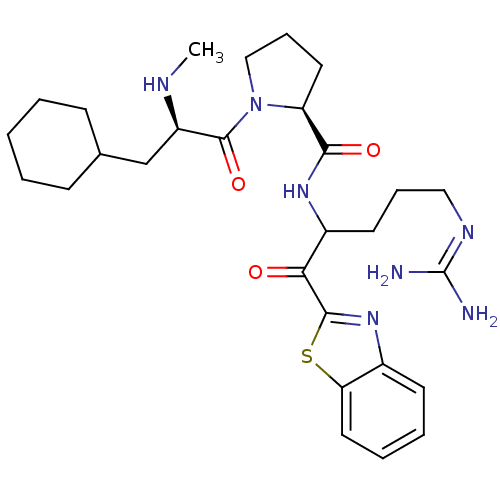 BDBM14068((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14068((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 0.180nM ΔG°: -57.9kJ/mole IC50: 48nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
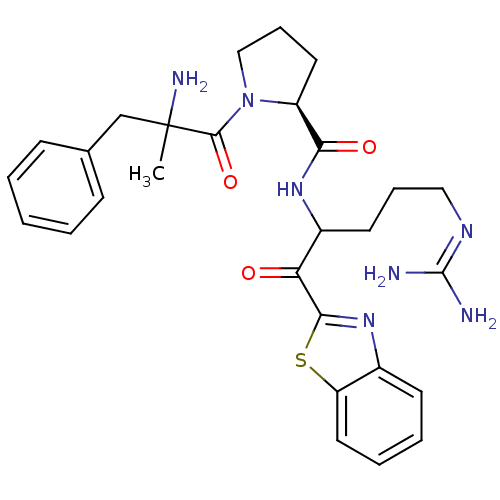 BDBM14071((2S)-1-(2-amino-2-benzylpropanoyl)-N-[1-(1,3-benzo...)
BDBM14071((2S)-1-(2-amino-2-benzylpropanoyl)-N-[1-(1,3-benzo...)Ki: 0.200nM ΔG°: -57.6kJ/mole IC50: 3.5nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14063((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14063((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 0.200nM ΔG°: -57.6kJ/mole IC50: 29nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
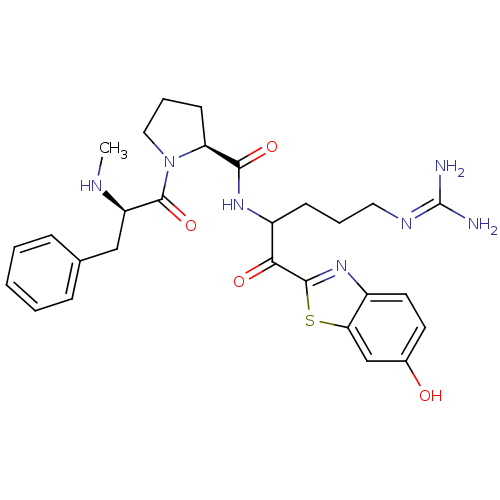 BDBM14124((2S)-N-[5-carbamimidamido-1-(6-hydroxy-1,3-benzoth...)
BDBM14124((2S)-N-[5-carbamimidamido-1-(6-hydroxy-1,3-benzoth...)Ki: 0.230nM IC50: 3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14086((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14086((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
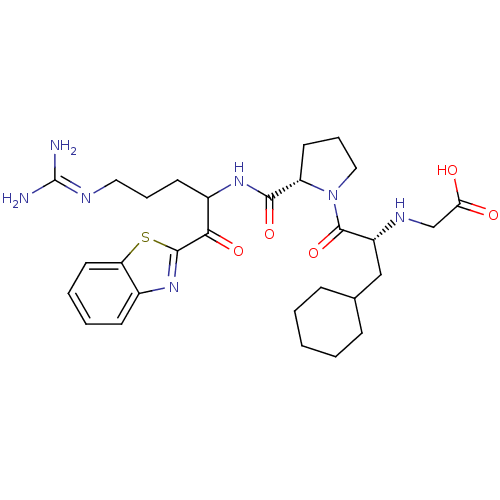 BDBM14072(2-ketobenzothiazole 12 | 2-{[(2R)-1-[(2S)-2-{[1-(1...)
BDBM14072(2-ketobenzothiazole 12 | 2-{[(2R)-1-[(2S)-2-{[1-(1...)Ki: 0.360nM ΔG°: -56.1kJ/mole IC50: 29nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
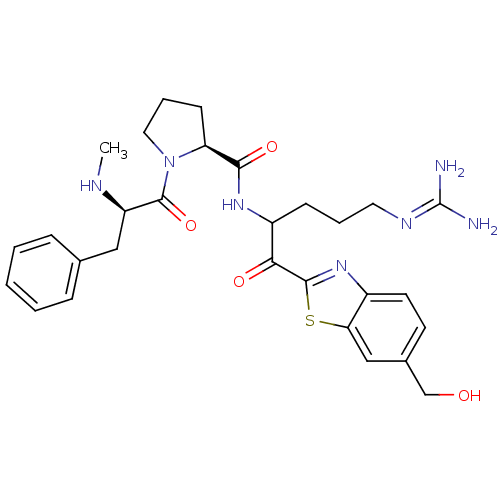 BDBM14126((2S)-N-{5-carbamimidamido-1-[6-(hydroxymethyl)-1,3...)
BDBM14126((2S)-N-{5-carbamimidamido-1-[6-(hydroxymethyl)-1,3...)Ki: 0.370nM IC50: 7nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
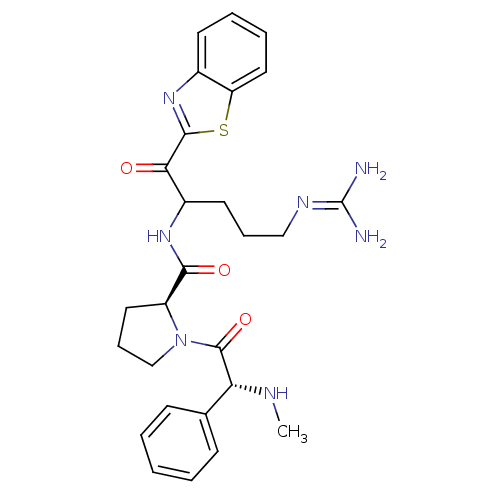 BDBM14075((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14075((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 0.460nM ΔG°: -55.4kJ/mole IC50: 34nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14122((2S)-N-(5-carbamimidamido-1-oxo-1-{3-thia-5-azatri...)
