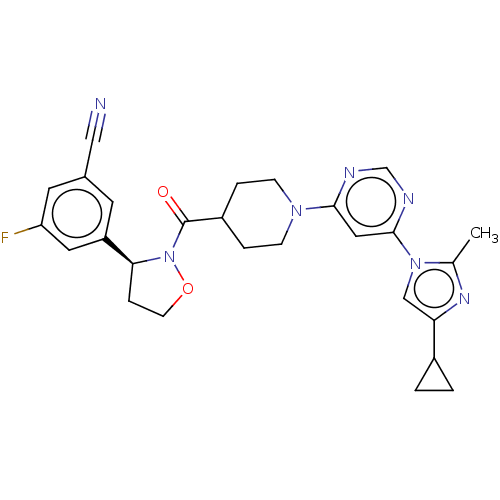
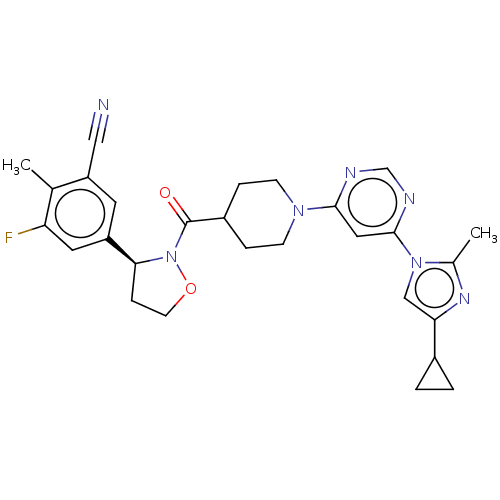
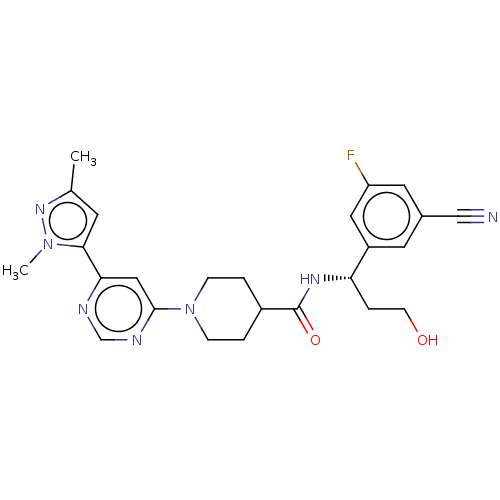
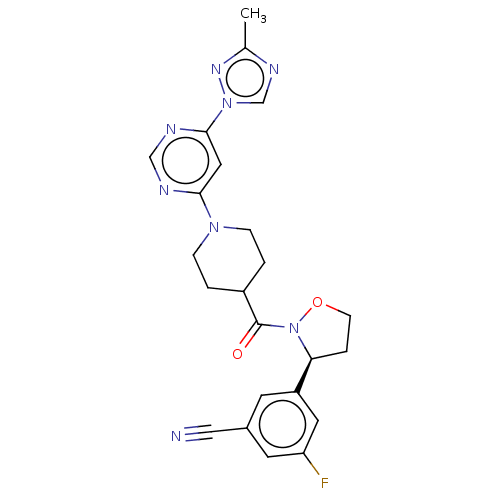
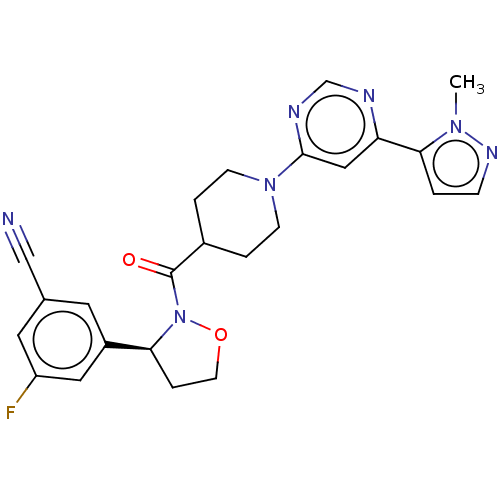
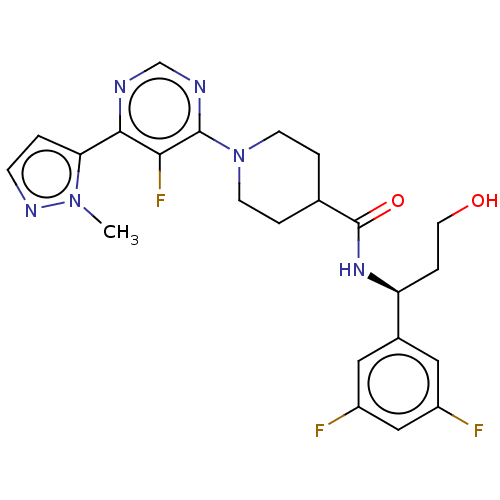
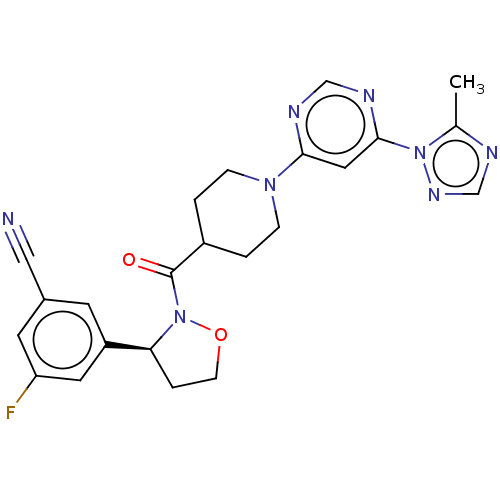
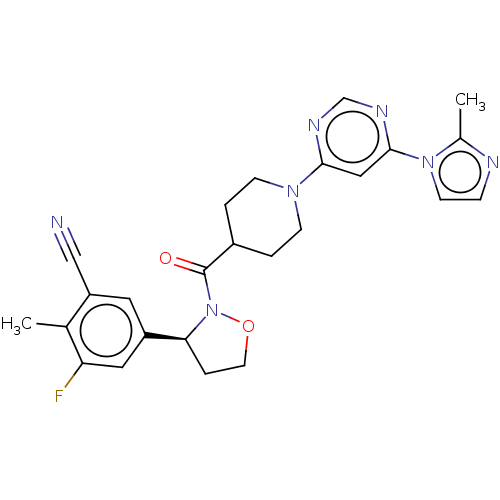
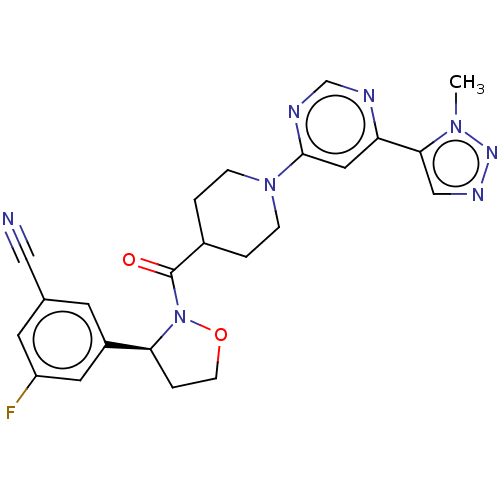
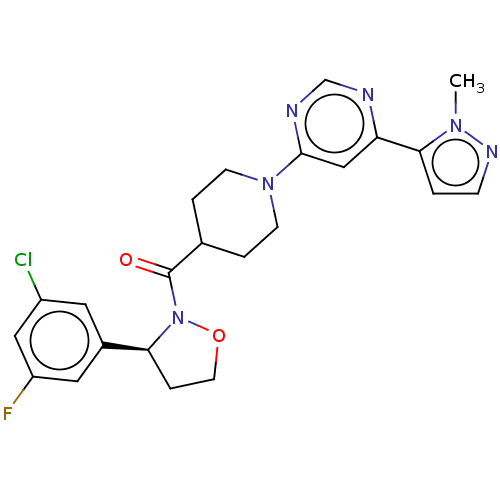
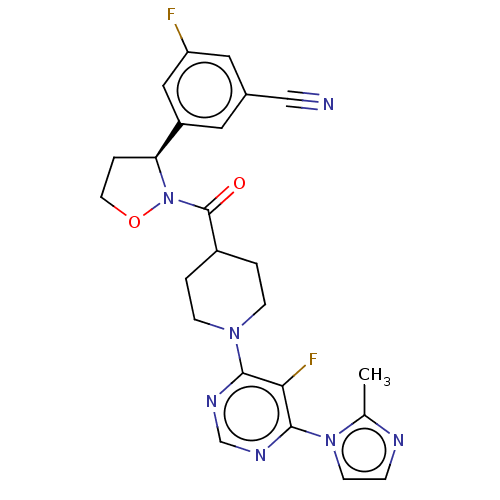
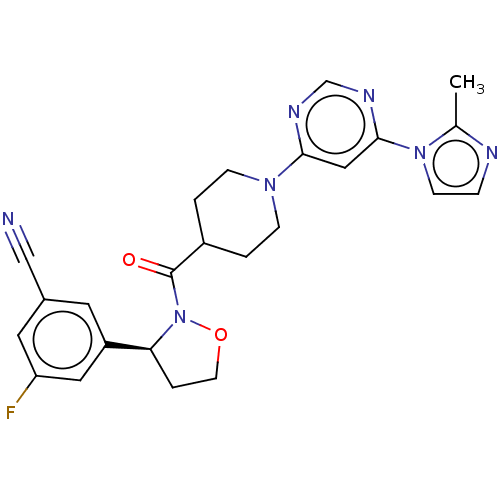
