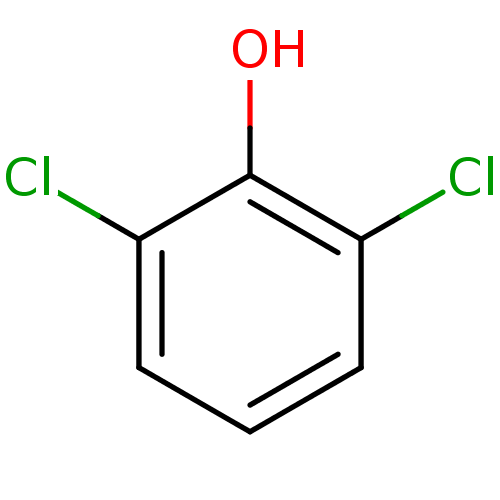
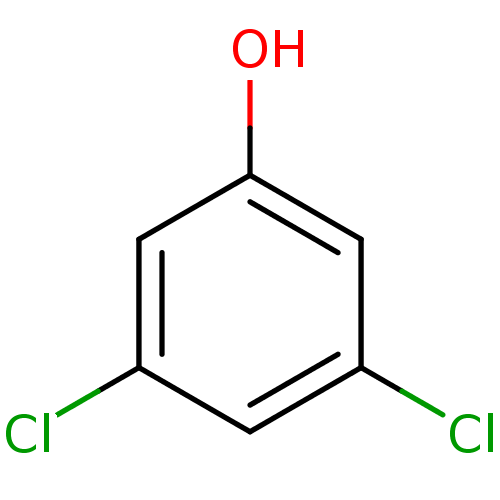
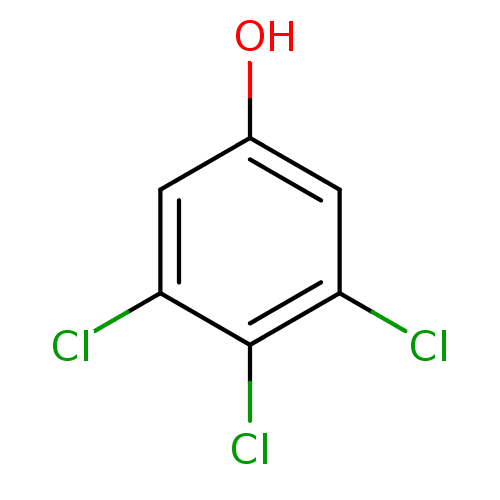
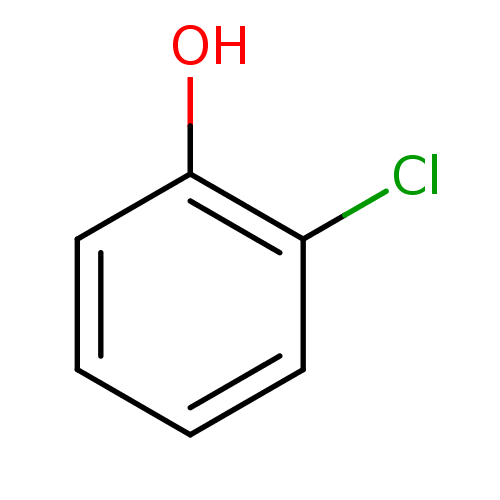
Report error Found 8 Enz. Inhib. hit(s) with all data for entry = 5706
Affinity DataKd: <50nMAssay Description:Formation of the hydroxylated product and depletion of NADPH were measured following ethyl acetate extraction of the reaction mixture.More data for this Ligand-Target Pair
Affinity DataKd: 4.20E+3nMAssay Description:Formation of the hydroxylated product and depletion of NADPH were measured following ethyl acetate extraction of the reaction mixture.More data for this Ligand-Target Pair
Affinity DataKd: 7.00E+3nMAssay Description:Formation of the hydroxylated product and depletion of NADPH were measured following ethyl acetate extraction of the reaction mixture.More data for this Ligand-Target Pair
Affinity DataKd: 1.10E+4nMAssay Description:Formation of the hydroxylated product and depletion of NADPH were measured following ethyl acetate extraction of the reaction mixture.More data for this Ligand-Target Pair
Affinity DataKd: 4.55E+4nMAssay Description:Formation of the hydroxylated product and depletion of NADPH were measured following ethyl acetate extraction of the reaction mixture.More data for this Ligand-Target Pair
Affinity DataKd: >5.00E+5nMAssay Description:Formation of the hydroxylated product and depletion of NADPH were measured following ethyl acetate extraction of the reaction mixture.More data for this Ligand-Target Pair
Affinity DataKd: >5.00E+5nMAssay Description:Formation of the hydroxylated product and depletion of NADPH were measured following ethyl acetate extraction of the reaction mixture.More data for this Ligand-Target Pair
Affinity DataKd: >1.00E+6nMAssay Description:Formation of the hydroxylated product and depletion of NADPH were measured following ethyl acetate extraction of the reaction mixture.More data for this Ligand-Target Pair