BDBM14122((2S)-N-(5-carbamimidamido-1-oxo-1-{3-thia-5-azatri...)Ki: 0.580nM IC50: 60nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14139((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
BDBM14139((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)Ki: 0.780nM IC50: 110nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
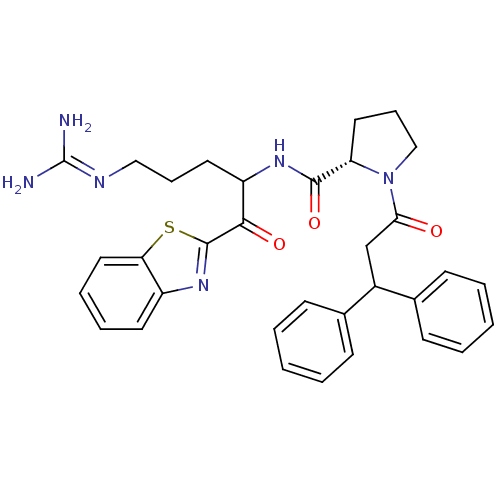 BDBM14074((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14074((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 1.10nM ΔG°: -53.2kJ/mole IC50: 11nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
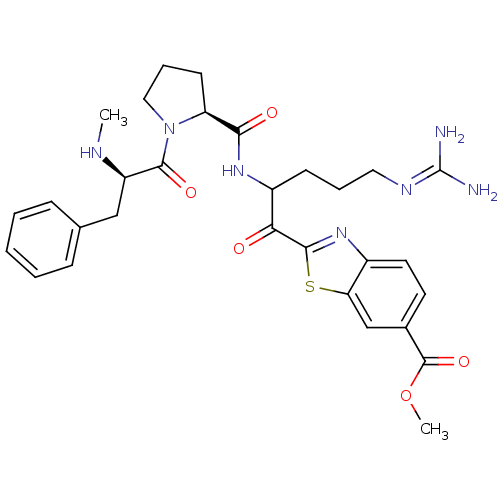 BDBM14128(2-ketobenzothiazole 68 | methyl 2-(5-carbamimidami...)
BDBM14128(2-ketobenzothiazole 68 | methyl 2-(5-carbamimidami...)Ki: 1.30nM IC50: 38nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
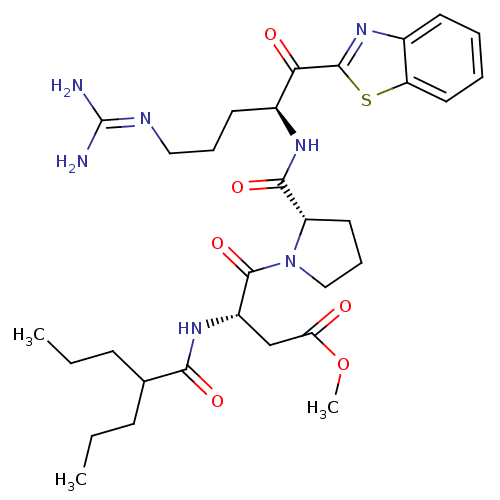 BDBM14137(2-ketobenzothiazole 75 | methyl (3S)-4-[(2S)-2-{[(...)
BDBM14137(2-ketobenzothiazole 75 | methyl (3S)-4-[(2S)-2-{[(...)Ki: 1.40nM IC50: 56nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14138((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
BDBM14138((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)Ki: 1.5nM IC50: 95nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
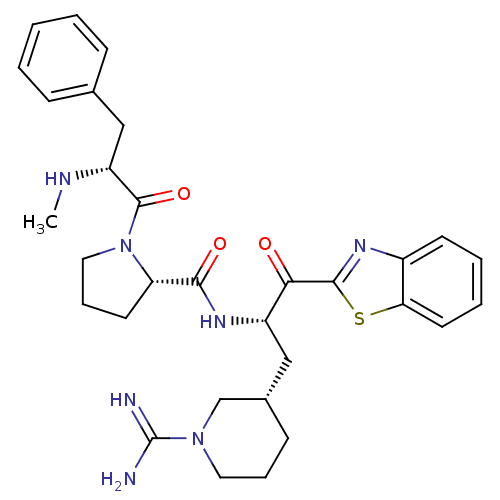 BDBM14094((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-3-[(3S)-1-c...)
BDBM14094((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-3-[(3S)-1-c...)Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
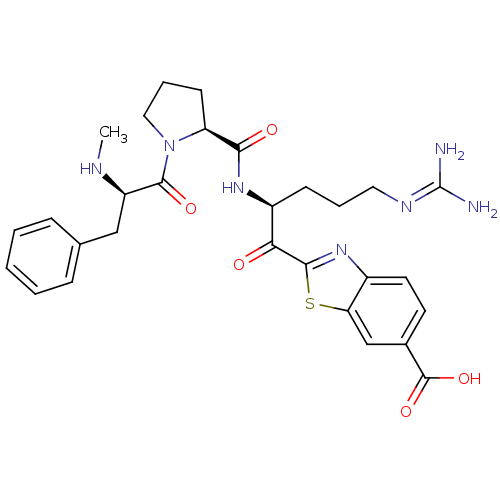 BDBM14129(2-[(2S)-5-carbamimidamido-2-{[(2S)-1-[(2R)-2-(meth...)
BDBM14129(2-[(2S)-5-carbamimidamido-2-{[(2S)-1-[(2R)-2-(meth...)Ki: 2nM IC50: 45nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
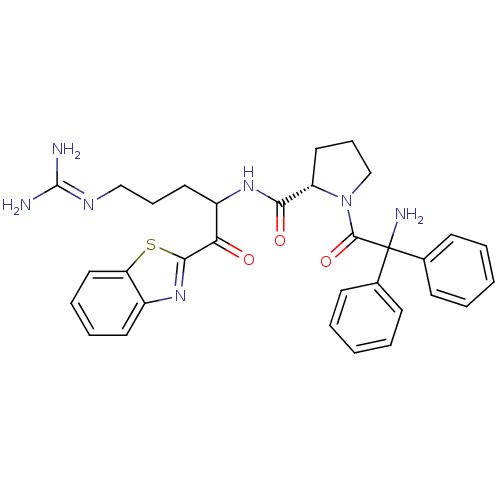 BDBM14078((2S)-1-(2-amino-2,2-diphenylacetyl)-N-[1-(1,3-benz...)
BDBM14078((2S)-1-(2-amino-2,2-diphenylacetyl)-N-[1-(1,3-benz...)Ki: 2.10nM ΔG°: -51.5kJ/mole IC50: 15nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
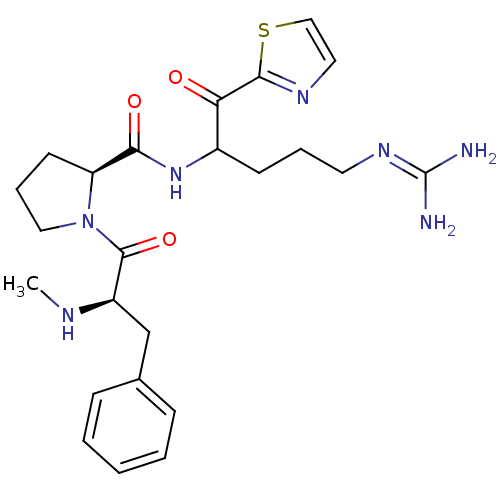 BDBM14117((2S)-N-[5-carbamimidamido-1-oxo-1-(1,3-thiazol-2-y...)
