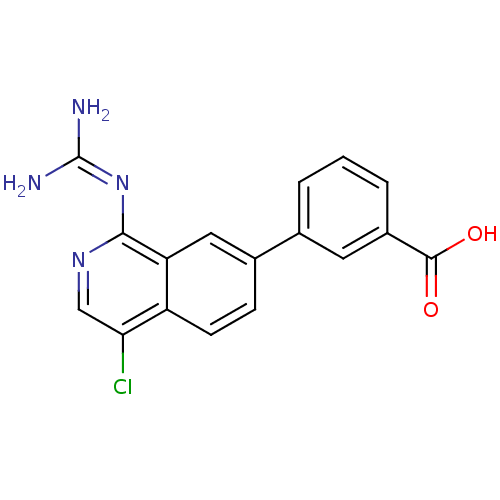
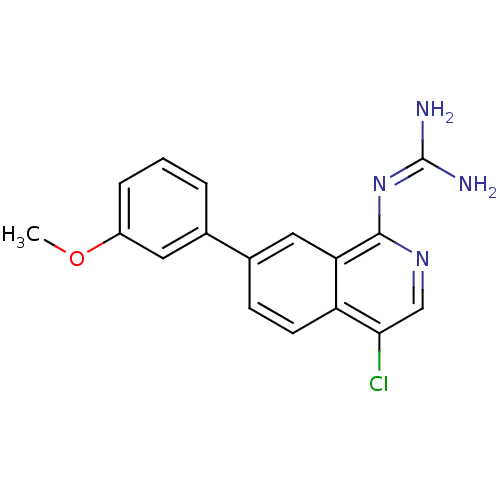
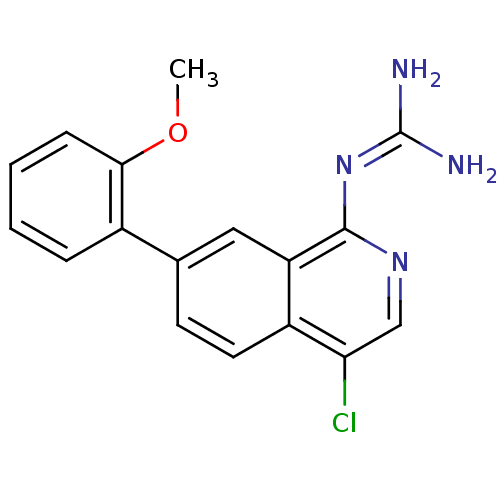
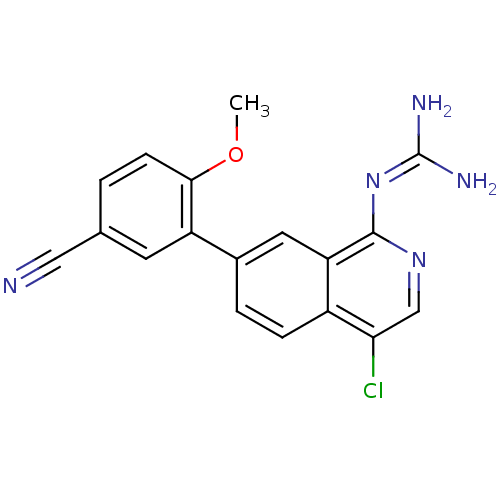
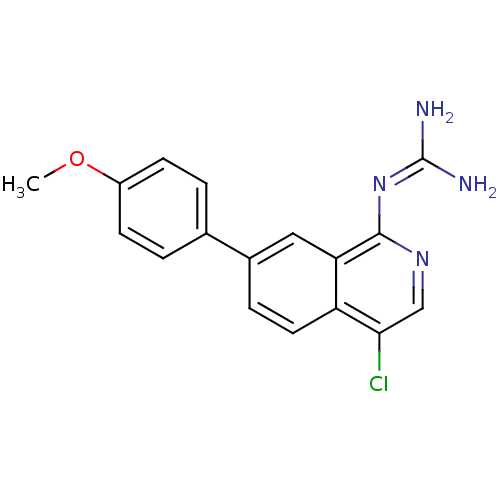
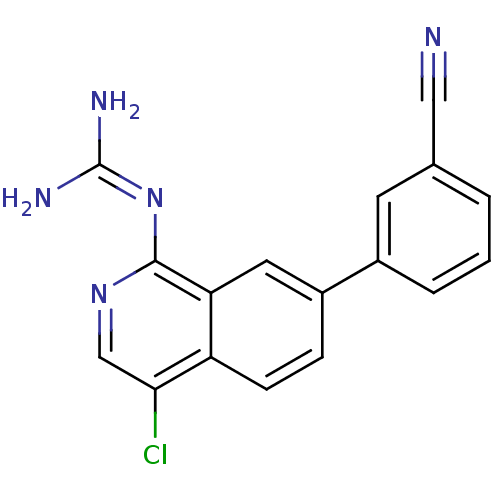
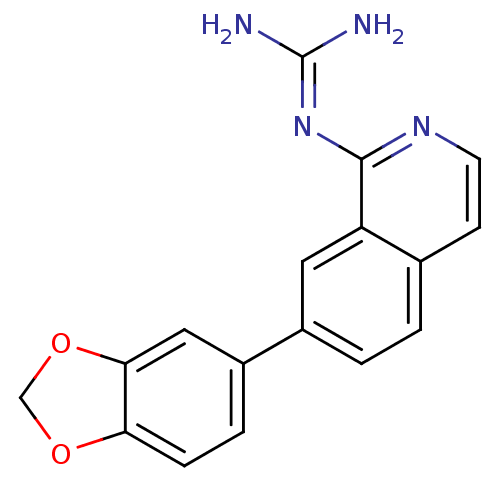
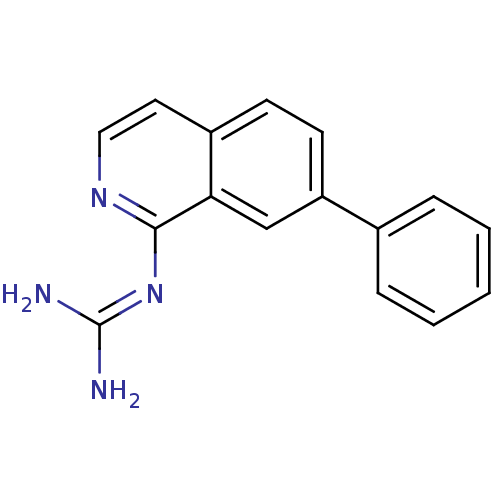
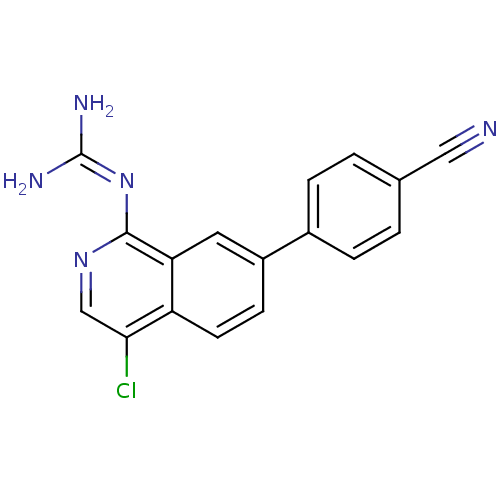
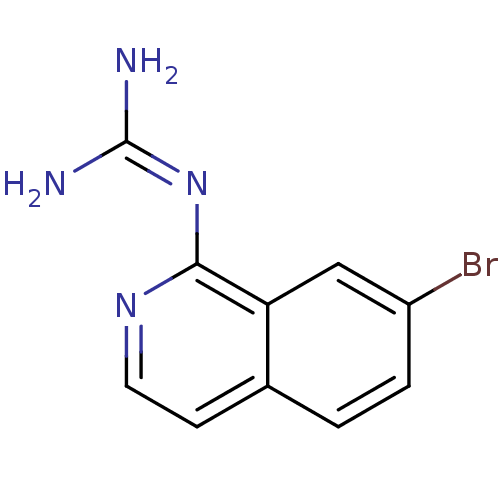
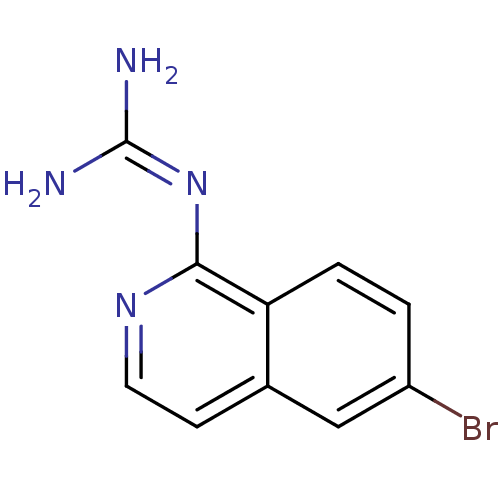
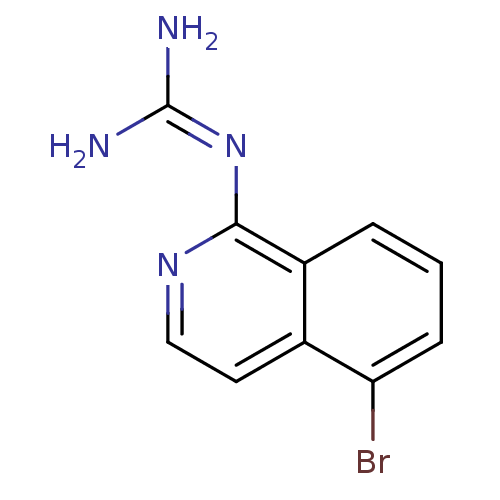
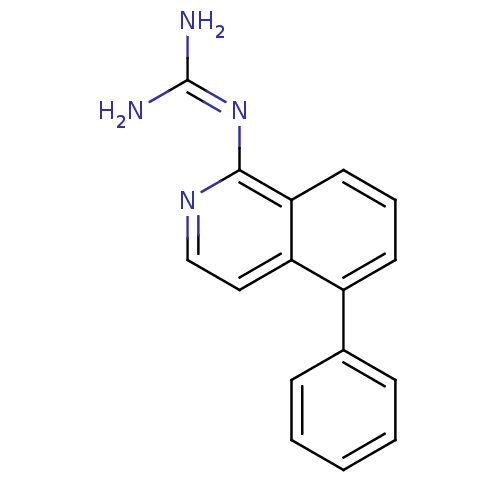
Report error Found 58 Enz. Inhib. hit(s) with all data for entry = 50014788
Affinity DataKi: 9nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 37nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 82nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 83nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 100nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 100nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 130nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 180nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 200nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 300nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 310nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 340nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 400nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 490nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 580nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 640nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 660nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 800nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 830nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 1.00E+3nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 1.30E+3nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 1.40E+3nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 1.92E+3nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 2.34E+3nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 3.00E+3nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 3.20E+3nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 3.62E+3nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 4.10E+3nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 4.75E+3nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 4.80E+3nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 4.80E+3nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 4.93E+3nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 7.30E+3nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 9.30E+3nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 1.20E+4nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 1.30E+4nMAssay Description:Inhibitory activity against human tissue type plasminogen activator using S-2444More data for this Ligand-Target Pair
Affinity DataKi: 1.30E+4nMAssay Description:Inhibitory activity against human tissue type plasminogen activator using S-2444More data for this Ligand-Target Pair
Affinity DataKi: 1.45E+4nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 1.55E+4nMAssay Description:Inhibitory activity against human tissue type plasminogen activator using S-2444More data for this Ligand-Target Pair
Affinity DataKi: 1.90E+4nMAssay Description:Inhibitory activity against human tissue type plasminogen activator using S-2444More data for this Ligand-Target Pair
Affinity DataKi: 2.40E+4nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 2.90E+4nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 4.75E+4nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 4.75E+4nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 5.08E+4nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 6.80E+4nMAssay Description:Inhibitory activity against human plasmin was evaluated using chromozym-PL as substrate at 1 mMMore data for this Ligand-Target Pair
Affinity DataKi: 9.51E+4nMAssay Description:Inhibitory activity against human tissue type plasminogen activator using S-2444More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+5nMAssay Description:Inhibitory activity against human urokinase-type plasminogen activator was evaluated using S-2444 as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 1.24E+5nMAssay Description:Inhibitory activity against human tissue type plasminogen activator using S-2444More data for this Ligand-Target Pair
Affinity DataKi: 1.24E+5nMAssay Description:Inhibitory activity against human tissue type plasminogen activator using S-2444More data for this Ligand-Target Pair