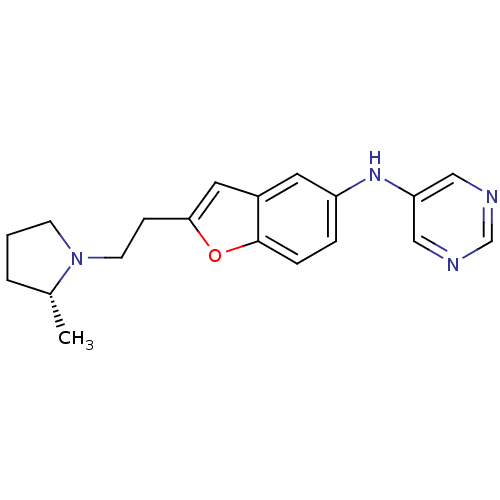
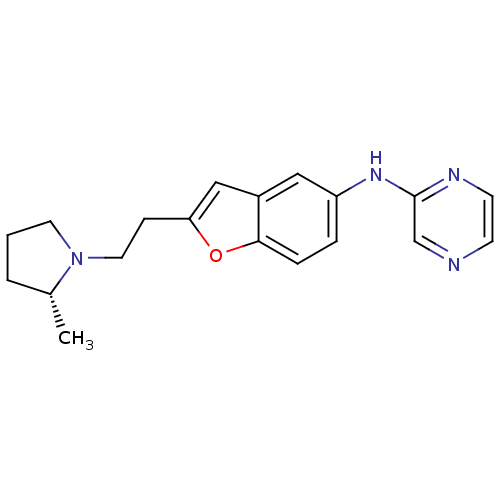
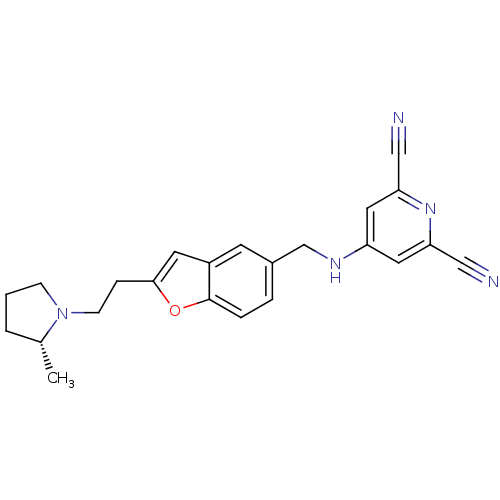
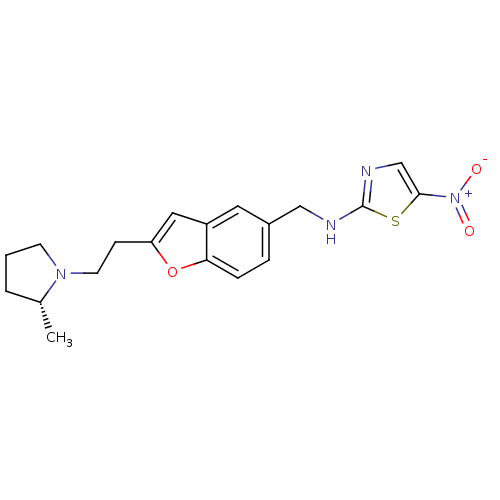
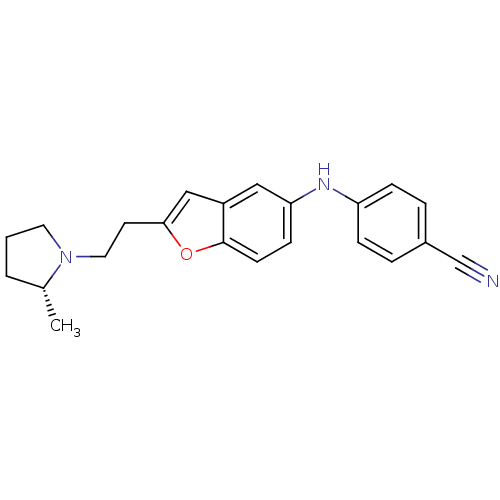
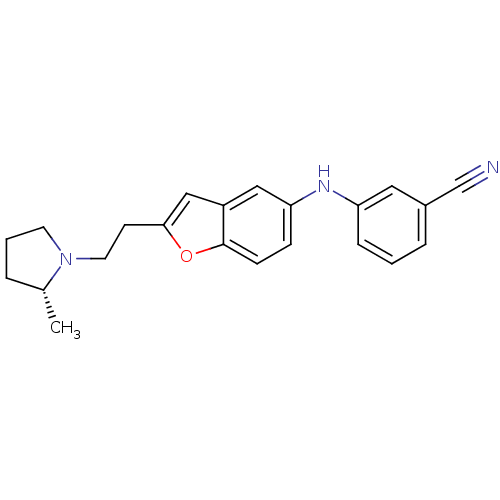
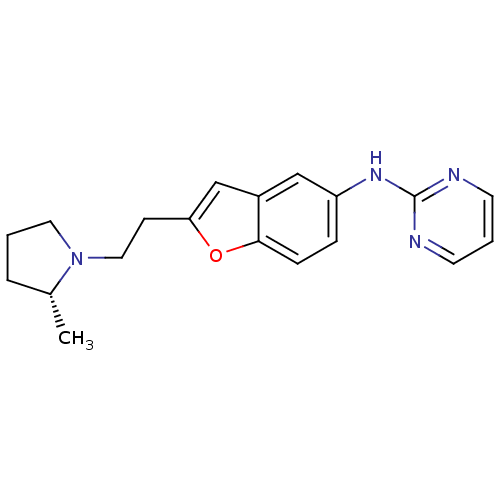
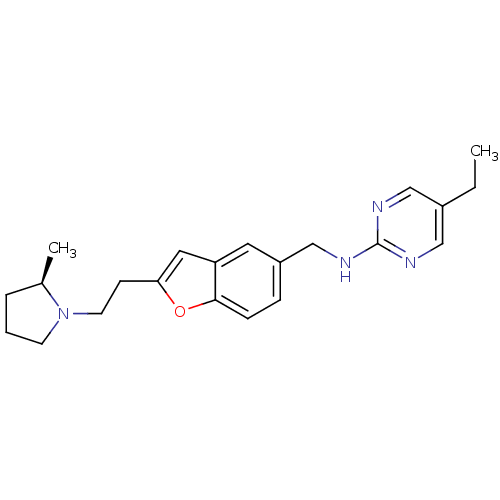
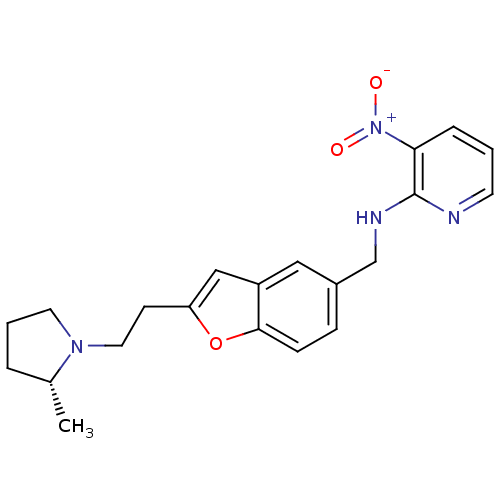
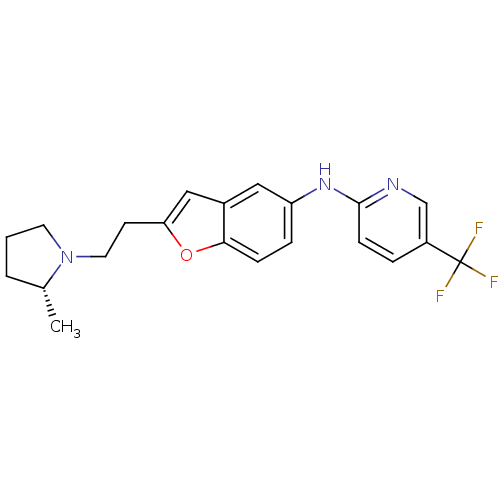
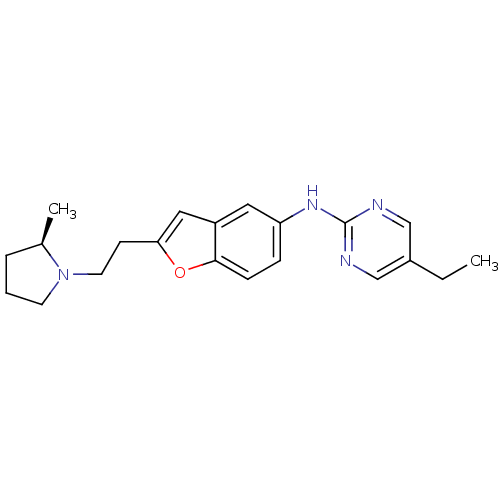
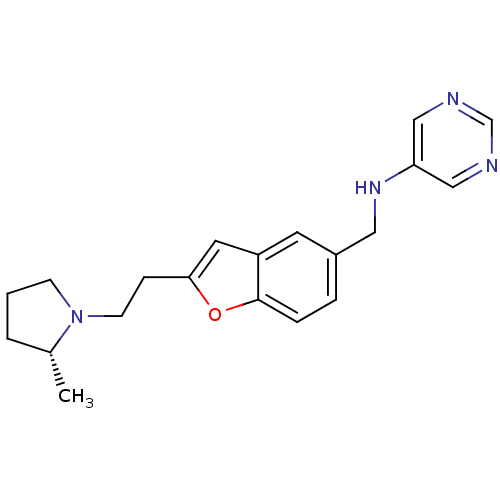
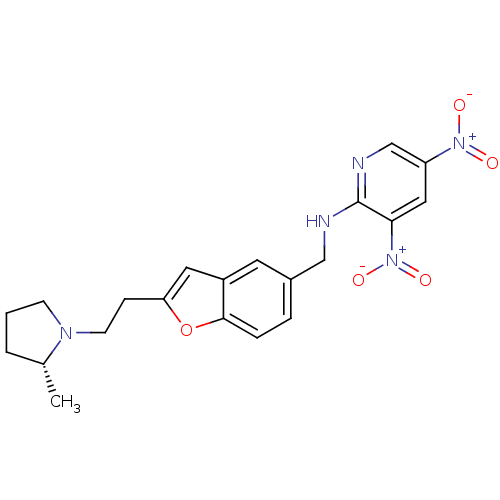
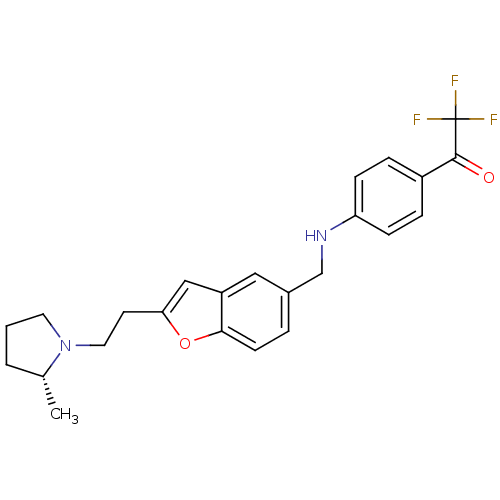
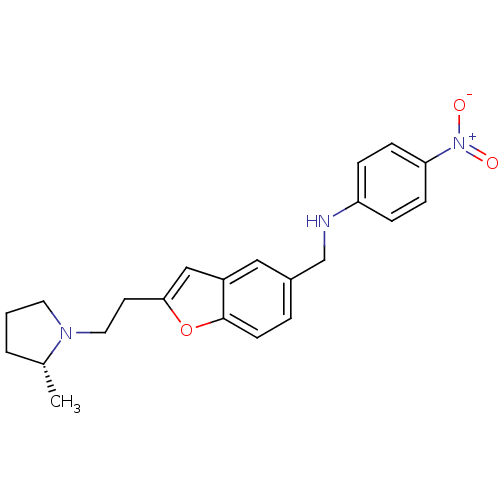
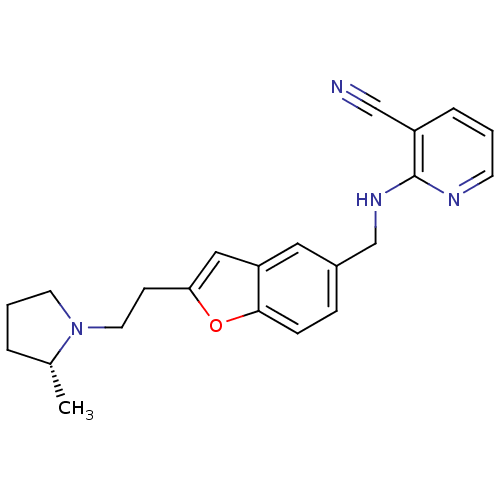
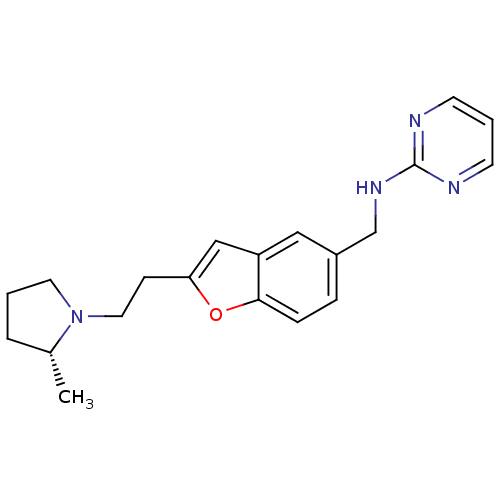
Report error Found 60 Enz. Inhib. hit(s) with all data for entry = 50016874
Affinity DataKi: 0.0500nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.0800nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.110nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.110nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.130nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.140nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.190nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.220nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.270nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.270nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.290nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.290nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.300nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.310nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.310nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.310nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.330nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.340nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.350nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.380nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.400nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.400nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.430nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.450nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.540nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.580nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.620nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.680nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.690nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.710nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.740nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.740nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.740nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.840nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.870nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 0.870nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 1.04nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 1.18nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 1.19nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 1.22nMAssay Description:In vitro binding affinity for human histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 1.23nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 1.35nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 1.42nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 1.47nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 1.58nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 1.92nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 2.01nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 2.12nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair
Affinity DataKi: 2.36nMAssay Description:In vitro binding affinity for rat histamine H3 receptor using [3H]N-alpha-methylhistamineMore data for this Ligand-Target Pair