BDBM14117((2S)-N-[5-carbamimidamido-1-oxo-1-(1,3-thiazol-2-y...)Ki: 2.10nM IC50: 19nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
 BDBM14063((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14063((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
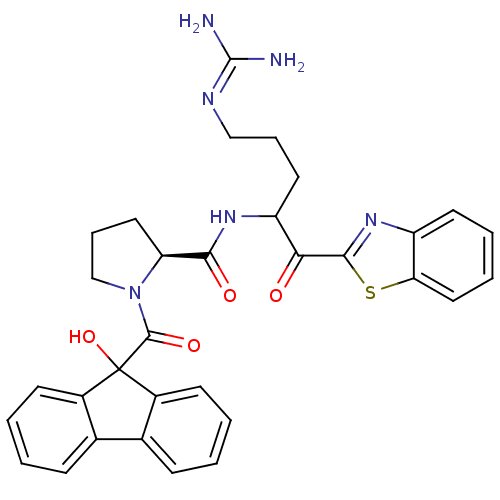 BDBM14079((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14079((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 3.10nM IC50: 95nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14121((2S)-N-[5-carbamimidamido-1-oxo-1-(4,5,6,7-tetrahy...)
BDBM14121((2S)-N-[5-carbamimidamido-1-oxo-1-(4,5,6,7-tetrahy...)Ki: 3.40nM IC50: 80nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14067((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14067((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 3.80nM ΔG°: -50.0kJ/mole IC50: 150nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14119(2-ketobenzothiazole 59 | ethyl 2-(5-carbamimidamid...)
BDBM14119(2-ketobenzothiazole 59 | ethyl 2-(5-carbamimidamid...)Ki: 4.5nM IC50: 120nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
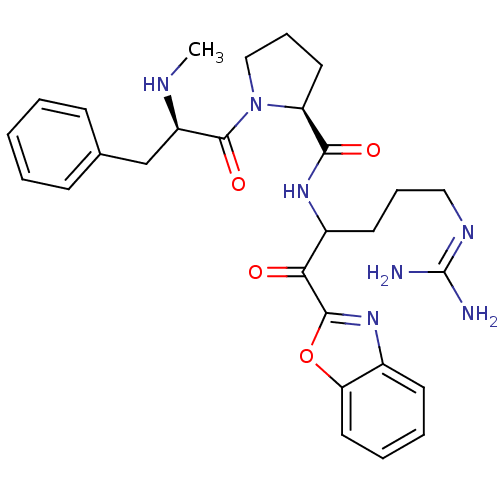 BDBM14115((2S)-N-[1-(1,3-benzoxazol-2-yl)-5-carbamimidamido-...)
BDBM14115((2S)-N-[1-(1,3-benzoxazol-2-yl)-5-carbamimidamido-...)Ki: 6.20nM IC50: 110nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
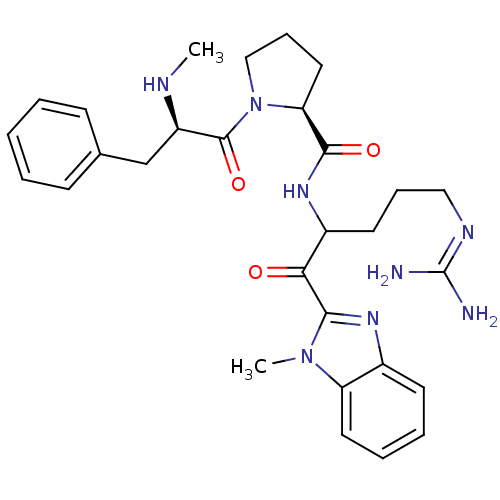 BDBM14114((2S)-N-[5-carbamimidamido-1-(1-methyl-1H-1,3-benzo...)
BDBM14114((2S)-N-[5-carbamimidamido-1-(1-methyl-1H-1,3-benzo...)Ki: 8.10nM IC50: 110nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
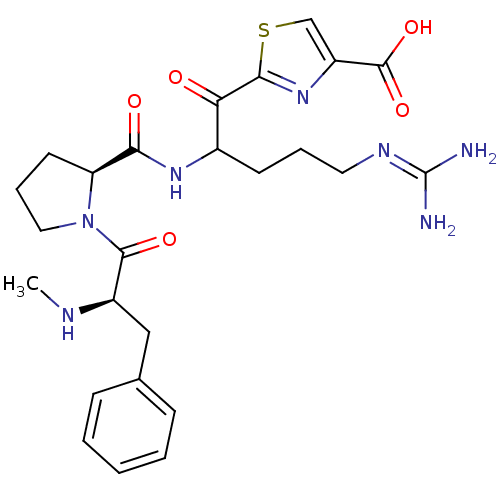 BDBM14118(2-(5-carbamimidamido-2-{[(2S)-1-[(2R)-2-(methylami...)
BDBM14118(2-(5-carbamimidamido-2-{[(2S)-1-[(2R)-2-(methylami...)Ki: 9.10nM IC50: 112nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
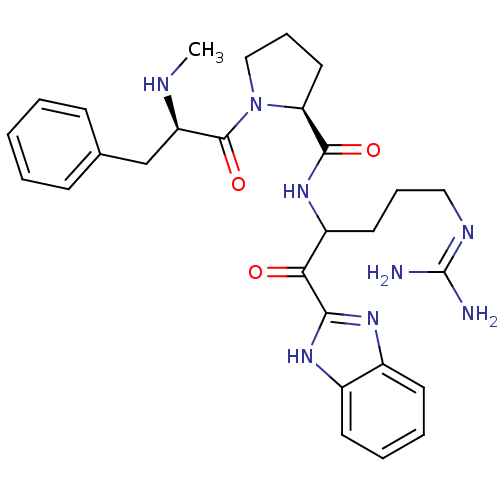 BDBM14113((2S)-N-[1-(1H-1,3-benzodiazol-2-yl)-5-carbamimidam...)
BDBM14113((2S)-N-[1-(1H-1,3-benzodiazol-2-yl)-5-carbamimidam...)Ki: 9.60nM IC50: 360nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14069((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14069((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 13nM ΔG°: -46.8kJ/mole IC50: 430nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
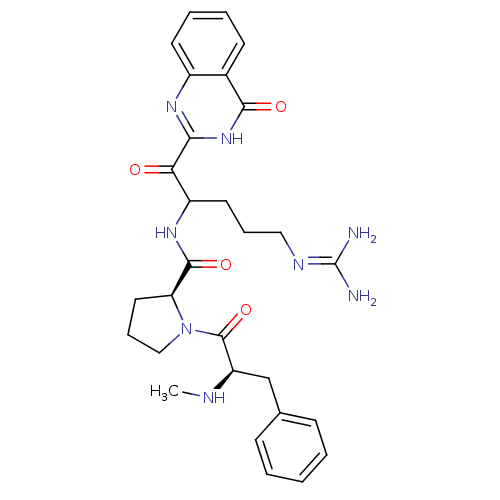 BDBM14116((2S)-N-[5-carbamimidamido-1-oxo-1-(4-oxo-3,4-dihyd...)
BDBM14116((2S)-N-[5-carbamimidamido-1-oxo-1-(4-oxo-3,4-dihyd...)Ki: 15nM IC50: 106nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14093((2S)-N-[(2R)-1-(1,3-benzothiazol-2-yl)-3-(3-carbam...)