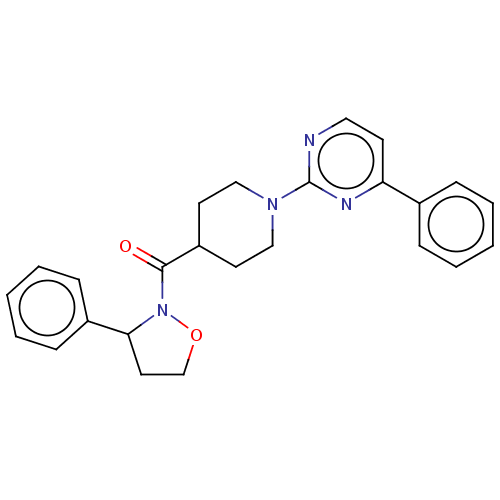
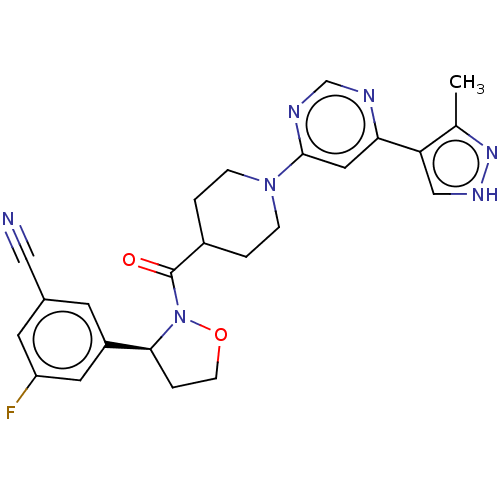
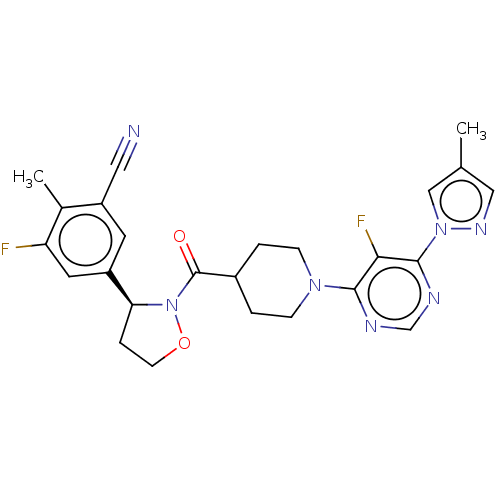
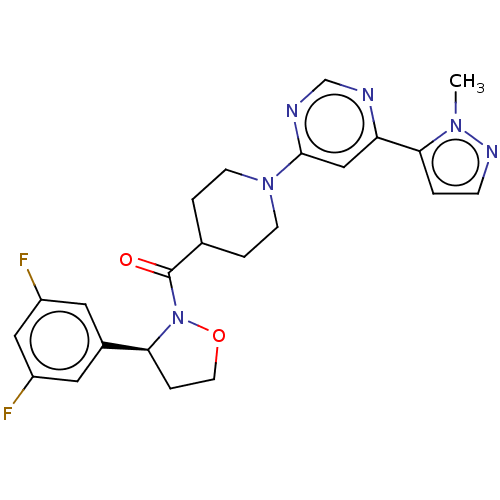
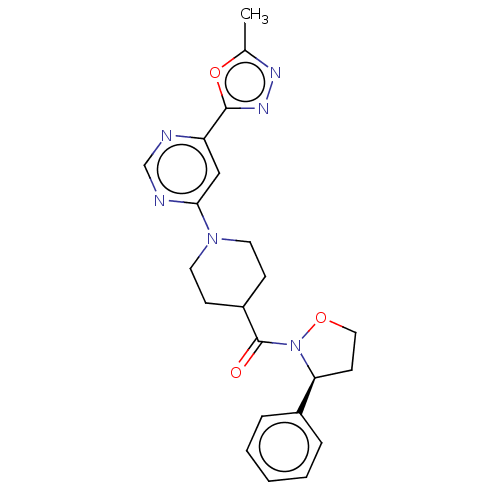
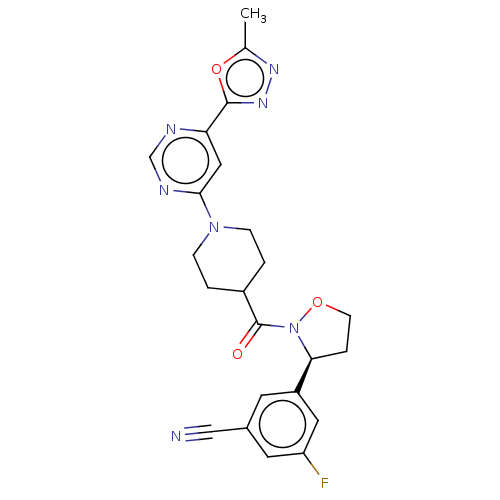
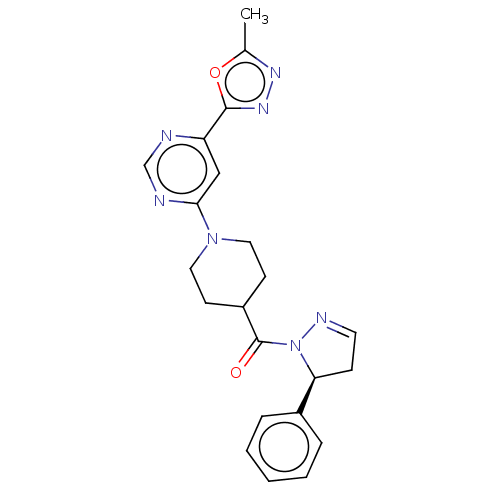
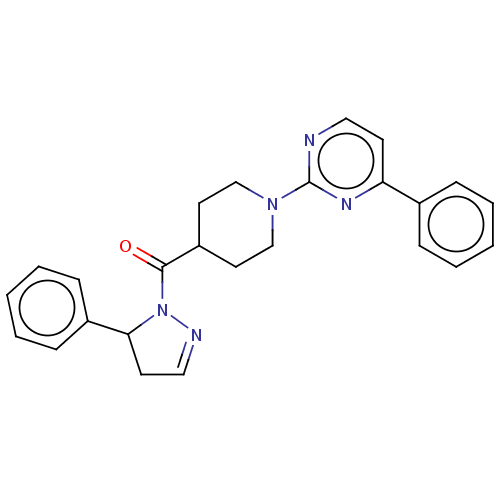
Report error Found 20 Enz. Inhib. hit(s) with all data for entry = 10174
Affinity DataIC50: 4.10nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 5.90nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 8.10nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 8.80nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 9.90nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 12.1nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 14.9nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 15.8nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 16.4nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 17.8nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 18.5nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 31nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 35nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 37nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 41nMAssay Description:In detail, 2 μl recombinantly produced hRIPK1 (aa 1-375) fusion protein (end concentration 3.6 μg/ml) and 2 μl compound (end concentra...More data for this Ligand-Target Pair