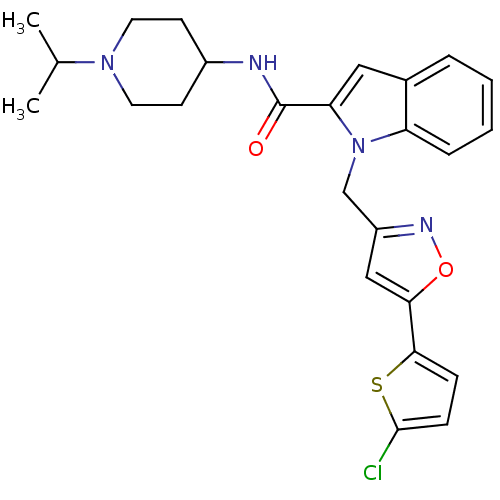
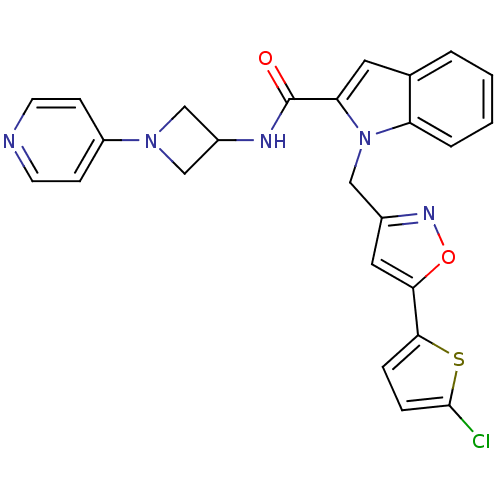
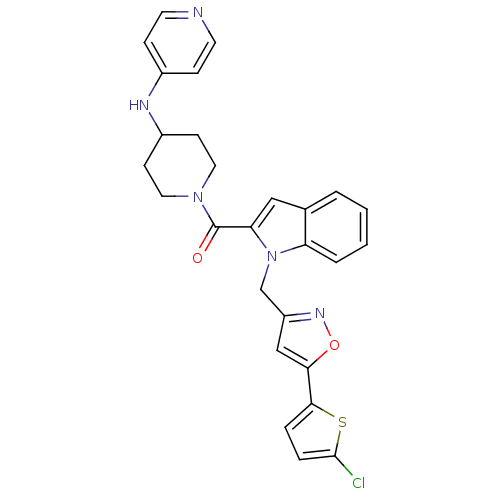
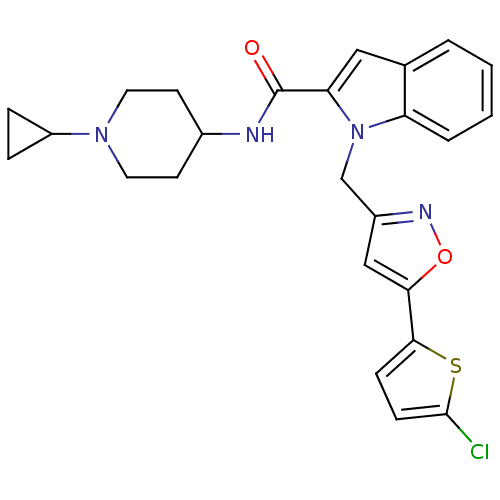
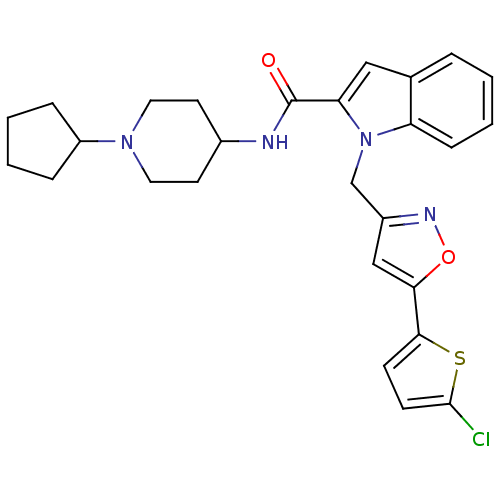
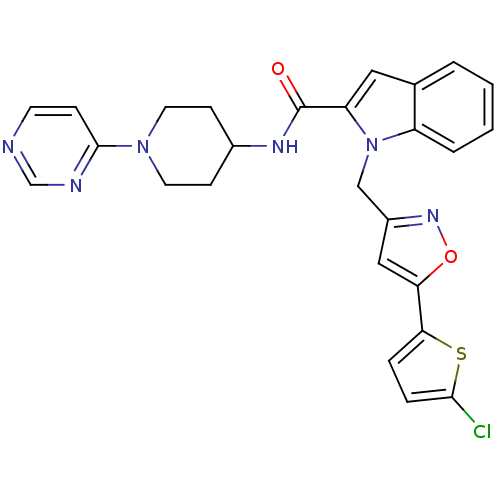
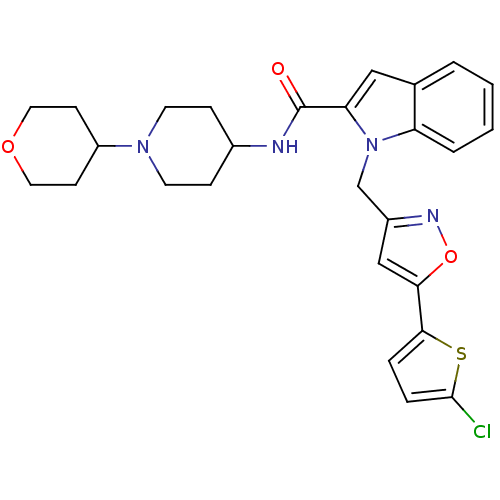
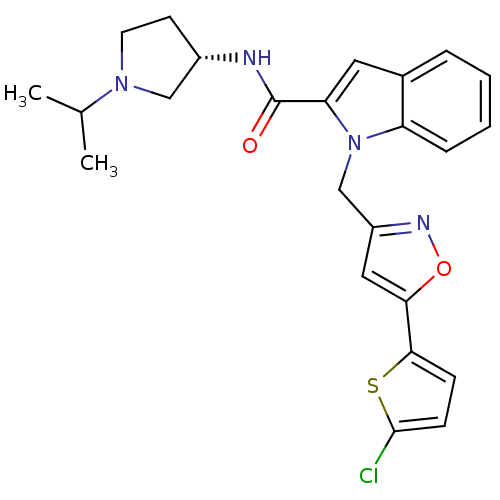
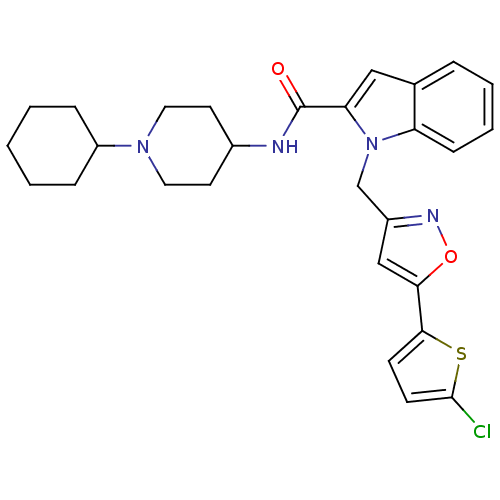
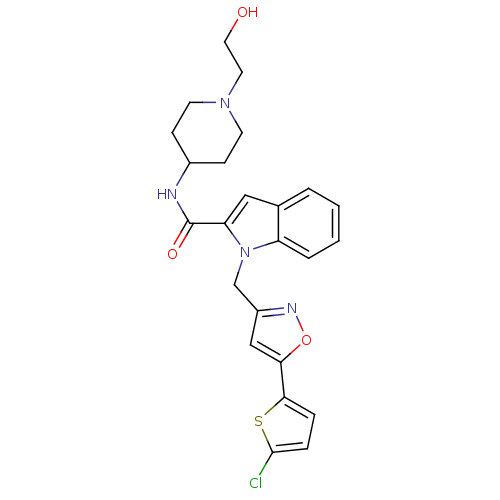
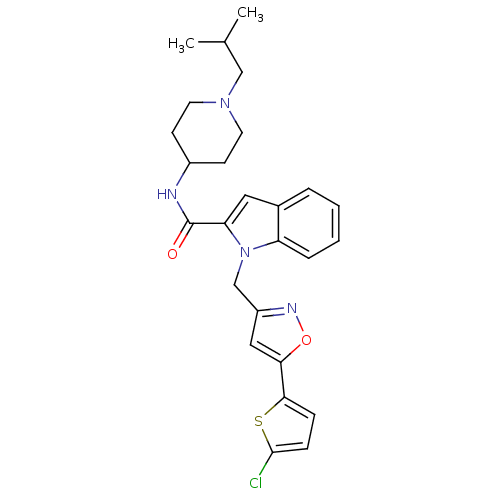
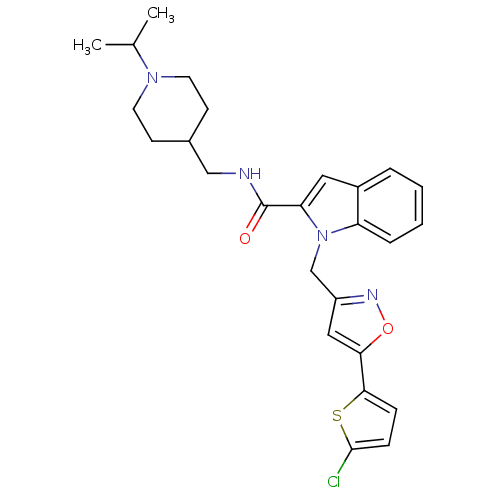
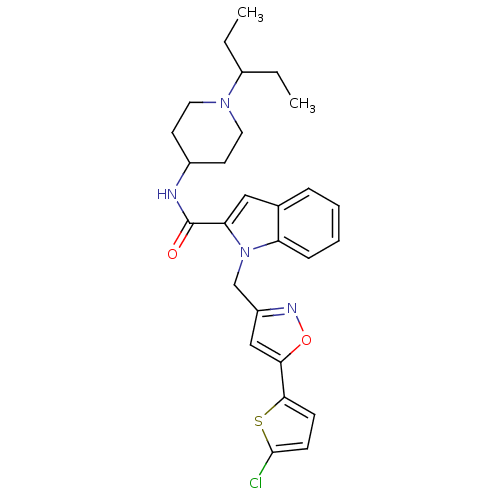
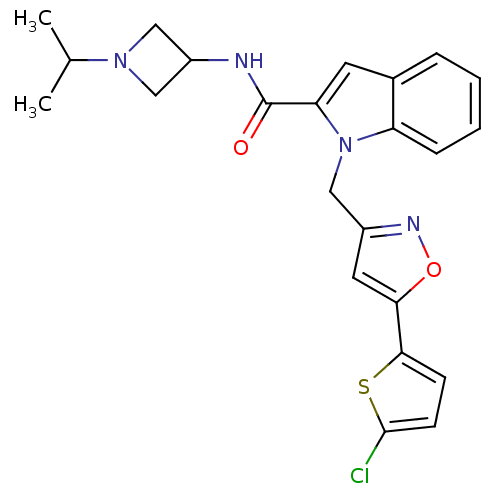
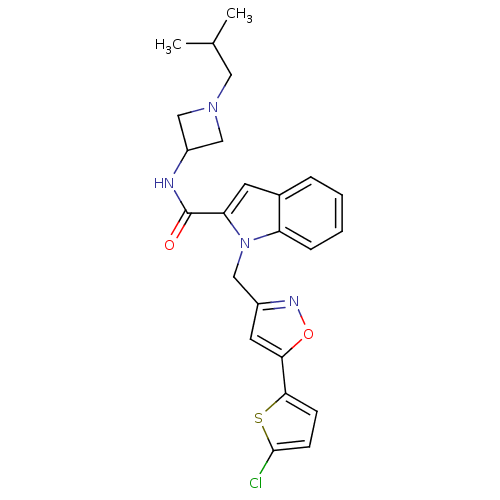
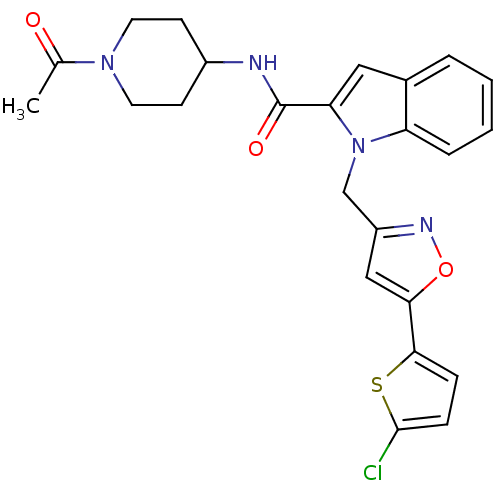
Report error Found 35 Enz. Inhib. hit(s) with all data for entry = 1987
Affinity DataKi: 3nM ΔG°: -48.6kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 14nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 18nM ΔG°: -44.2kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 18nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 27nM ΔG°: -43.2kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 40nM ΔG°: -42.2kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 48nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 64nM ΔG°: -41.1kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 64nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 71nM ΔG°: -40.8kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 95nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 95nM ΔG°: -40.1kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 103nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 106nM ΔG°: -39.8kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 125nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 131nM ΔG°: -39.3kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 142nM ΔG°: -39.1kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 161nM ΔG°: -38.8kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 220nM ΔG°: -38.0kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 225nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 345nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 763nM ΔG°: -34.9kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 839nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 1.08E+3nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 2.76E+3nM ΔG°: -31.7kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human thrombin was determined by using the chromogenic substrates S-2366. The hydrolysis rates of chromoge...More data for this Ligand-Target Pair
Affinity DataKi: 2.90E+3nMAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 3.00E+3nM ΔG°: -31.5kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s...More data for this Ligand-Target Pair
Affinity DataKi: 3.16E+3nMAssay Description:The inhibitory effect of test compound for human thrombin was determined by using the chromogenic substrates S-2366. The hydrolysis rates of chromoge...More data for this Ligand-Target Pair
Affinity DataKi: 8.22E+3nM ΔG°: -29.0kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human thrombin was determined by using the chromogenic substrates S-2366. The hydrolysis rates of chromoge...More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:The inhibitory effect of test compound for human thrombin was determined by using the chromogenic substrates S-2366. The hydrolysis rates of chromoge...More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nM ΔG°: >-28.5kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human thrombin was determined by using the chromogenic substrates S-2366. The hydrolysis rates of chromoge...More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nM ΔG°: >-28.5kJ/molepH: 7.8 T: 2°CAssay Description:The inhibitory effect of test compound for human thrombin was determined by using the chromogenic substrates S-2366. The hydrolysis rates of chromoge...More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:The inhibitory effect of test compound for human thrombin was determined by using the chromogenic substrates S-2366. The hydrolysis rates of chromoge...More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:The inhibitory effect of test compound for human thrombin was determined by using the chromogenic substrates S-2366. The hydrolysis rates of chromoge...More data for this Ligand-Target Pair