BDBM14093((2S)-N-[(2R)-1-(1,3-benzothiazol-2-yl)-3-(3-carbam...)Ki: 15nM IC50: 370nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
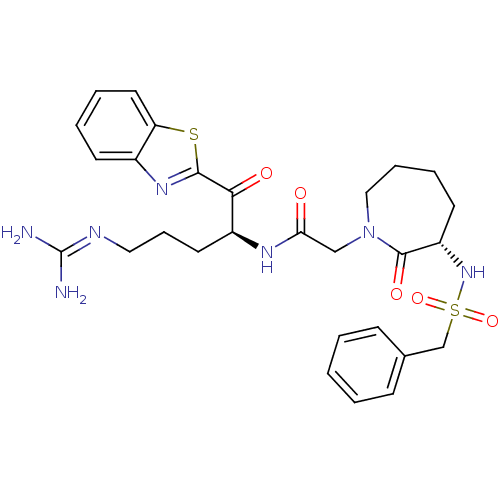 BDBM14136(2-ketobenzothiazole 74 | N-[(2S)-1-(1,3-benzothiaz...)
BDBM14136(2-ketobenzothiazole 74 | N-[(2S)-1-(1,3-benzothiaz...)Ki: 16nM IC50: 270nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
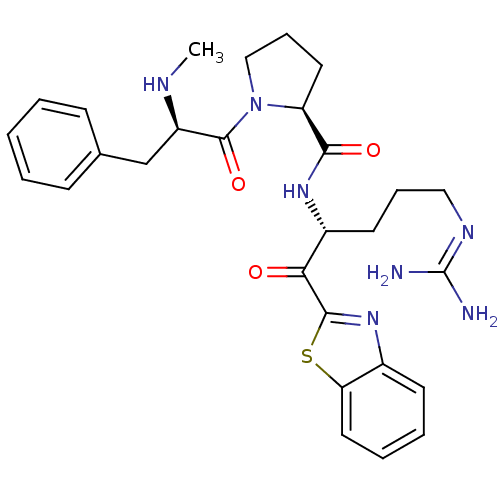 BDBM14087((2S)-N-[(2R)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
BDBM14087((2S)-N-[(2R)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)Ki: 17nM IC50: 80nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
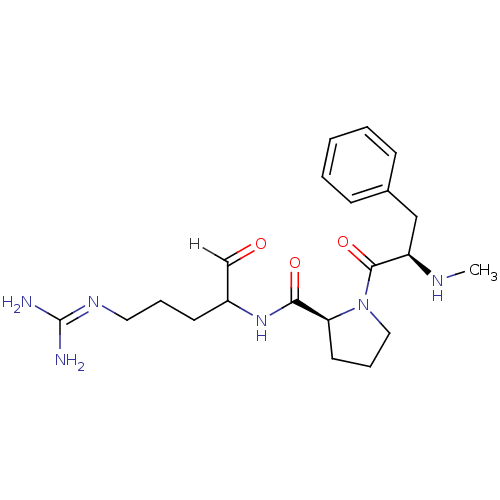 BDBM14130((2S)-N-(5-carbamimidamido-1-oxopentan-2-yl)-1-[(2R...)
BDBM14130((2S)-N-(5-carbamimidamido-1-oxopentan-2-yl)-1-[(2R...)Ki: 18nM IC50: 85nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
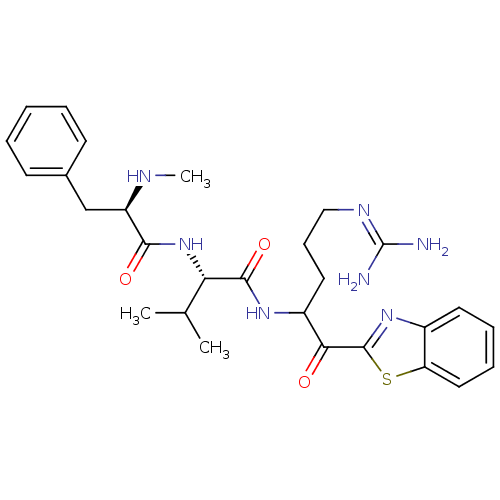 BDBM14085((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14085((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 21nM IC50: 440nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
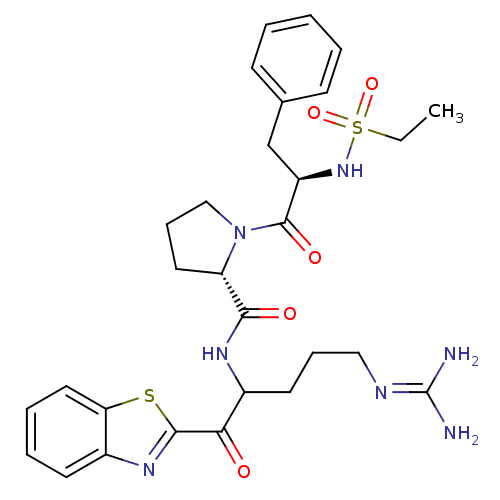 BDBM14070((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14070((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 24nM ΔG°: -45.2kJ/mole IC50: 500nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
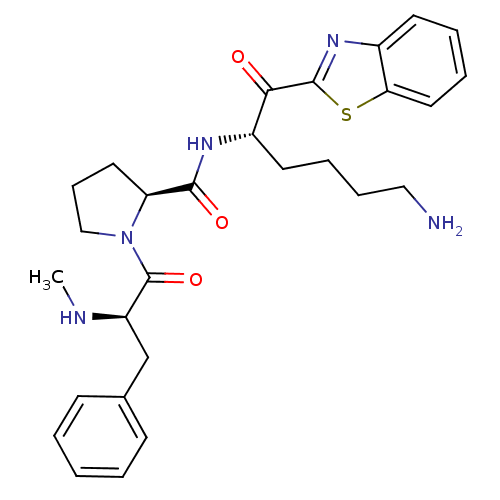 BDBM14097((2S)-N-[(2S)-6-amino-1-(1,3-benzothiazol-2-yl)-1-o...)
BDBM14097((2S)-N-[(2S)-6-amino-1-(1,3-benzothiazol-2-yl)-1-o...)Ki: 38nM IC50: 650nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
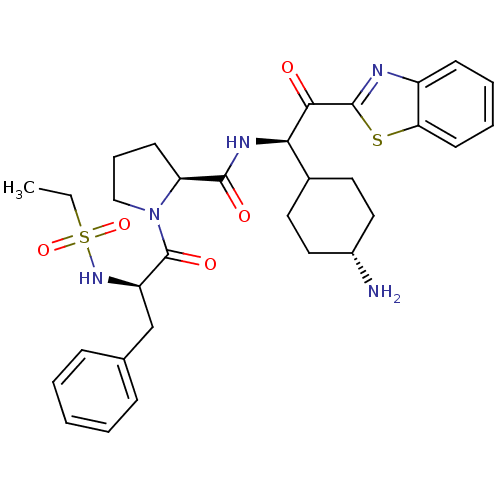 BDBM14132((2S)-N-[(1R)-1-(4-aminocyclohexyl)-2-(1,3-benzothi...)
BDBM14132((2S)-N-[(1R)-1-(4-aminocyclohexyl)-2-(1,3-benzothi...)Ki: 53nM IC50: 460nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14077((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14077((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 73nM ΔG°: -42.4kJ/mole IC50: 490nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
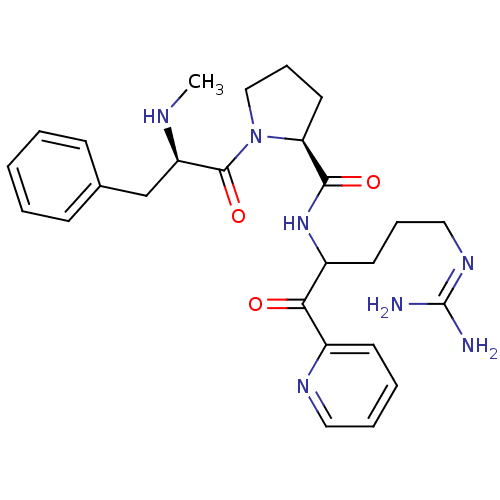 BDBM14110((2S)-N-[5-carbamimidamido-1-oxo-1-(pyridin-2-yl)pe...)
