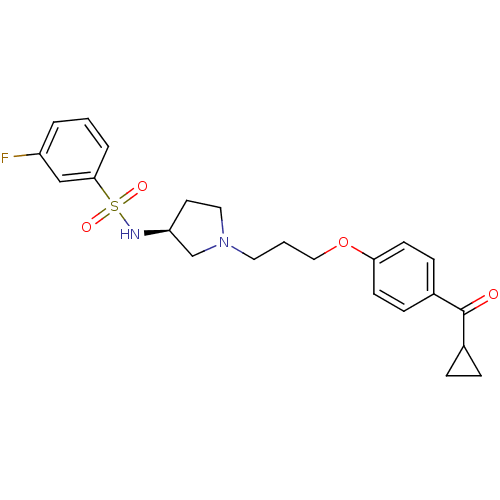
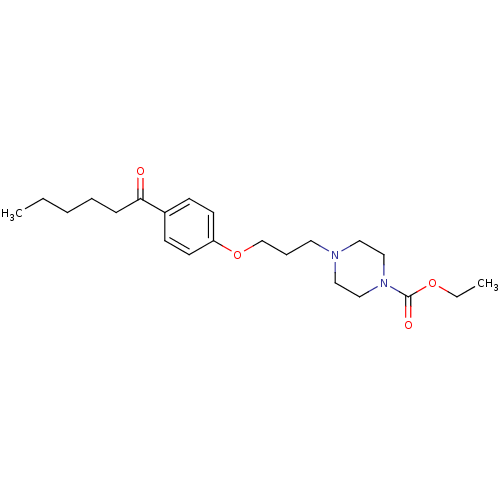
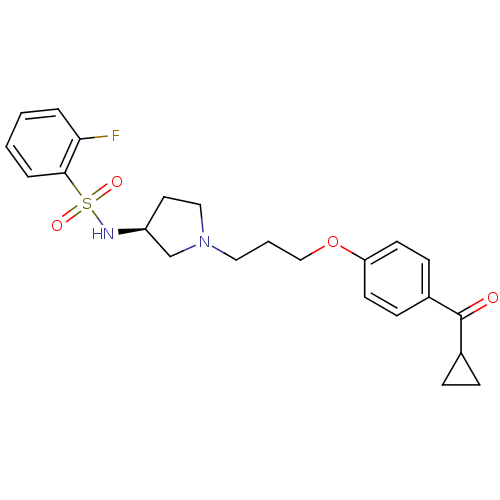
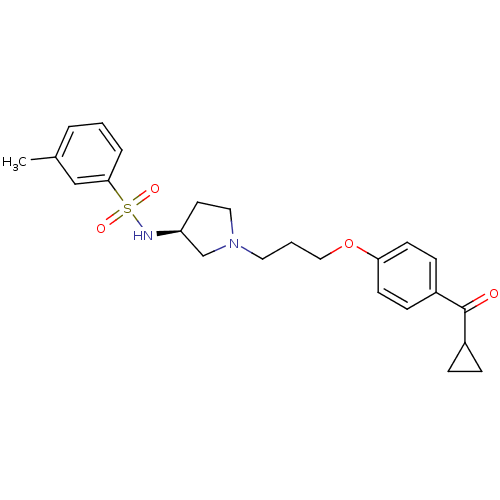
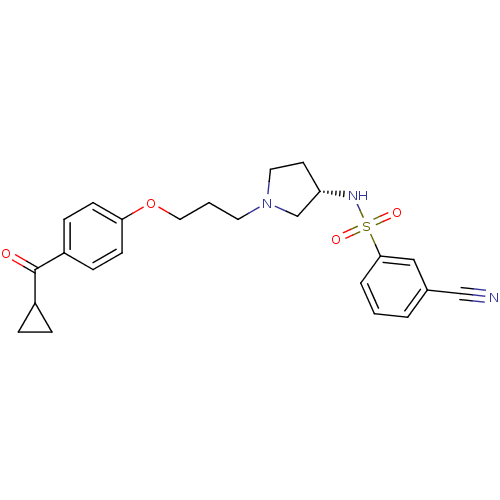
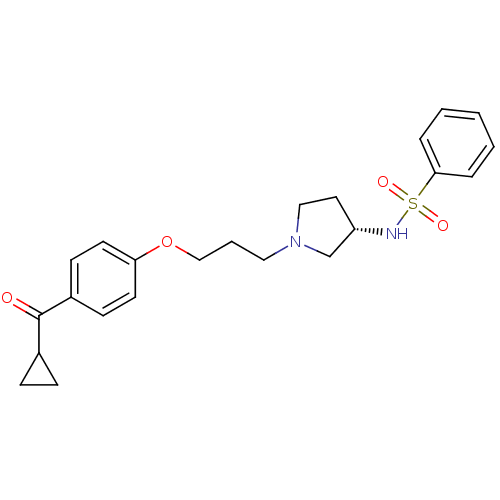
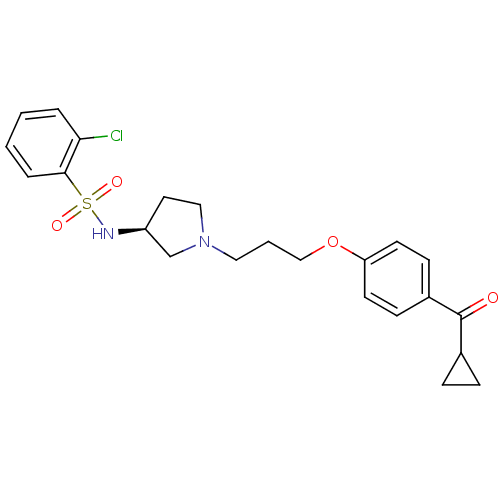
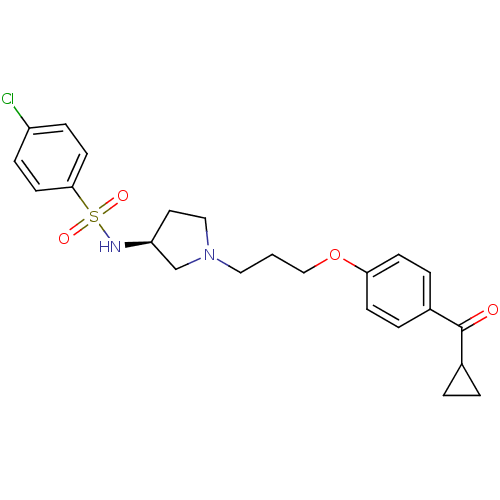
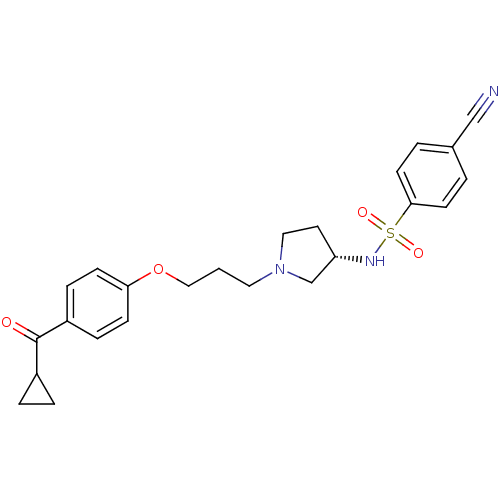
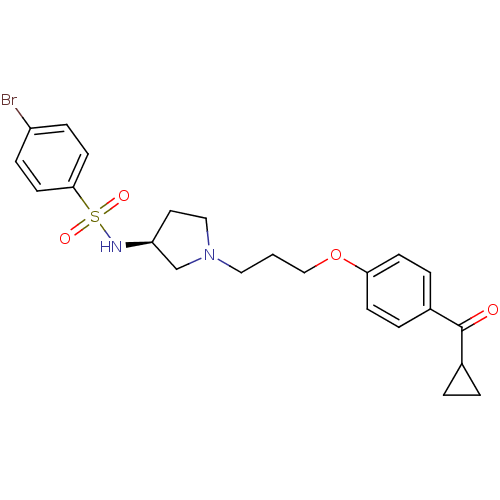
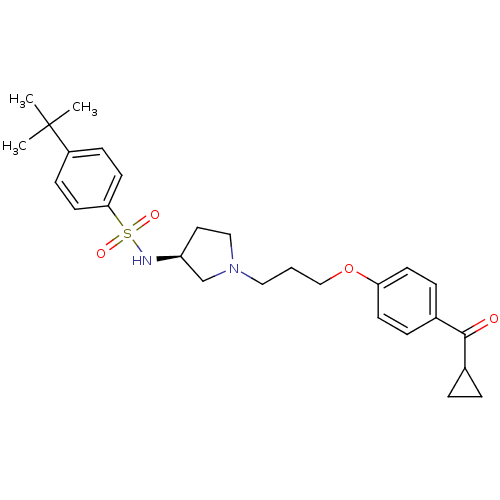
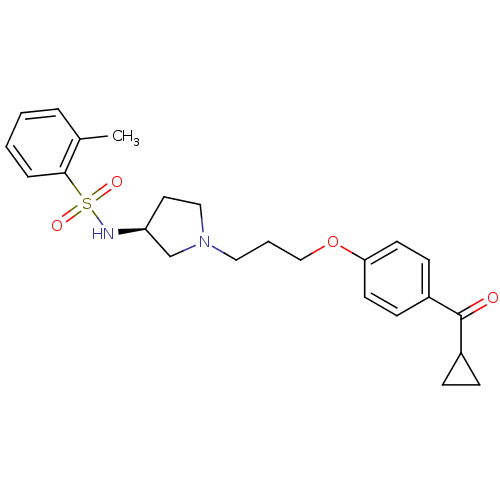
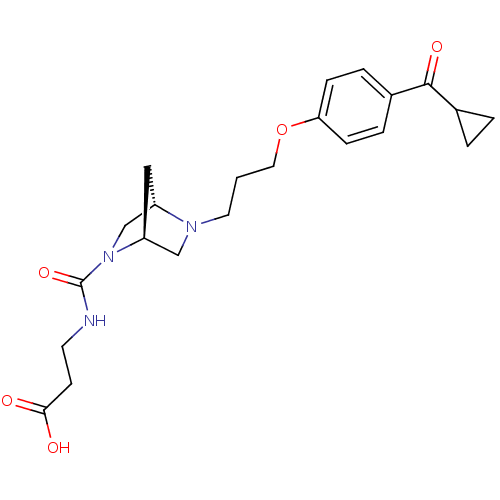
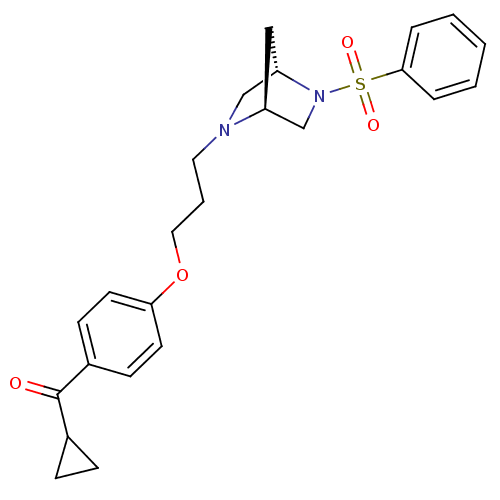
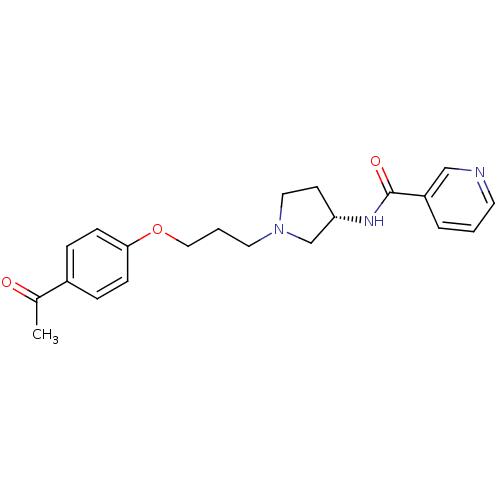
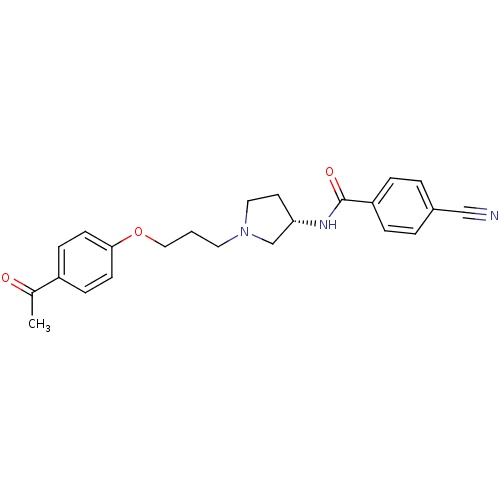
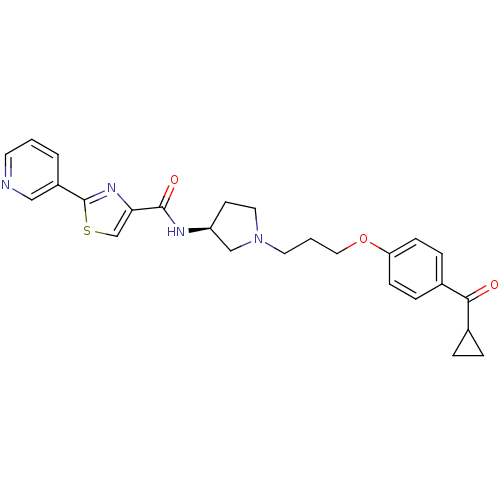
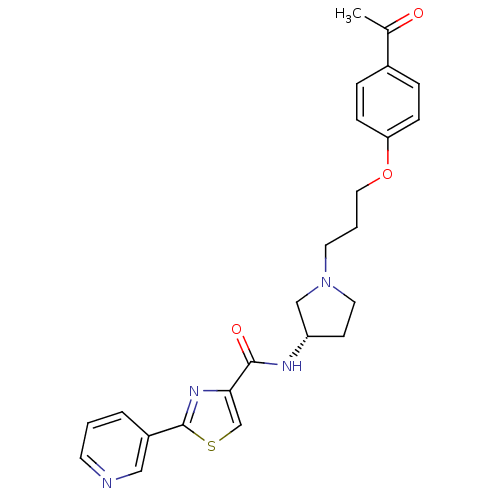
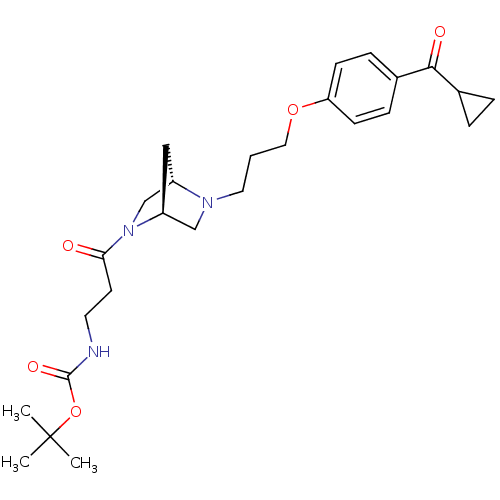
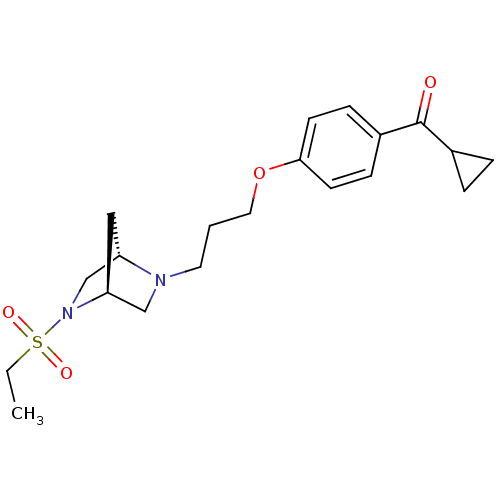
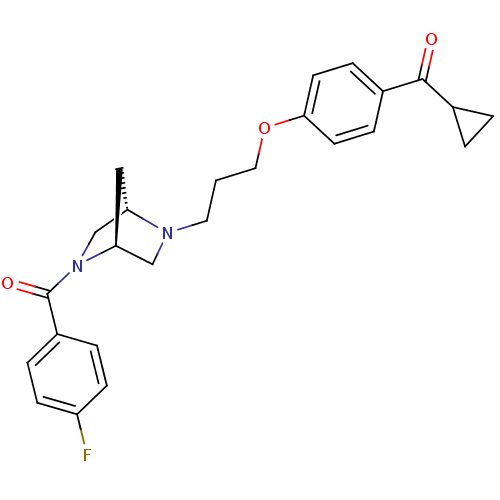
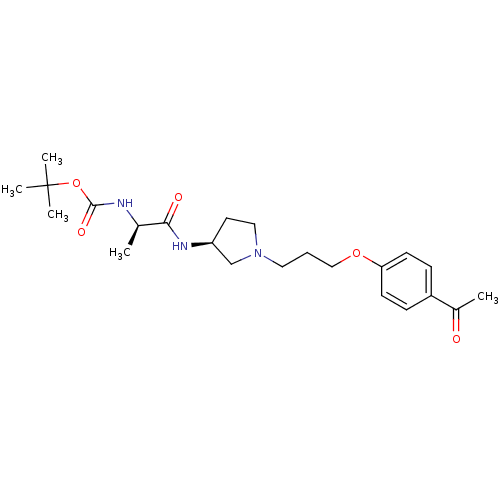
