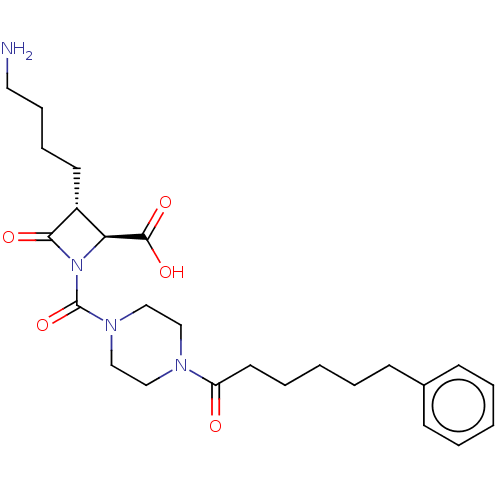
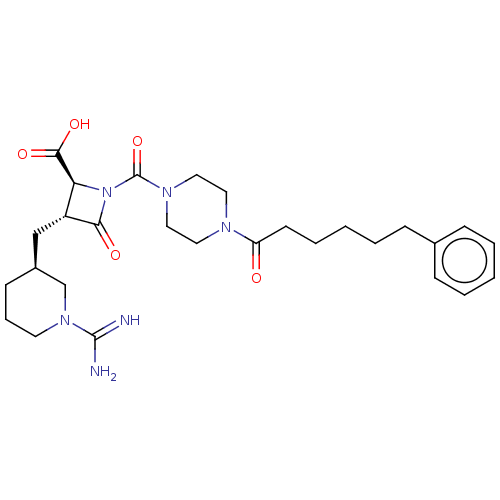
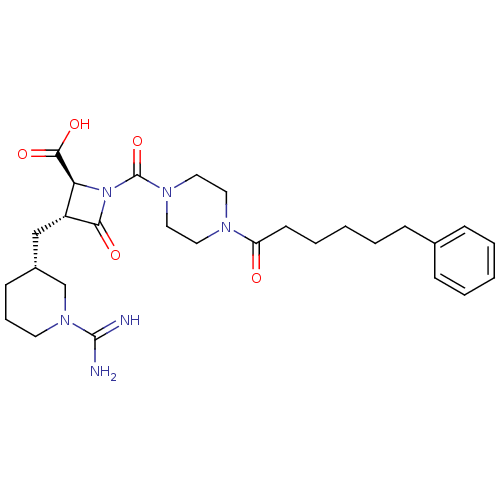
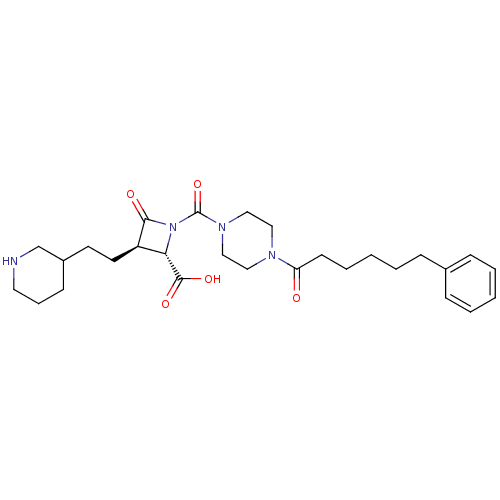
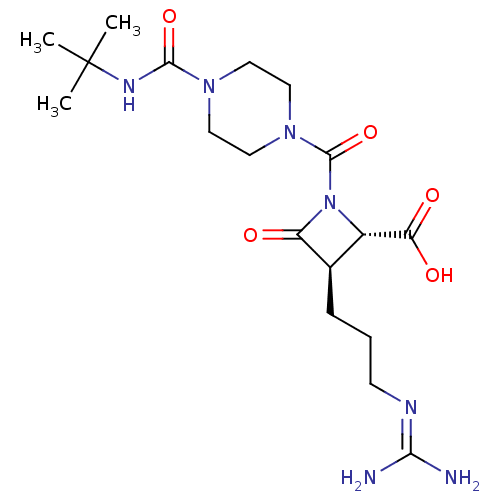
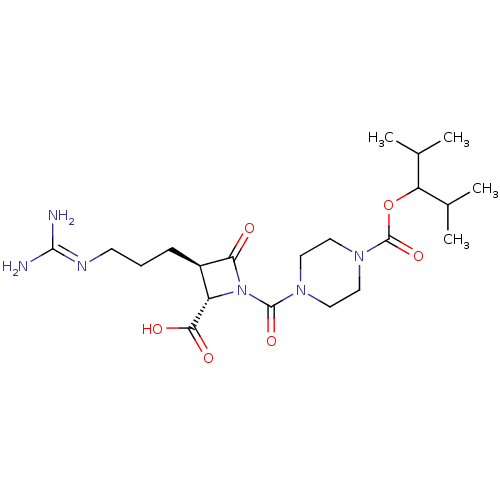
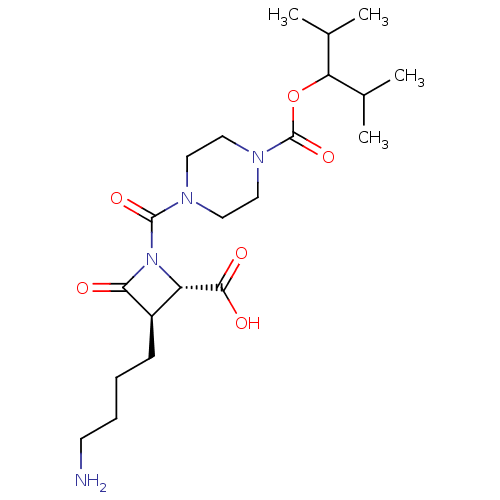
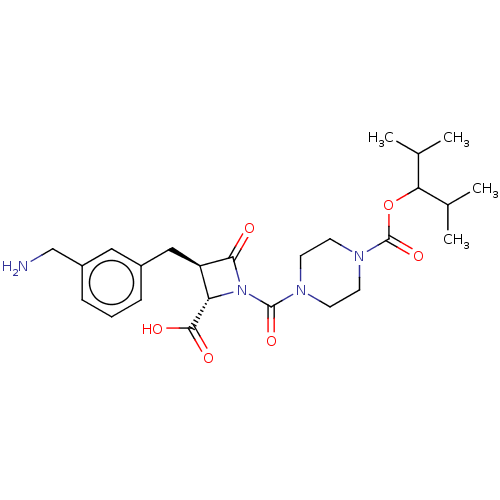
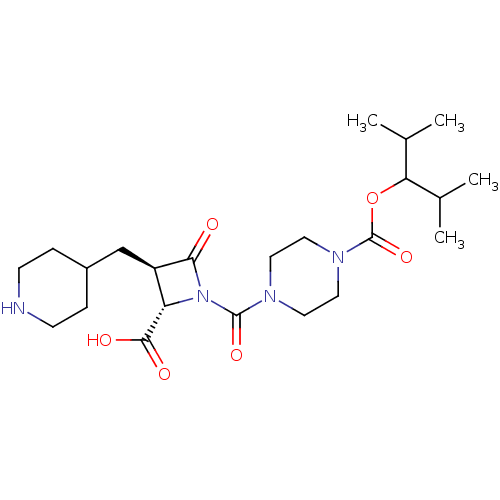
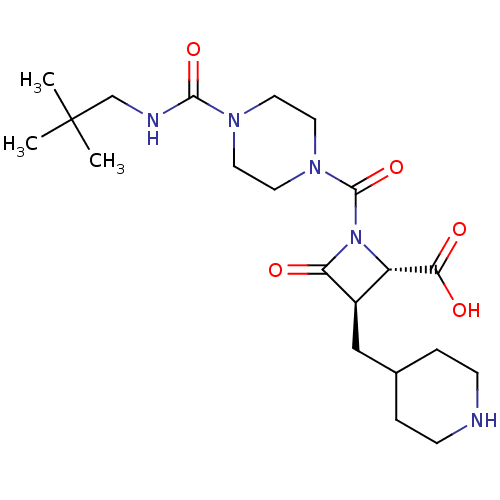
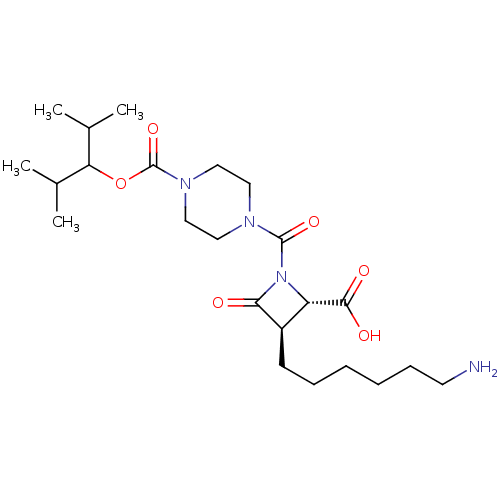
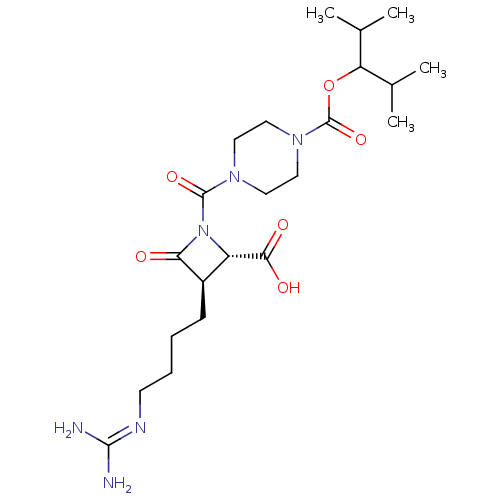
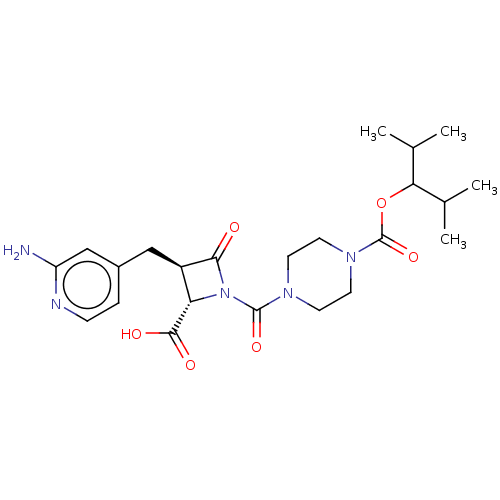
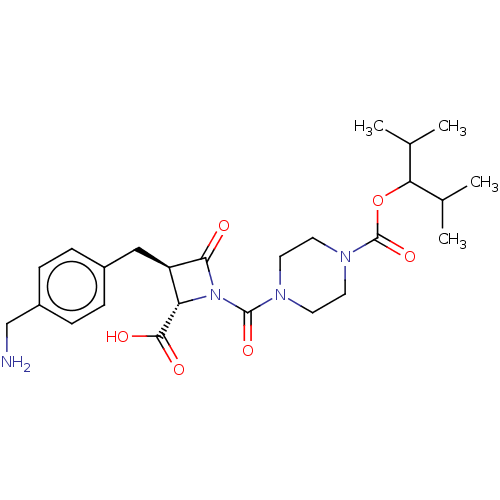
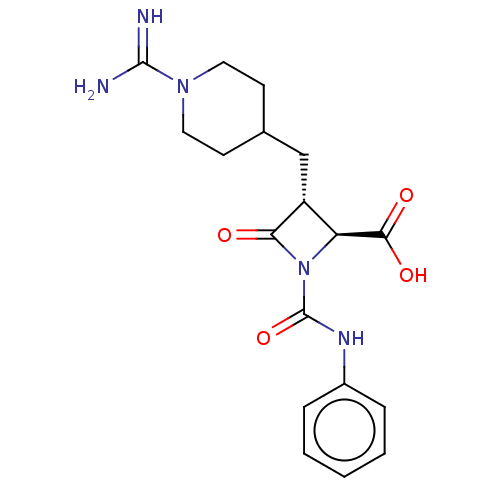
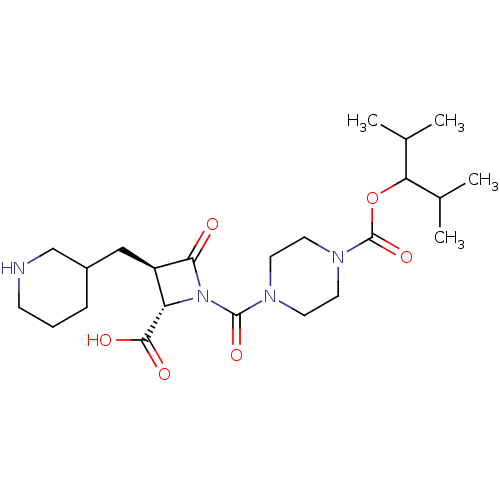
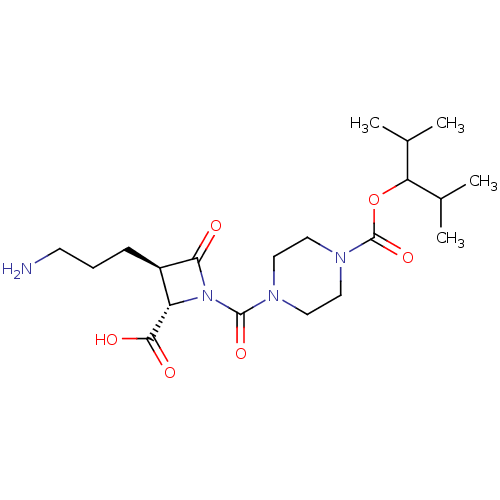
Report error Found 35 Enz. Inhib. hit(s) with all data for entry = 50014563
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.70nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.70nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.70nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.80nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 7nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 19nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 29nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 32nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 34nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 39nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 61nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 147nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 211nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 309nMAssay Description:Inhibitory activity of compound against Plasmin was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 515nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetSerine protease 1(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 935nMAssay Description:Inhibitory activity of compound against Trypsin was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.05E+3nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.21E+3nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 2.61E+3nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 6.22E+3nMAssay Description:Inhibitory activity of compound against Plasmin was determinedMore data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 7.00E+3nMAssay Description:Inhibitory activity of compound against Urokinase-type plasminogen activator was determinedMore data for this Ligand-Target Pair
TargetSerine protease 1(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibitory activity of compound against Trypsin was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.79E+4nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3.30E+4nMAssay Description:Inhibitory activity of compound against Urokinase-type plasminogen activator was determinedMore data for this Ligand-Target Pair
TargetCoagulation factor X(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3.30E+4nMAssay Description:Inhibitory activity of compound against Coagulation factor X was determinedMore data for this Ligand-Target Pair
TargetTissue-type plasminogen activator(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3.30E+4nMAssay Description:Inhibitory activity of compound against tissue type plasminogen activator was determinedMore data for this Ligand-Target Pair
TargetTissue-type plasminogen activator(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3.30E+4nMAssay Description:Inhibitory activity of compound against tissue type plasminogen activator was determinedMore data for this Ligand-Target Pair
TargetCoagulation factor X(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3.30E+4nMAssay Description:Inhibitory activity of compound against Coagulation factor X was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 3.30E+4nMAssay Description:Inhibitory activity of compound against Thrombin was determinedMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Human)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3.30E+4nMAssay Description:Inhibitory activity of compound against human tryptase was determinedMore data for this Ligand-Target Pair
Affinity DataIC50: 3.30E+4nMAssay Description:Inhibitory activity of compound against Thrombin was determinedMore data for this Ligand-Target Pair