BDBM14110((2S)-N-[5-carbamimidamido-1-oxo-1-(pyridin-2-yl)pe...)Ki: 85nM IC50: 930nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
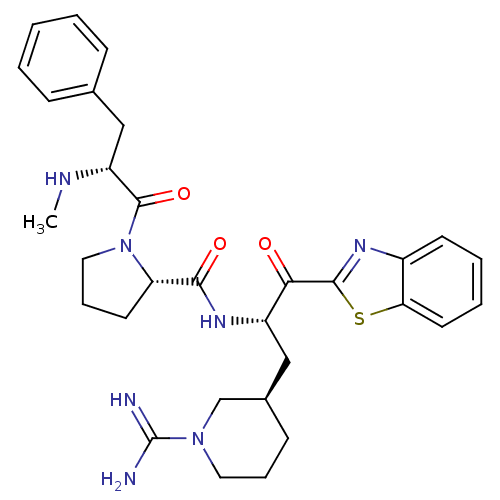 BDBM14095((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-3-[(3R)-1-c...)
BDBM14095((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-3-[(3R)-1-c...)Ki: 99nM IC50: 1.00E+3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
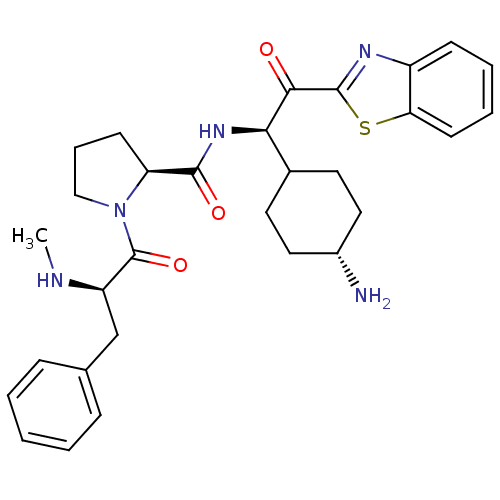 BDBM14131((2S)-N-[(1R)-1-(4-aminocyclohexyl)-2-(1,3-benzothi...)
BDBM14131((2S)-N-[(1R)-1-(4-aminocyclohexyl)-2-(1,3-benzothi...)Ki: 110nM IC50: 1.90E+3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
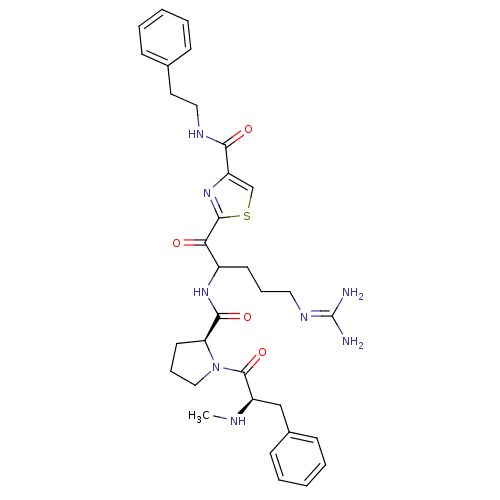 BDBM14120(2-(5-carbamimidamido-2-{[(2S)-1-[(2R)-2-(methylami...)
BDBM14120(2-(5-carbamimidamido-2-{[(2S)-1-[(2R)-2-(methylami...)Ki: 120nM IC50: 830nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
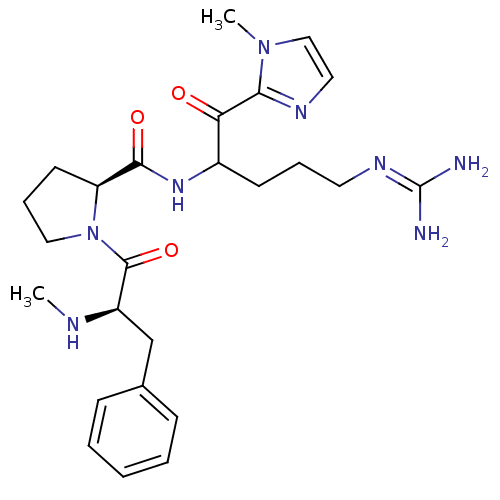 BDBM14112((2S)-N-[5-carbamimidamido-1-(1-methyl-1H-imidazol-...)
BDBM14112((2S)-N-[5-carbamimidamido-1-(1-methyl-1H-imidazol-...)Ki: 200nM IC50: 1.50E+3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
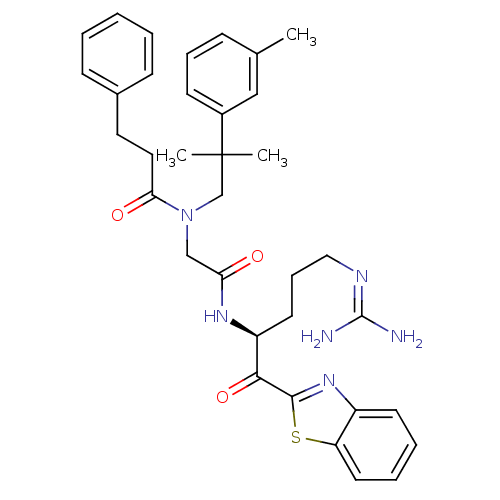 BDBM14135(2-ketobenzothiazole 73 | N-({[(2S)-1-(1,3-benzothi...)
BDBM14135(2-ketobenzothiazole 73 | N-({[(2S)-1-(1,3-benzothi...)Ki: 400nM IC50: 4.00E+3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
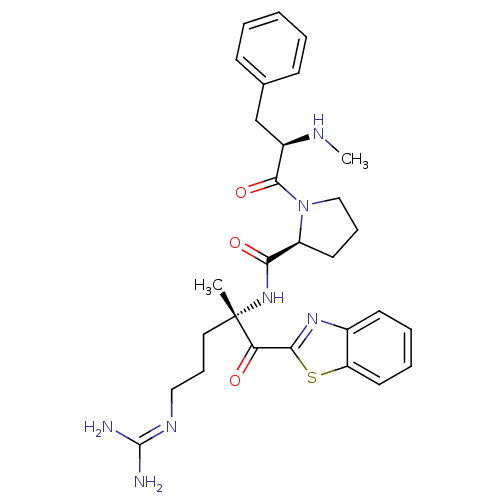 BDBM14088((2S)-N-[(2S)-2-(1,3-benzothiazol-2-ylcarbonyl)-5-c...)