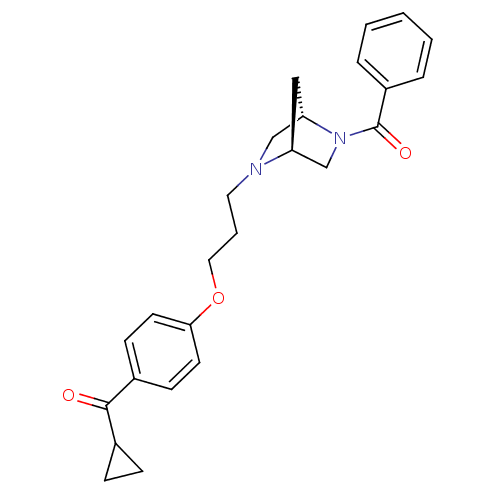
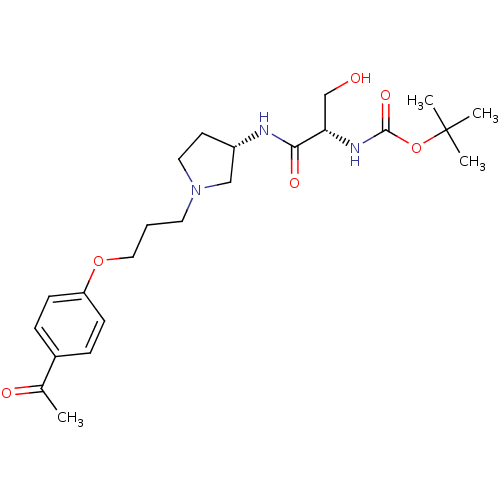
Report error Found 108 Enz. Inhib. hit(s) with all data for entry = 50012508
Affinity DataKi: 1.60nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 2.70nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 2.70nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 2.80nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 2.90nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 3.5nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 3.70nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 3.80nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 4nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 4.20nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 4.60nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 5nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 5.20nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 6nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 10nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 17nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 19nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 37nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 40nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 50nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 55nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 71nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 120nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 134nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 151nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 191nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 202nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 208nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 232nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 258nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 290nMAssay Description:Binding affinity towards human Histamine H2 receptor (For compound 11)More data for this Ligand-Target Pair
Affinity DataKi: 290nMAssay Description:Binding affinity towards human Histamine H2 receptor (For compound 11)More data for this Ligand-Target Pair
Affinity DataKi: 307nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 310nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 311nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 330nMAssay Description:Binding affinity towards human Histamine H2 receptor (For compound 11)More data for this Ligand-Target Pair
Affinity DataKi: 344nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 380nMAssay Description:Binding affinity towards human Histamine H2 receptor (For compound 11)More data for this Ligand-Target Pair
Affinity DataKi: 413nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 500nMAssay Description:Binding affinity towards human Histamine H2 receptor (For compound 11)More data for this Ligand-Target Pair
Affinity DataKi: 587nMAssay Description:Binding affinity towards rats Histamine type 3 (H3) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 676nMAssay Description:Binding affinity towards rat Histamine H3 receptor (For compound 11)More data for this Ligand-Target Pair
Affinity DataKi: 700nMAssay Description:Binding affinity towards human Histamine H2 receptor (For compound 11)More data for this Ligand-Target Pair
Affinity DataKi: 710nMAssay Description:Binding affinity towards human Histamine H2 receptor (For compound 11)More data for this Ligand-Target Pair
Affinity DataKi: 730nMAssay Description:Binding affinity towards human Histamine H2 receptor (For compound 11)More data for this Ligand-Target Pair