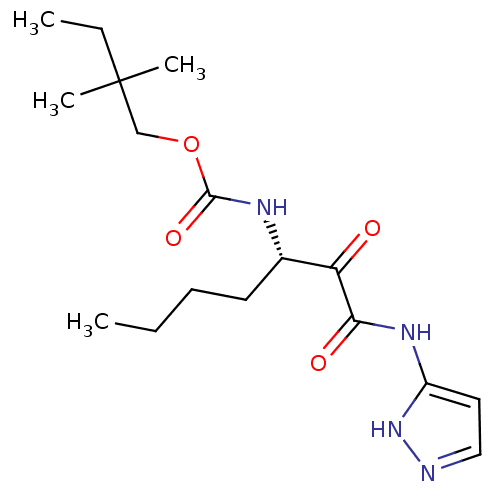
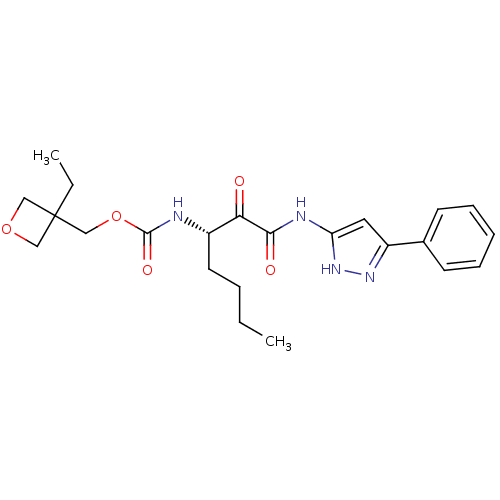
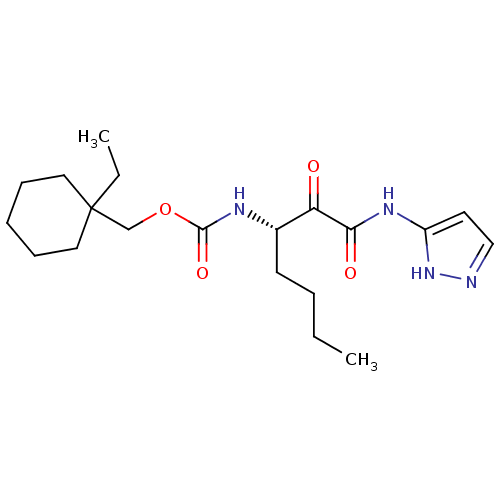
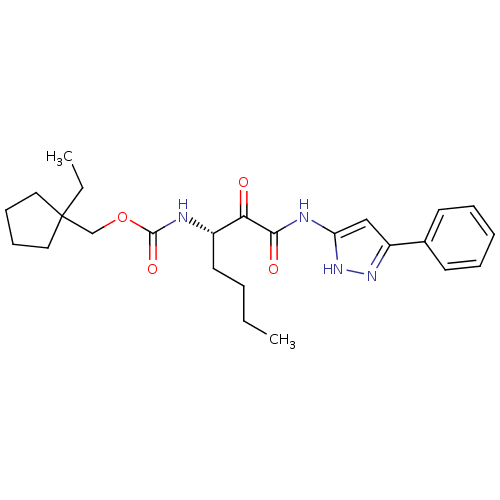
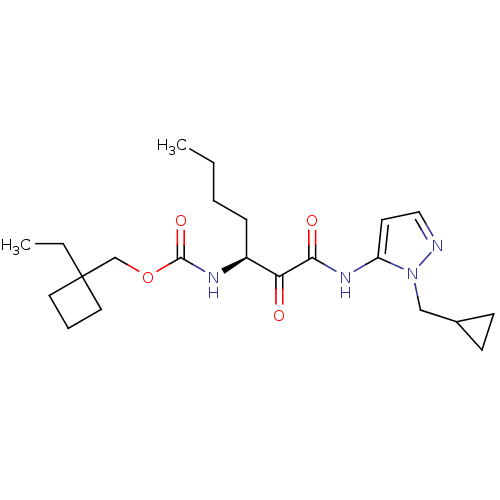
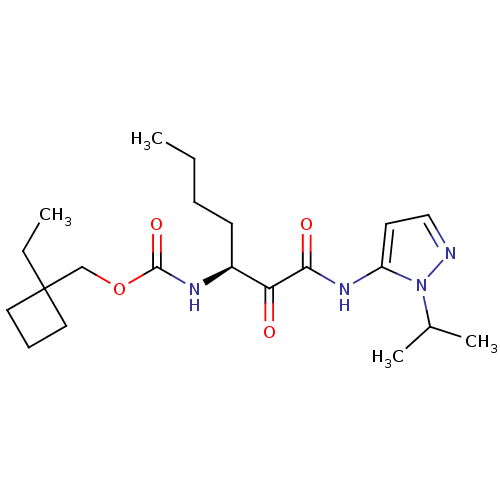
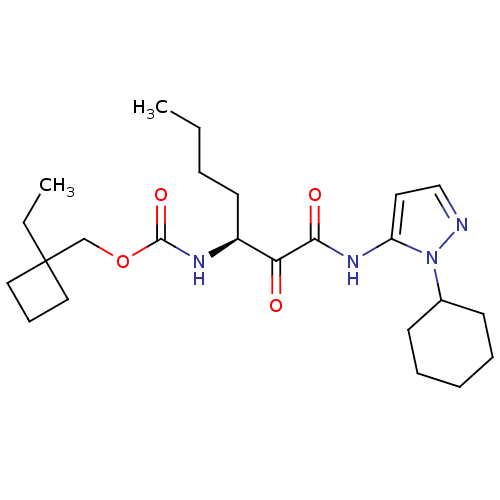
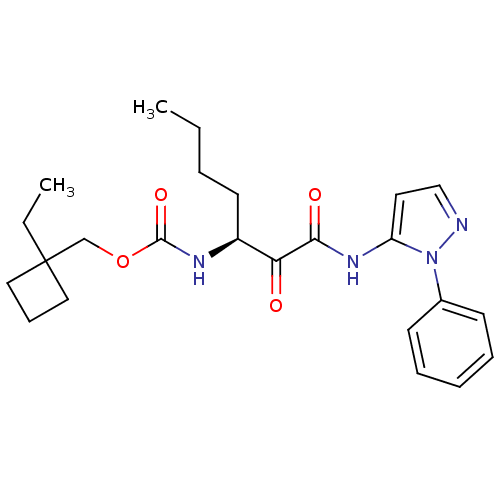
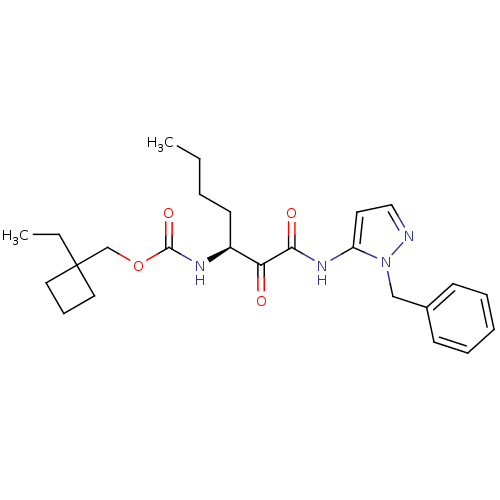
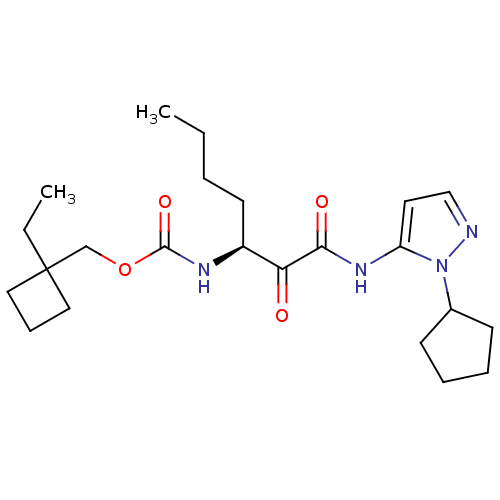
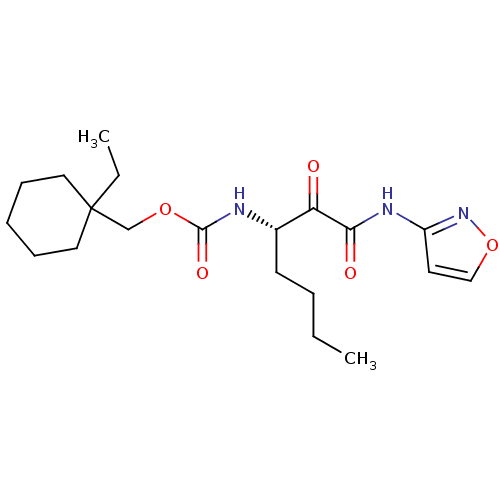
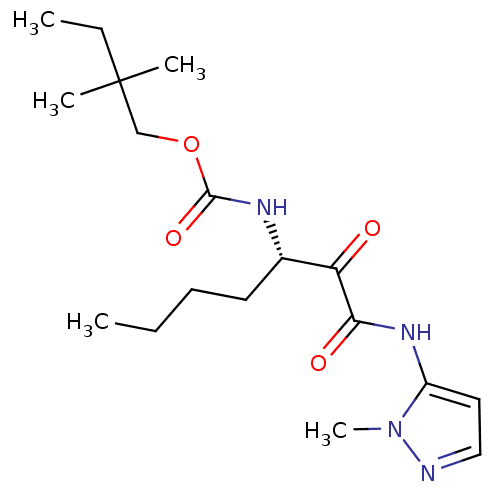
Report error Found 33 Enz. Inhib. hit(s) with all data for entry = 50015299
Affinity DataIC50: 1.60nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 3.40nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 6.20nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 6.20nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 36nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 45nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 51nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 52nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 54nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 58nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 63nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 65nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 68nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 68nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 71nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 71nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 76nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 83nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 120nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 135nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 140nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 350nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+3nMAssay Description:Inhibitory potency against human Cathepsin KMore data for this Ligand-Target Pair