BDBM14088((2S)-N-[(2S)-2-(1,3-benzothiazol-2-ylcarbonyl)-5-c...)Ki: 430nM IC50: 3.70E+3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
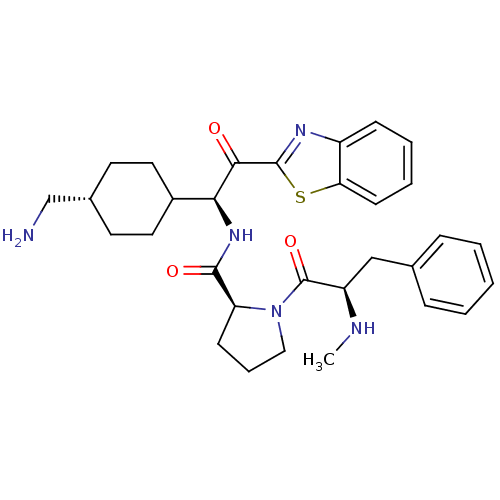 BDBM14101((2S)-N-[(1S)-1-[4-(aminomethyl)cyclohexyl]-2-(1,3-...)
BDBM14101((2S)-N-[(1S)-1-[4-(aminomethyl)cyclohexyl]-2-(1,3-...)Ki: 920nM IC50: 1.70E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
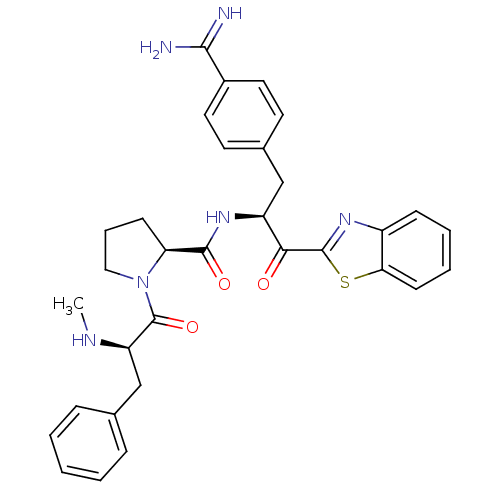 BDBM14091((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-3-(4-carbam...)
BDBM14091((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-3-(4-carbam...)Ki: 1.40E+3nM IC50: >1.00E+3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
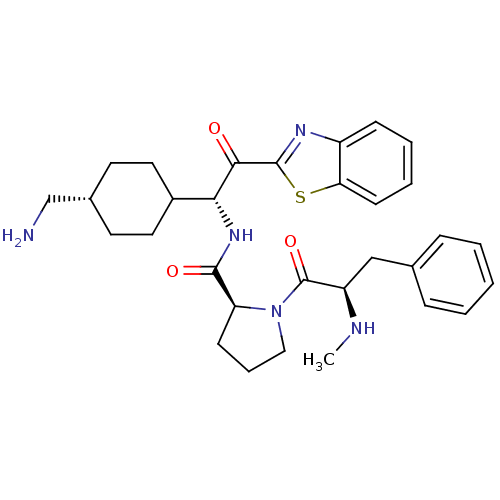 BDBM14102((2S)-N-[(1R)-1-[4-(aminomethyl)cyclohexyl]-2-(1,3-...)
BDBM14102((2S)-N-[(1R)-1-[4-(aminomethyl)cyclohexyl]-2-(1,3-...)Ki: 1.45E+3nM IC50: >1.00E+3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
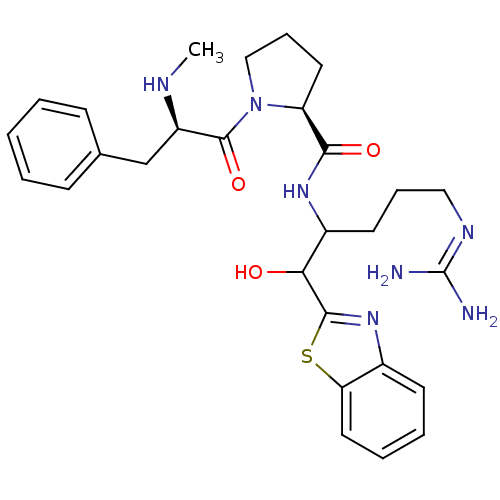 BDBM14108((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14108((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 1.60E+3nM IC50: 1.70E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
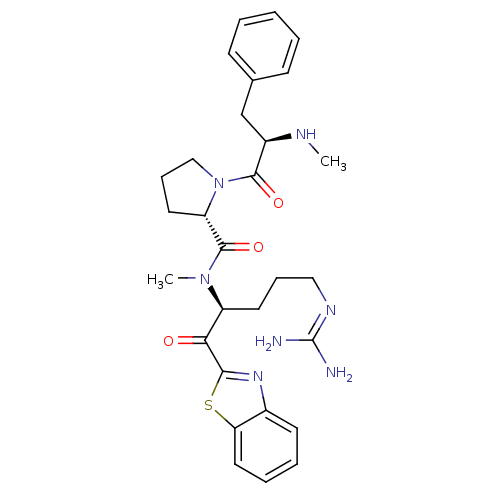 BDBM14089((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
BDBM14089((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)Ki: 2.10E+3nM IC50: 2.10E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
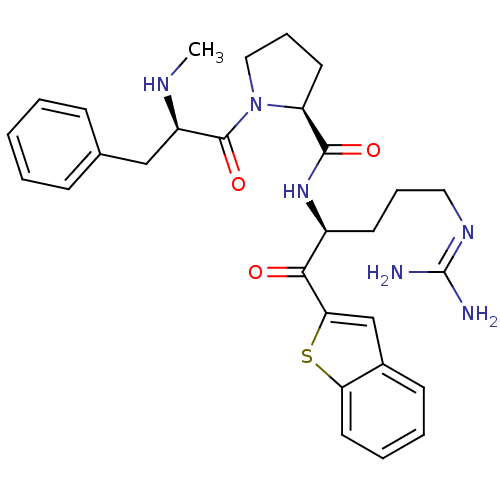 BDBM14111((2S)-N-[(2S)-1-(1-benzothiophen-2-yl)-5-carbamimid...)
BDBM14111((2S)-N-[(2S)-1-(1-benzothiophen-2-yl)-5-carbamimid...)Ki: 2.40E+3nM IC50: 1.30E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
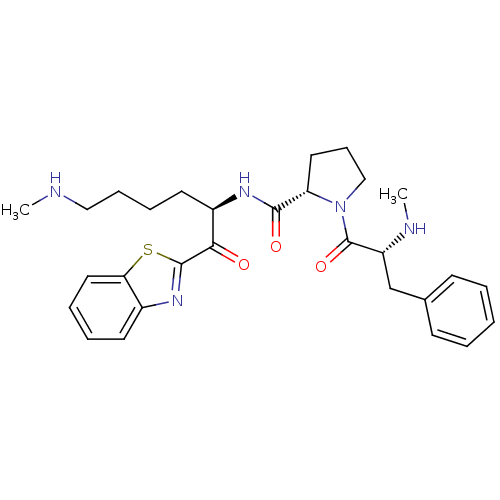 BDBM14100((2S)-N-[(2R)-1-(1,3-benzothiazol-2-yl)-6-(methylam...)
