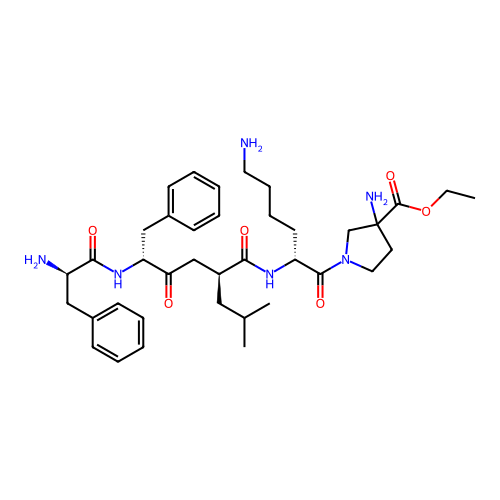
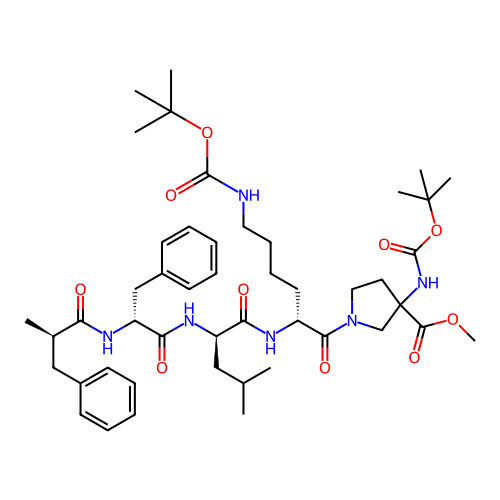
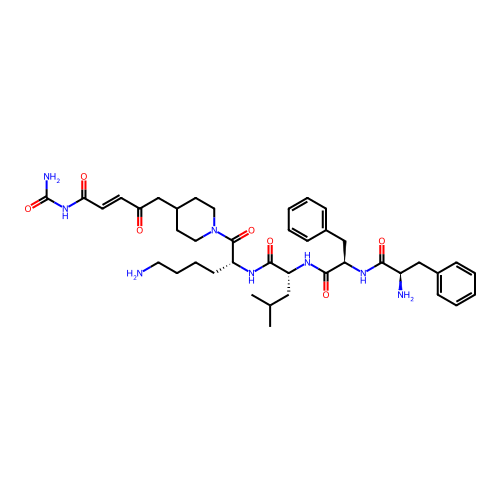
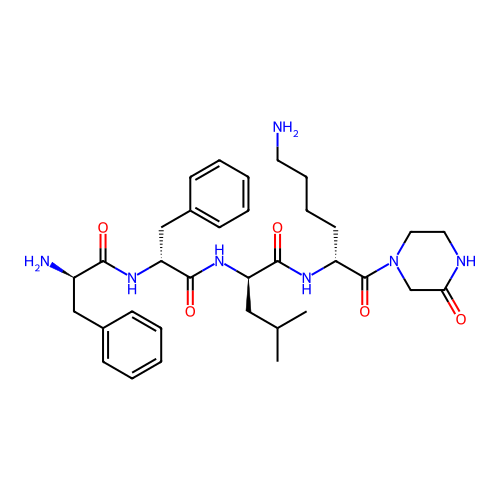
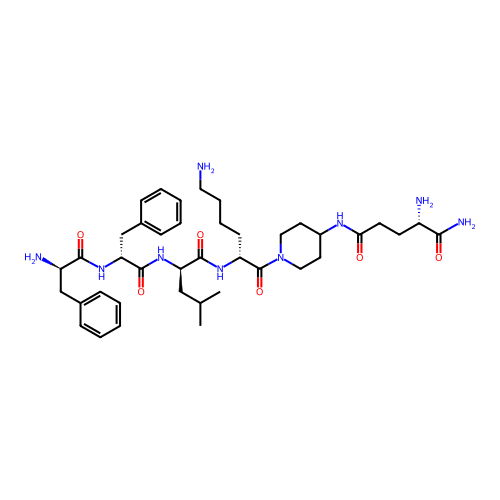
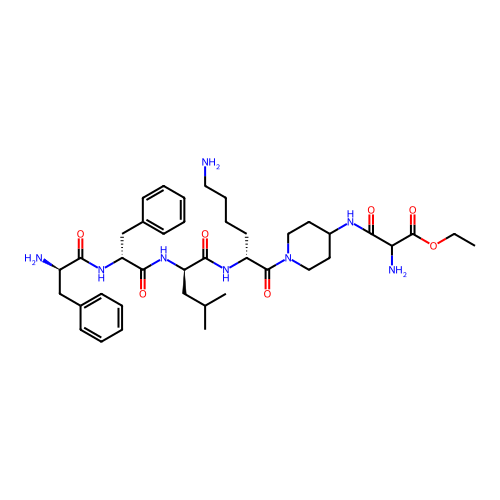
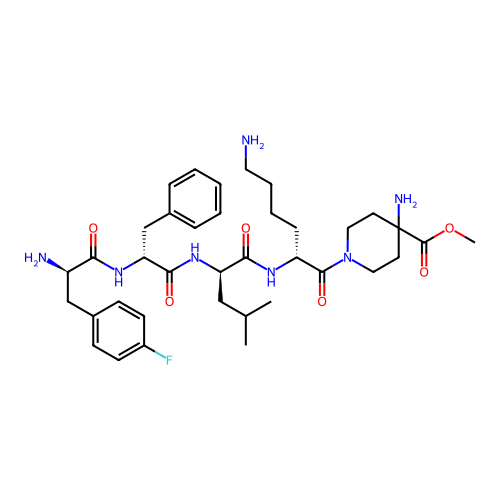
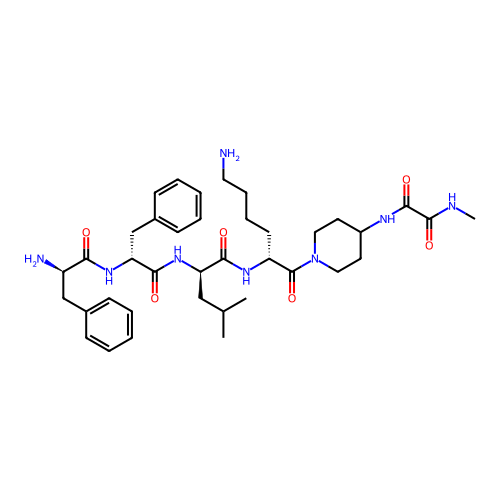
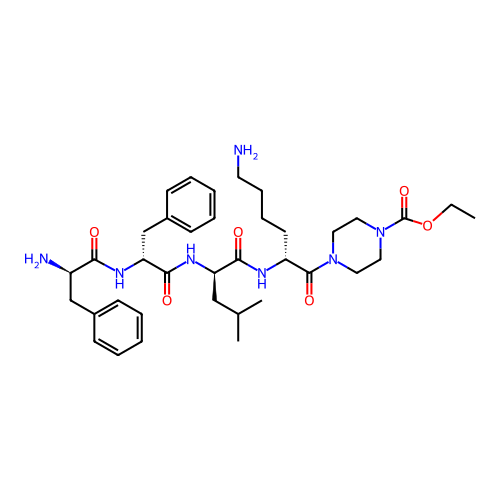
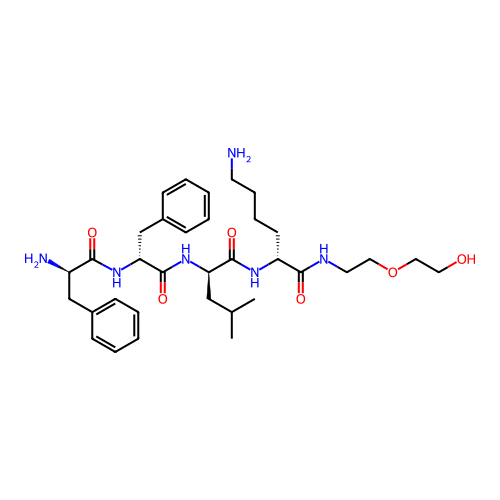
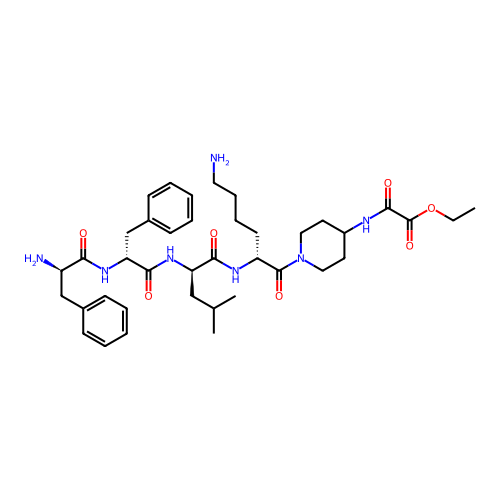
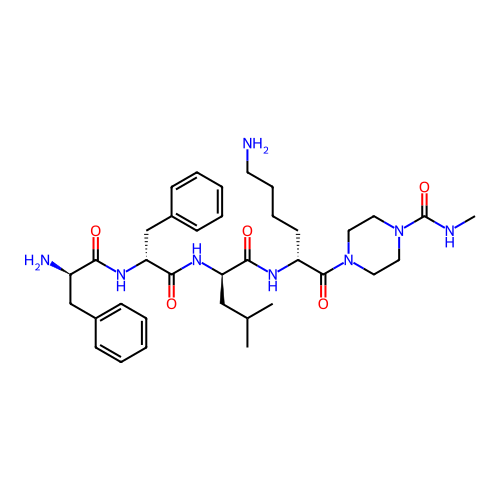
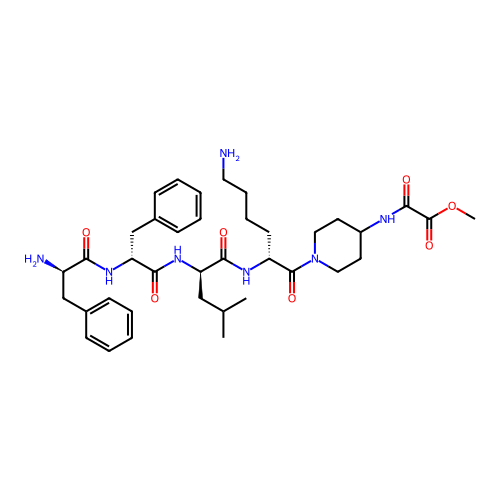
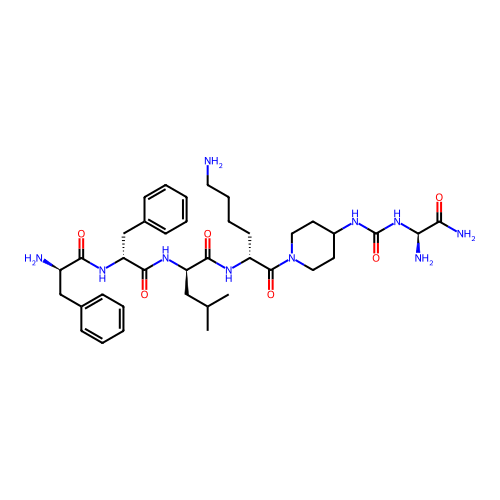
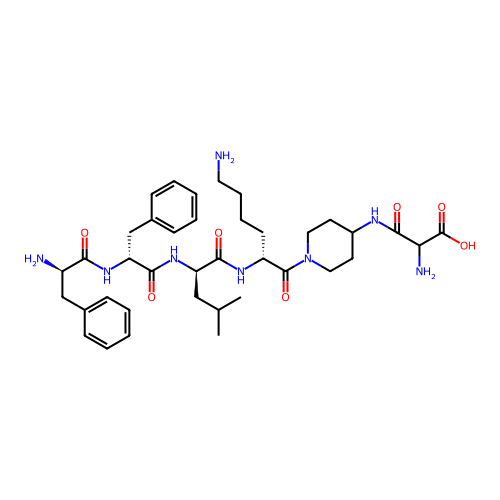
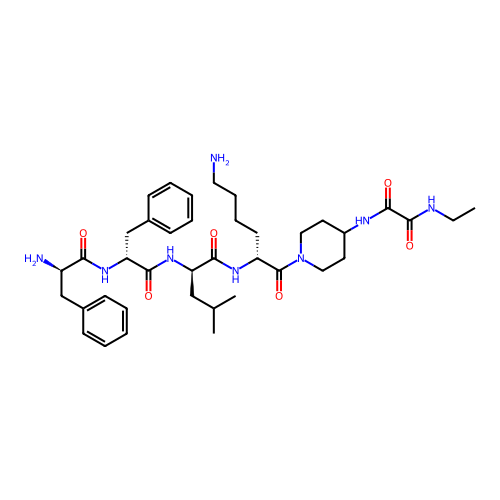
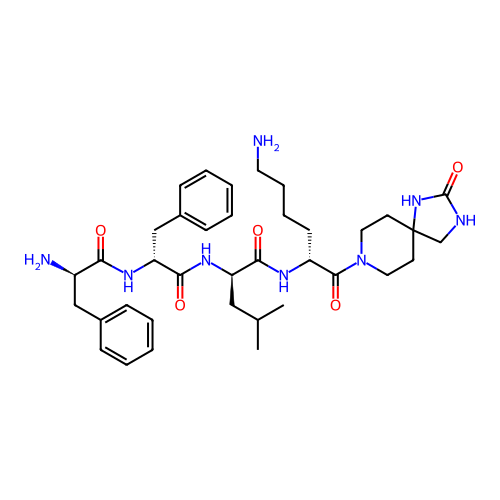
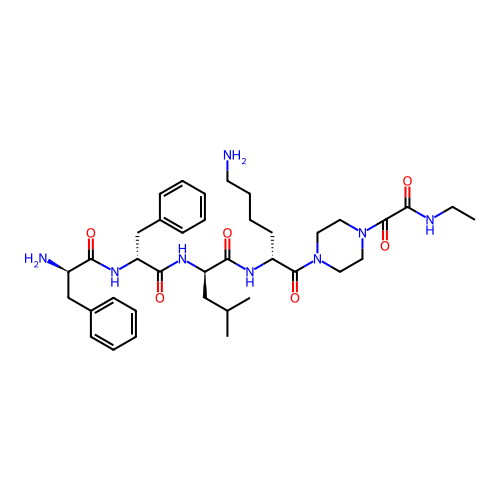
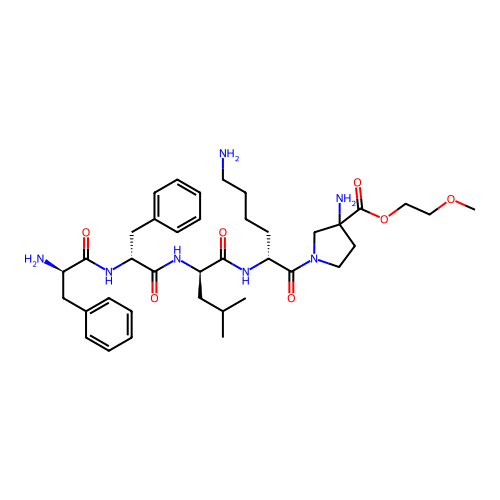
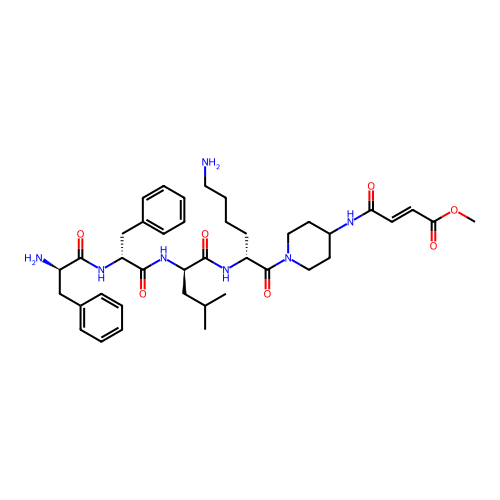
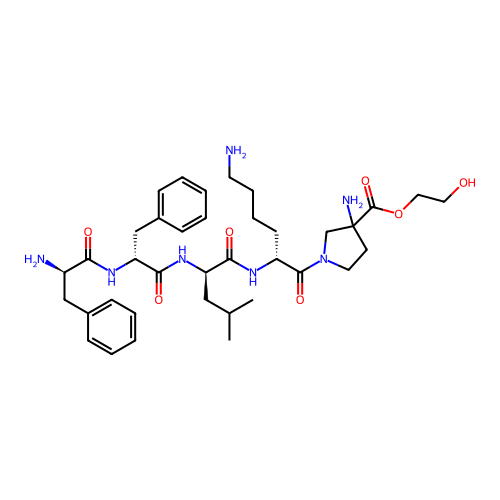
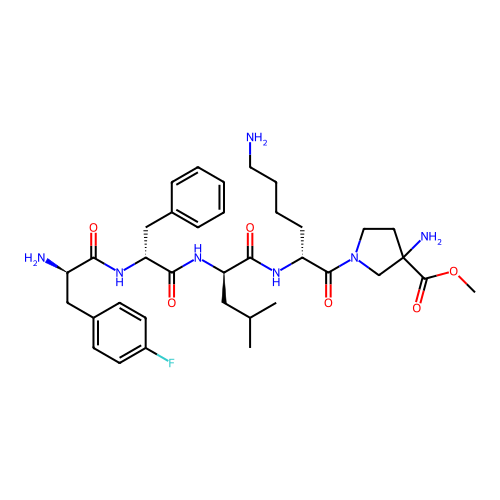
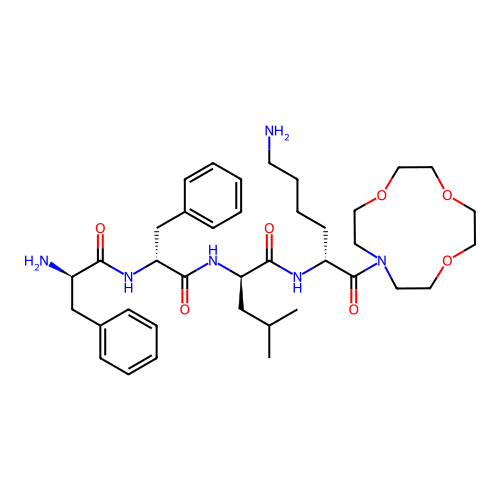
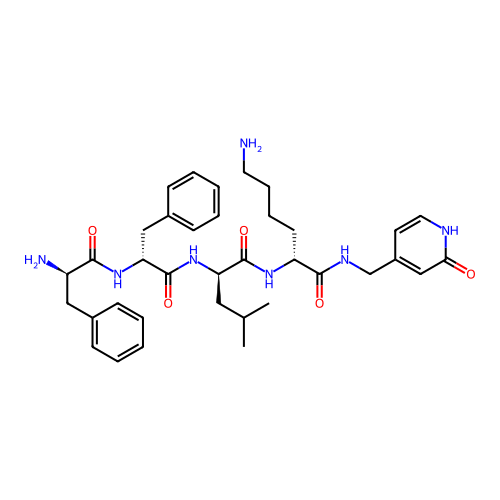
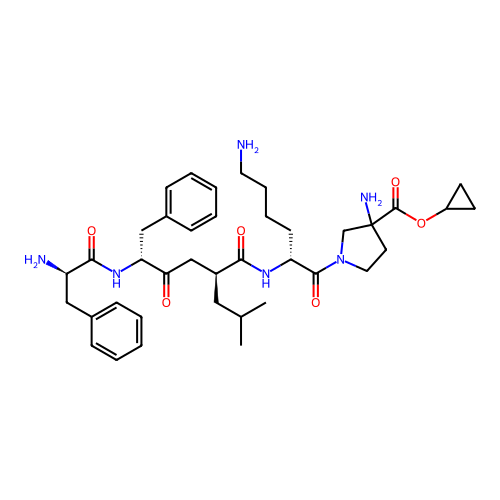
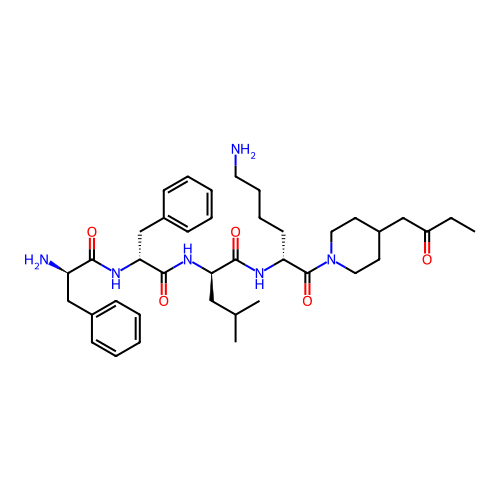
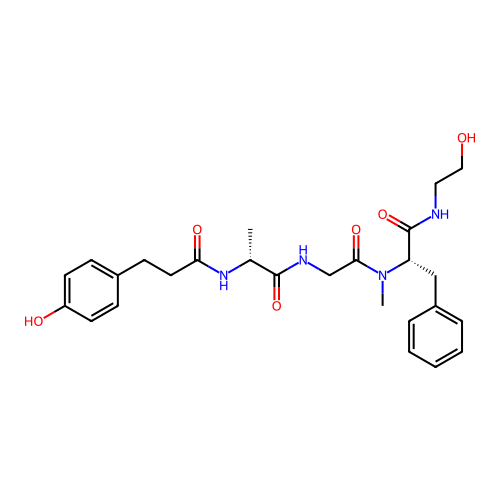
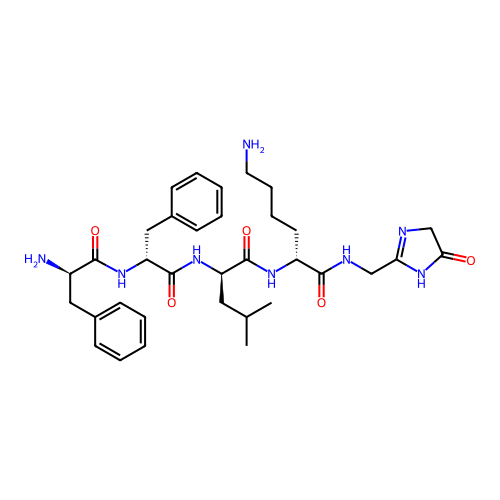
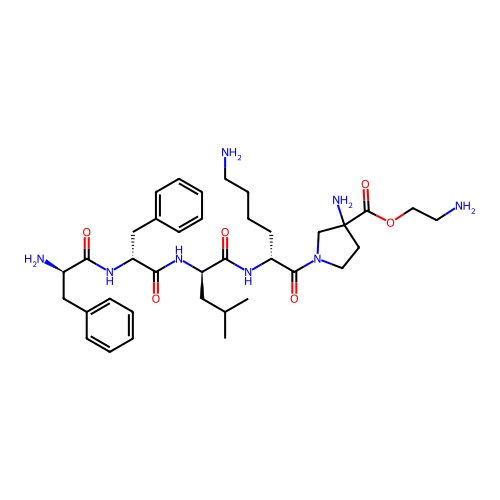
