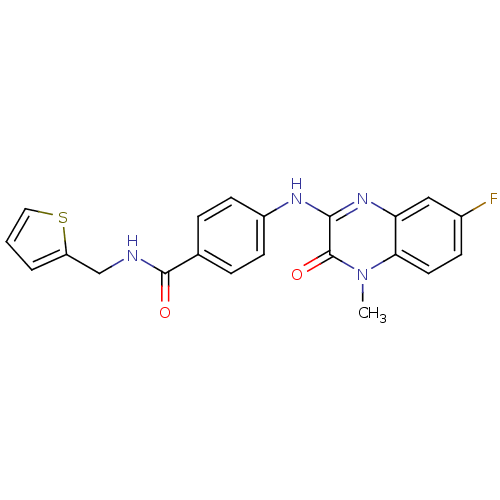
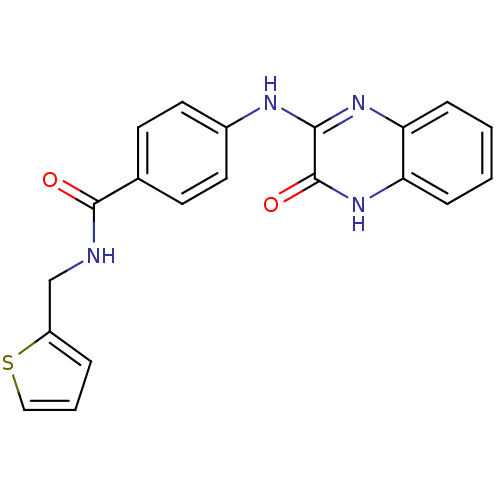
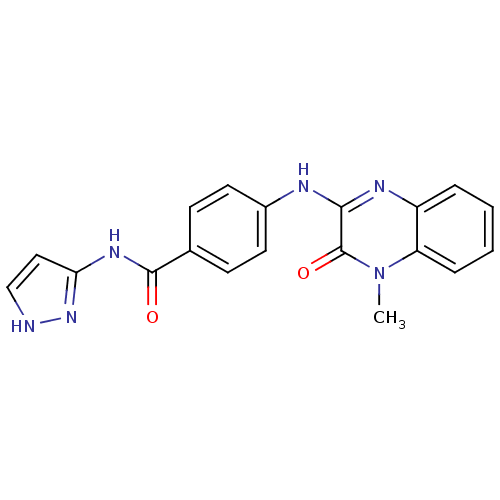
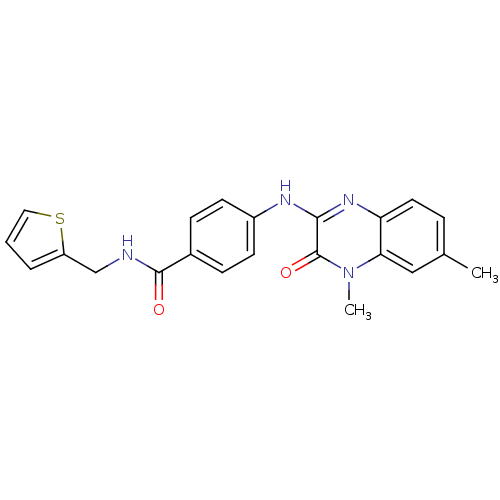
Report error Found 27 Enz. Inhib. hit(s) with all data for entry = 50037595
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
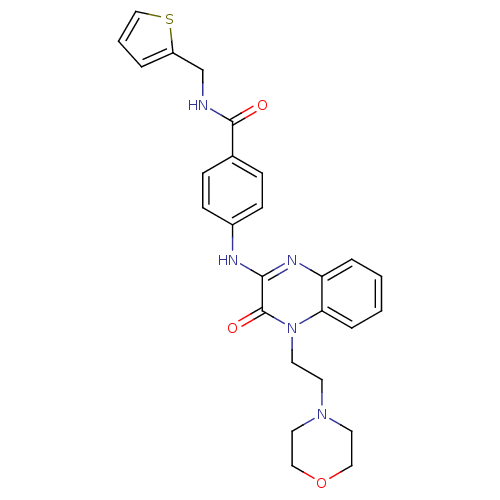
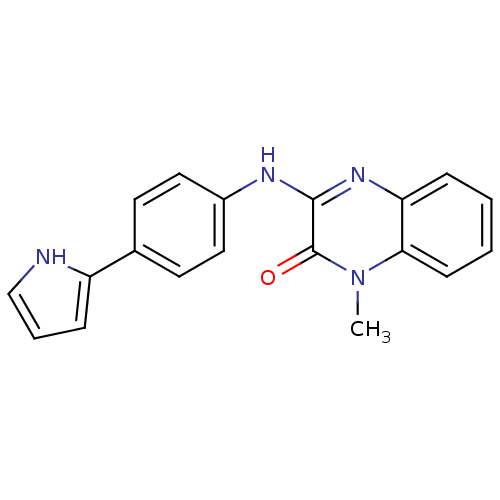
Affinity DataIC50: 110nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
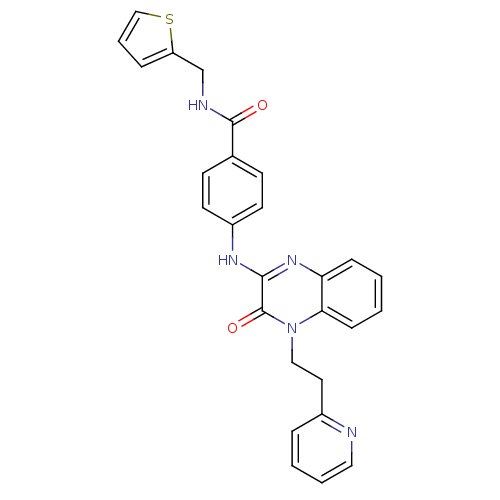
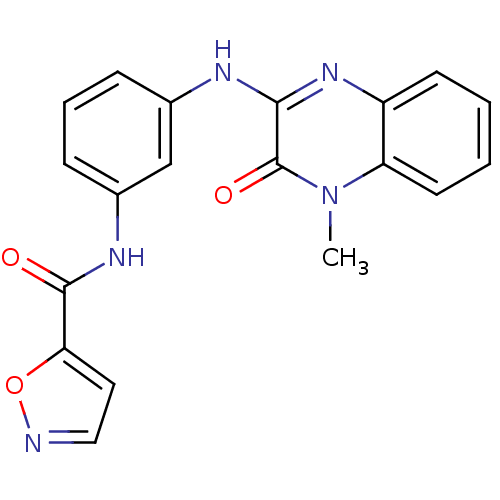
Affinity DataIC50: 110nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
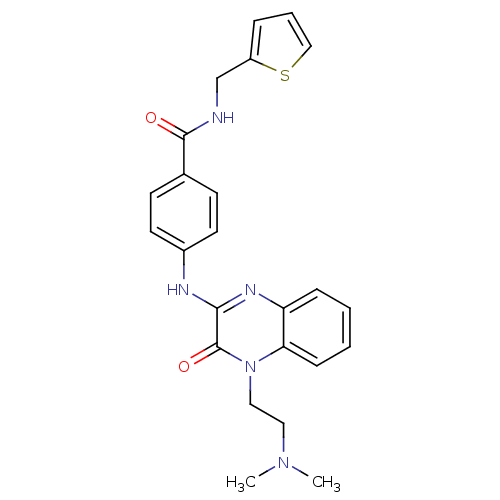
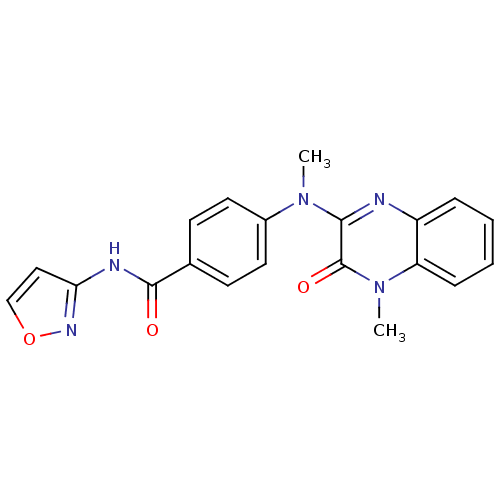
Affinity DataIC50: 120nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
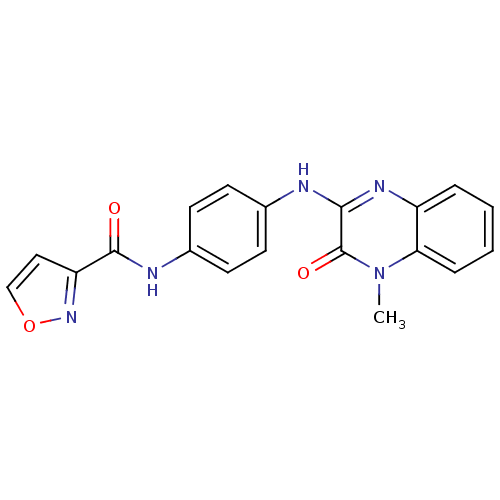
Affinity DataIC50: 140nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 160nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 190nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 190nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 200nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 280nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 320nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 350nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 420nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 480nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 580nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 710nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 710nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 730nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 2.50E+3nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 2.50E+3nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 2.50E+3nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 5.70E+3nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair
TargetGlycogen phosphorylase, muscle form(Human)
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibitory concentration against glycogen phosphorylase of rabbit muscleMore data for this Ligand-Target Pair