BDBM14100((2S)-N-[(2R)-1-(1,3-benzothiazol-2-yl)-6-(methylam...)Ki: 3.50E+3nM IC50: >1.00E+3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
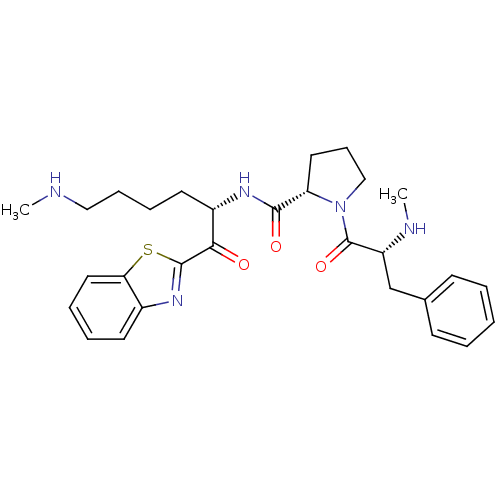 BDBM14099((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-6-(methylam...)
BDBM14099((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-6-(methylam...)Ki: 3.70E+3nM IC50: >1.00E+3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14103((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-methoxy-1...)
BDBM14103((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-methoxy-1...)Ki: 3.90E+3nM IC50: 4.60E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14108((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14108((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Ki: 5.30E+3nM IC50: 4.80E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
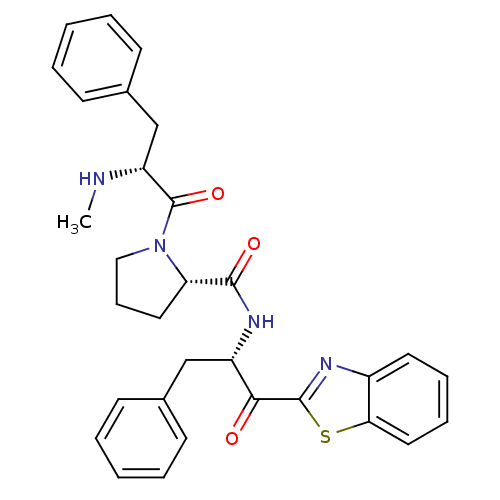 BDBM14105((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-1-oxo-3-phe...)
BDBM14105((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-1-oxo-3-phe...)Ki: 7.10E+3nM IC50: >1.00E+3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14083(2-ketobenzothiazole 23 | N-[1-(1,3-benzothiazol-2-...)
BDBM14083(2-ketobenzothiazole 23 | N-[1-(1,3-benzothiazol-2-...)Ki: 1.46E+4nM IC50: 8.20E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
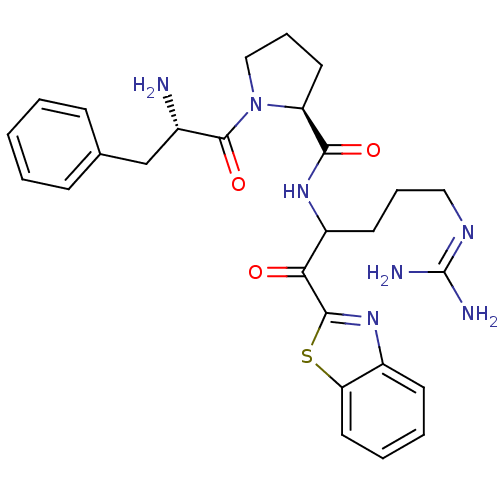 BDBM14140((2S)-1-[(2S)-2-amino-3-phenylpropanoyl]-N-[1-(1,3-...)
BDBM14140((2S)-1-[(2S)-2-amino-3-phenylpropanoyl]-N-[1-(1,3-...)
 BDBM14073((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
BDBM14073((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)IC50: 30nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
 BDBM14075((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14075((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)IC50: 32nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
 BDBM14091((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-3-(4-carbam...)
BDBM14091((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-3-(4-carbam...)
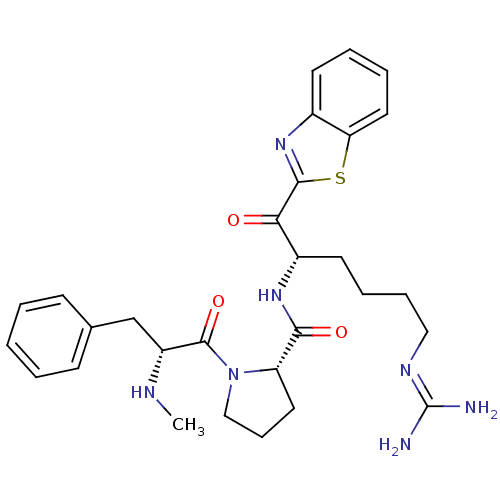 BDBM14090((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-6-carbamimi...)
BDBM14090((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-6-carbamimi...)IC50: 630nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14090((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-6-carbamimi...)
BDBM14090((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-6-carbamimi...)IC50: 8.40E+3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
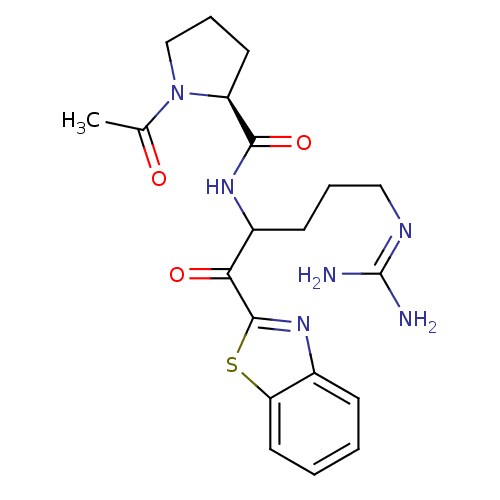 BDBM14080((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14080((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)IC50: 1.40E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14140((2S)-1-[(2S)-2-amino-3-phenylpropanoyl]-N-[1-(1,3-...)
BDBM14140((2S)-1-[(2S)-2-amino-3-phenylpropanoyl]-N-[1-(1,3-...)IC50: 1.70E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
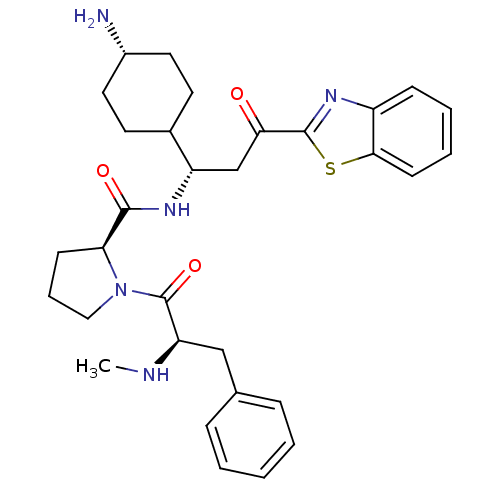 BDBM14134((2S)-N-[(1S)-1-(4-aminocyclohexyl)-3-(1,3-benzothi...)