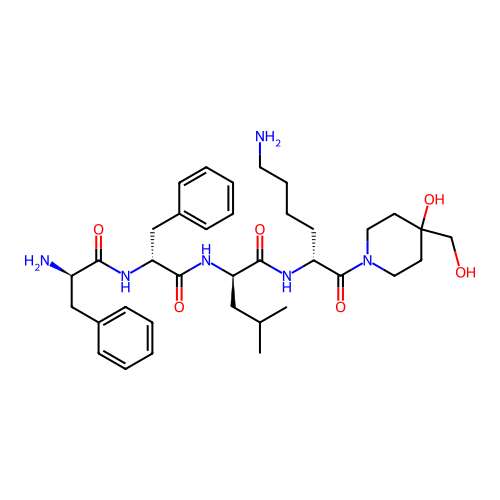
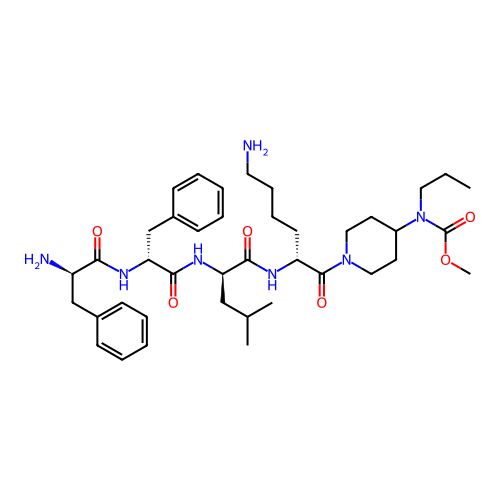
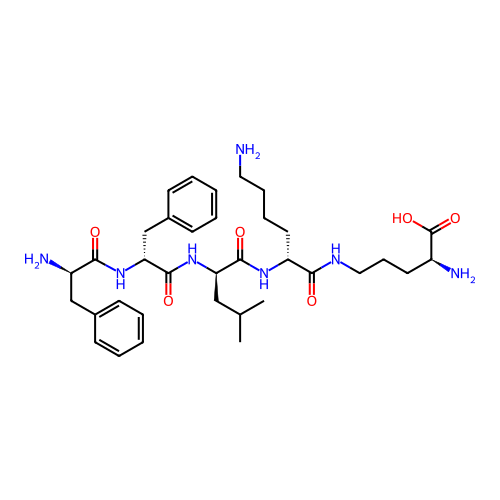
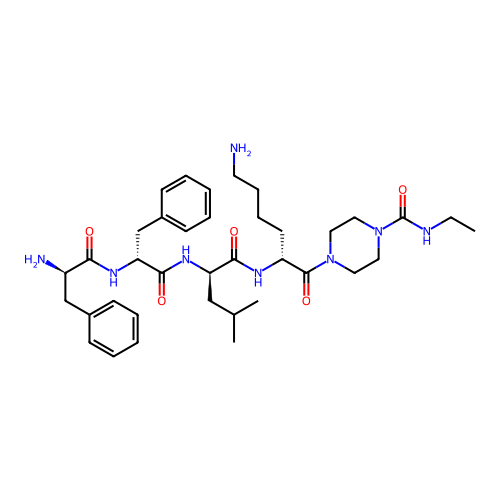
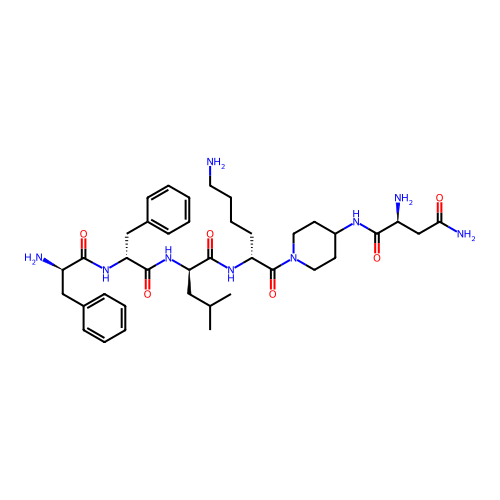
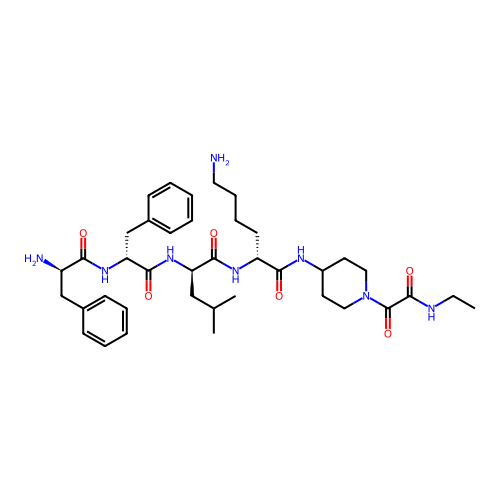
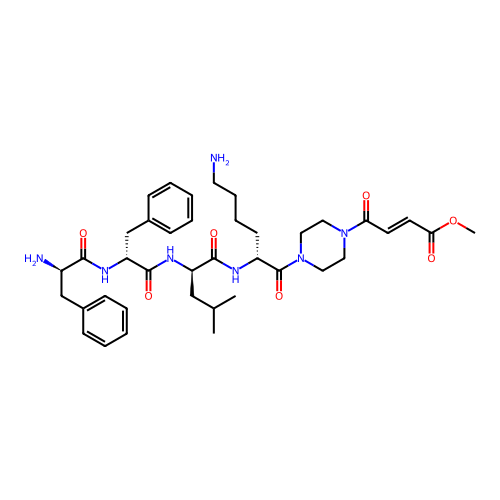
Report error Found 300 Enz. Inhib. hit(s) with all data for entry = 12650
Affinity DataIC50: 0.0960nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.150nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.160nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.170nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.183nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.190nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.210nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.210nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.210nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.230nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.25nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.260nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.270nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.270nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.280nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.290nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.290nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.310nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.310nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.330nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.330nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.330nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.330nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.350nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.350nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.360nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.360nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.360nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.370nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.370nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.390nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.400nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.410nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.410nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.410nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.420nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.426nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.430nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.450nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.450nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.460nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.460nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.470nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.480nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.490nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
Affinity DataIC50: 0.520nMAssay Description:The assay buffers used for opioid receptor binding studies were 50 mM Tris HCl (pH 7.4) for KOR, 50 mM Tris HCl (pH 7.4) with 5 mM MgCl2 for MOR, and...More data for this Ligand-Target Pair
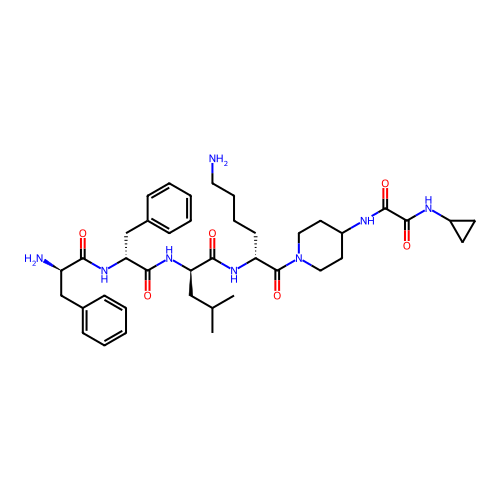
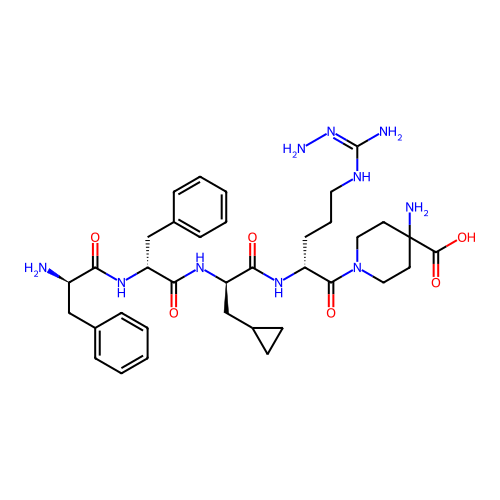
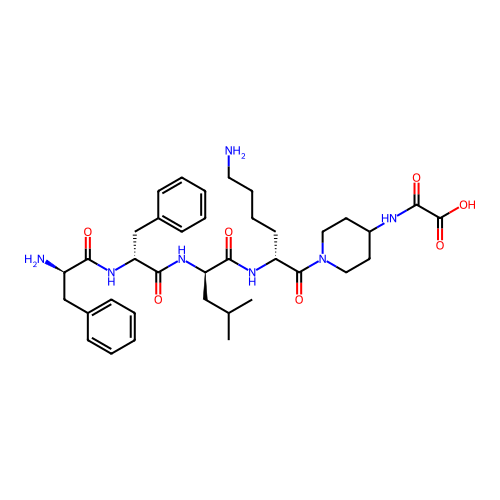
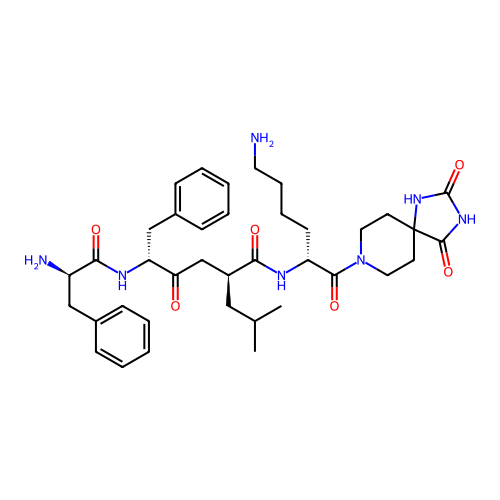
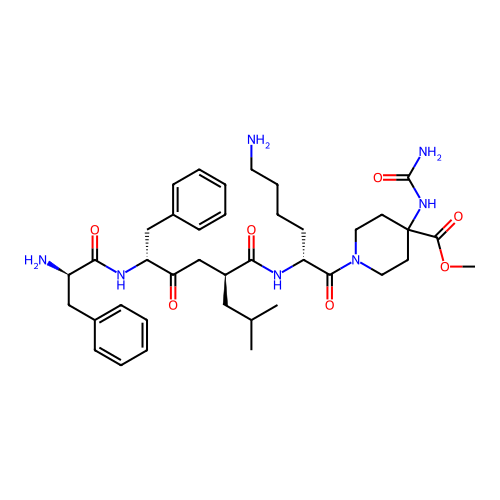
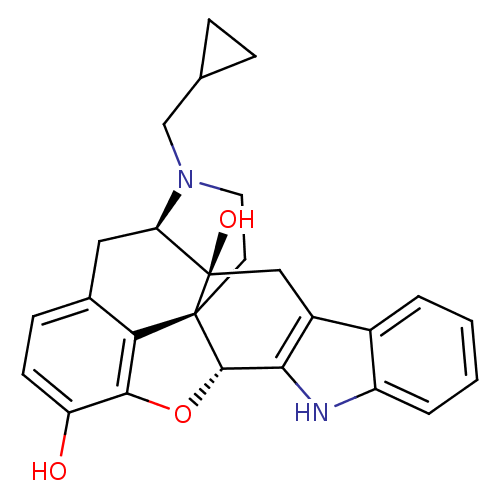
 3D Structure (crystal)
3D Structure (crystal)