BDBM14134((2S)-N-[(1S)-1-(4-aminocyclohexyl)-3-(1,3-benzothi...)IC50: 2.20E+4nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
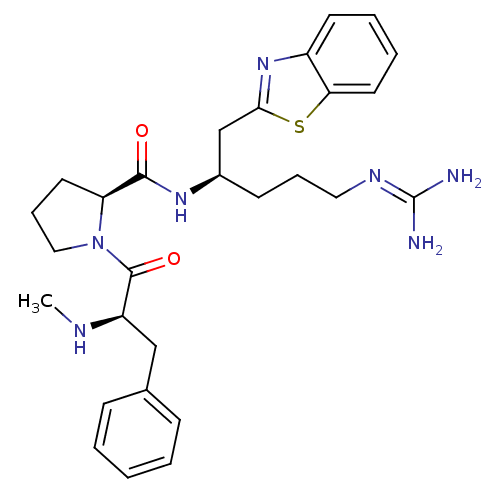 BDBM14107((2S)-N-[(2R)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
BDBM14107((2S)-N-[(2R)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
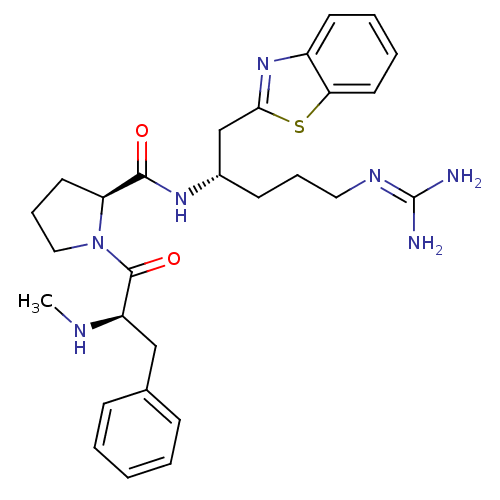 BDBM14106((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
BDBM14106((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)IC50: 3.20E+4nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
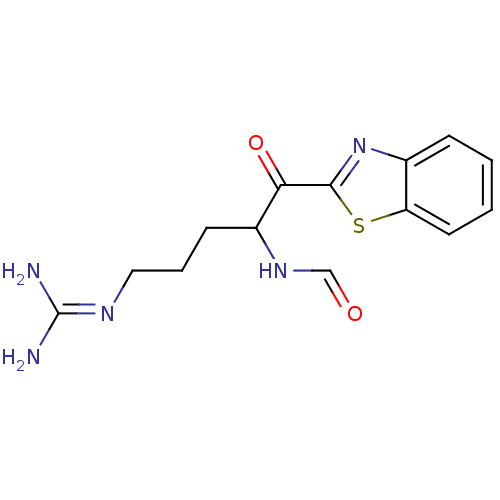 BDBM14082(2-ketobenzothiazole 22 | N-[1-(1,3-benzothiazol-2-...)
BDBM14082(2-ketobenzothiazole 22 | N-[1-(1,3-benzothiazol-2-...)IC50: >5.00E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14106((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
BDBM14106((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)IC50: 5.80E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
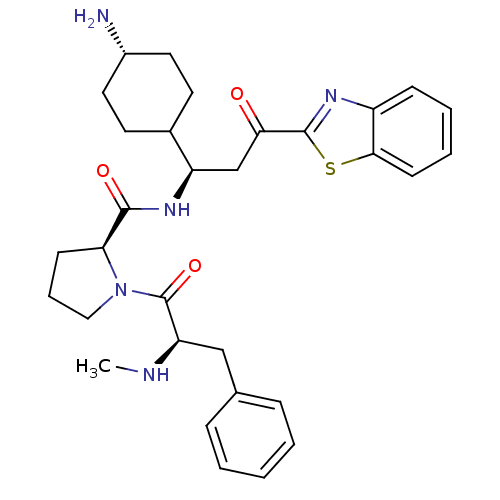 BDBM14133((2S)-N-[(1R)-1-(4-aminocyclohexyl)-3-(1,3-benzothi...)
BDBM14133((2S)-N-[(1R)-1-(4-aminocyclohexyl)-3-(1,3-benzothi...)IC50: 5.80E+4nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14107((2S)-N-[(2R)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)
BDBM14107((2S)-N-[(2R)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)IC50: 6.30E+4nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14133((2S)-N-[(1R)-1-(4-aminocyclohexyl)-3-(1,3-benzothi...)
BDBM14133((2S)-N-[(1R)-1-(4-aminocyclohexyl)-3-(1,3-benzothi...)IC50: >1.00E+5nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
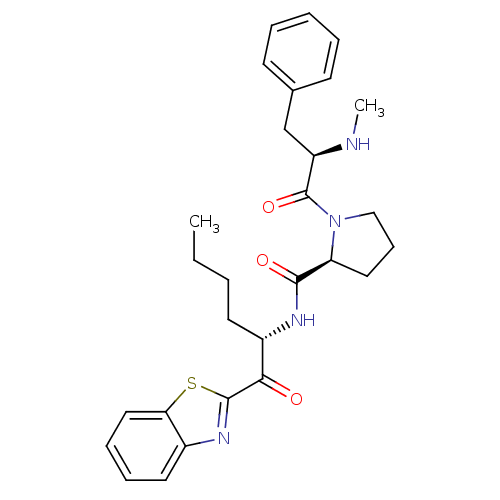 BDBM14104((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-1-oxohexan-...)
BDBM14104((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-1-oxohexan-...)IC50: >1.00E+5nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
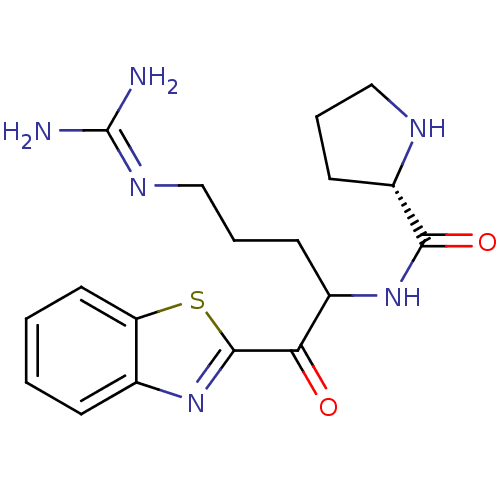 BDBM14084((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)
BDBM14084((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)IC50: >1.00E+5nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
 BDBM14134((2S)-N-[(1S)-1-(4-aminocyclohexyl)-3-(1,3-benzothi...)
BDBM14134((2S)-N-[(1S)-1-(4-aminocyclohexyl)-3-(1,3-benzothi...)IC50: >1.00E+5nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Prothrombin(Homo sapiens (Human))Johnson & Johnson Pharmaceutical
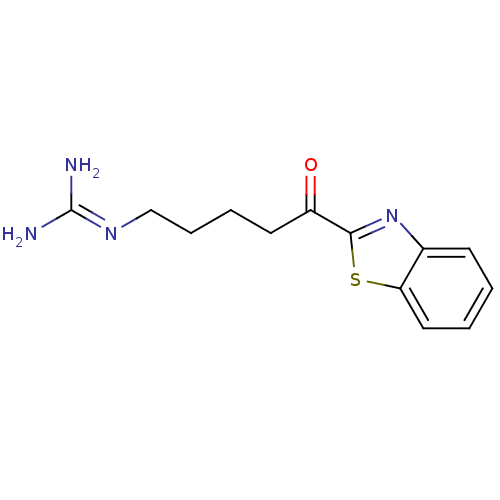 BDBM14081(2-ketobenzothiazole 21 | 3-[5-(1,3-benzothiazol-2-...)
BDBM14081(2-ketobenzothiazole 21 | 3-[5-(1,3-benzothiazol-2-...)IC50: >3.00E+5nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair