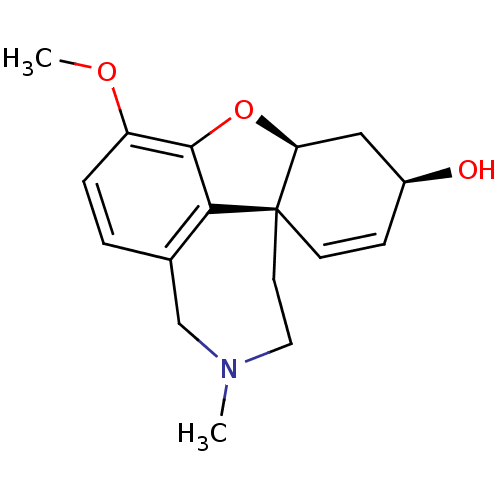
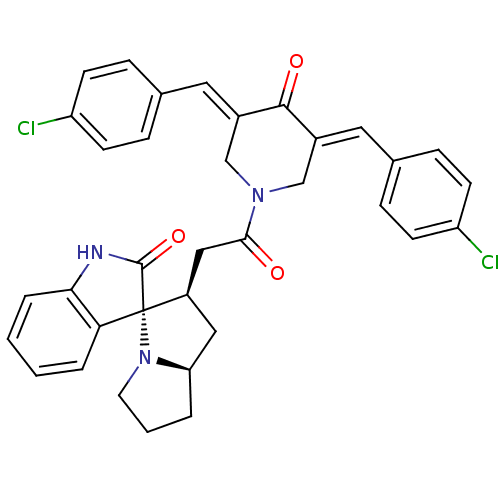
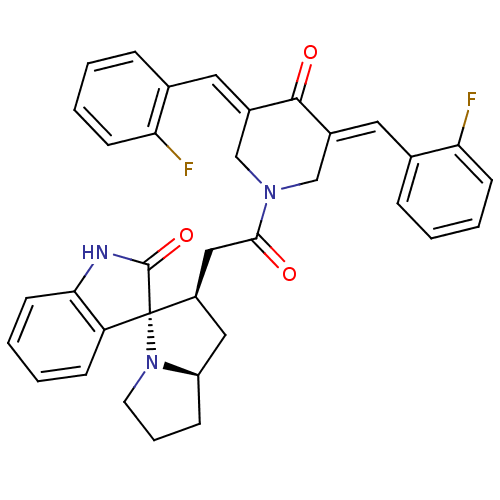
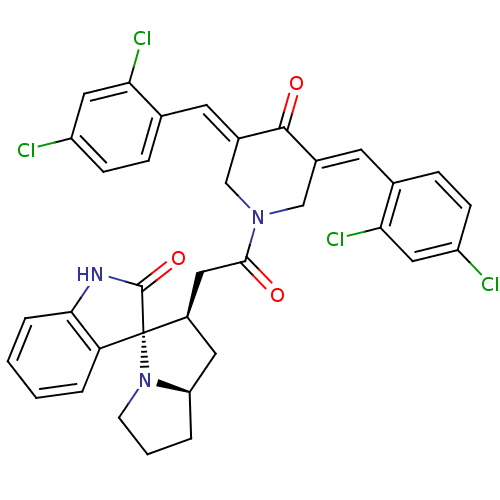
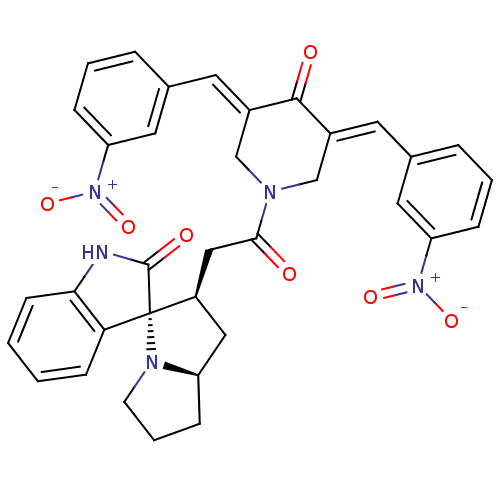
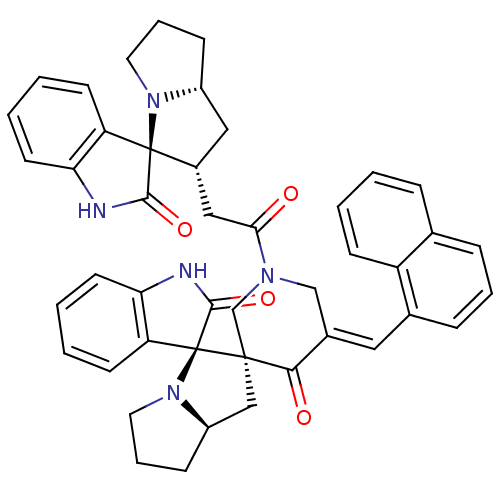
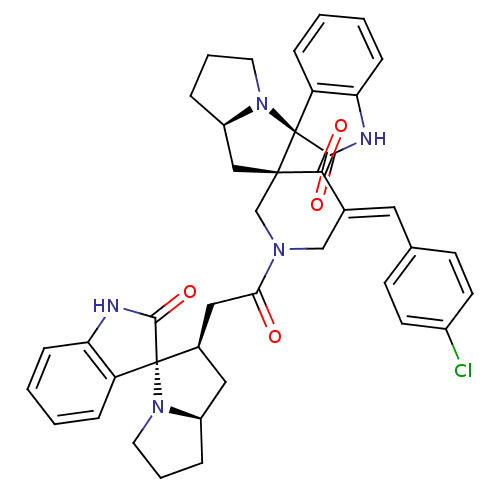
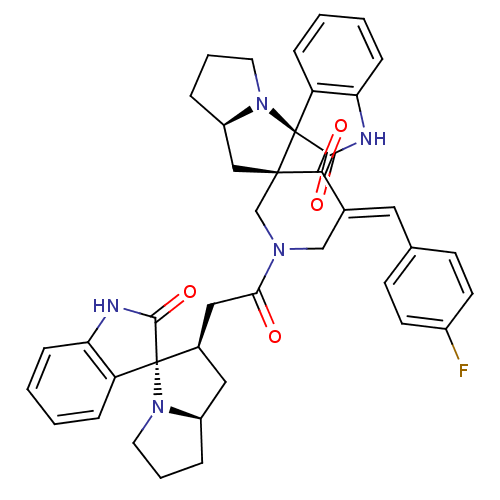
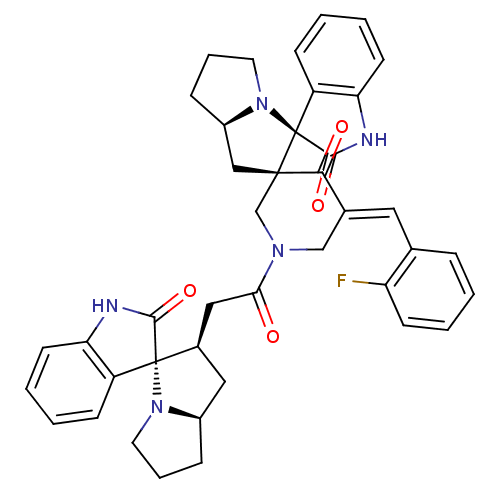
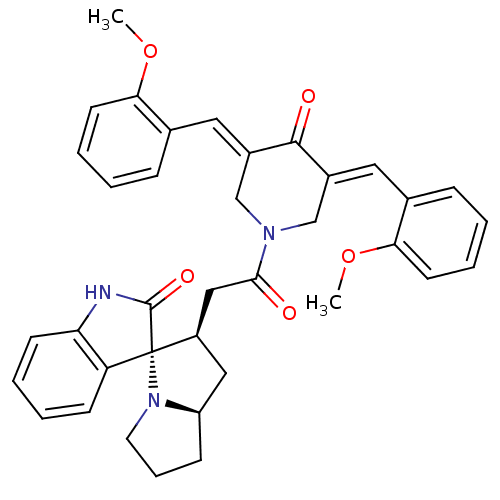
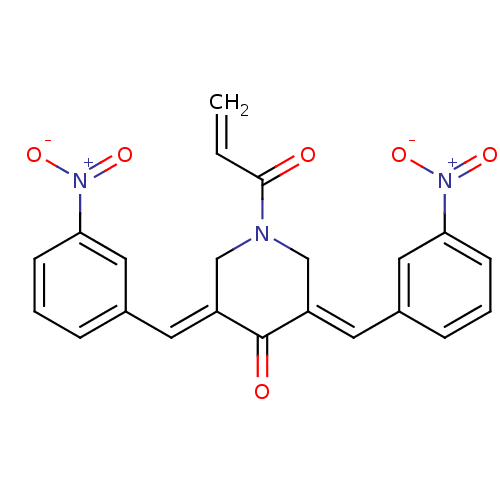
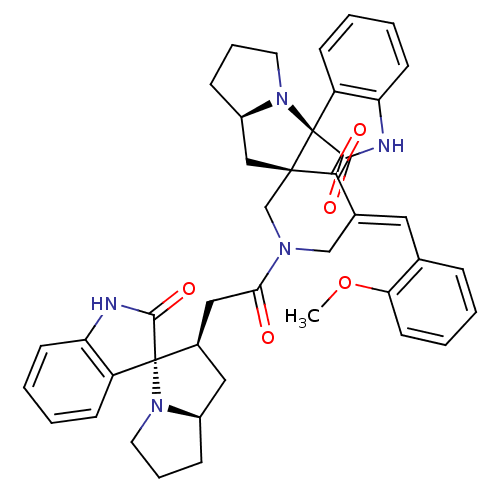
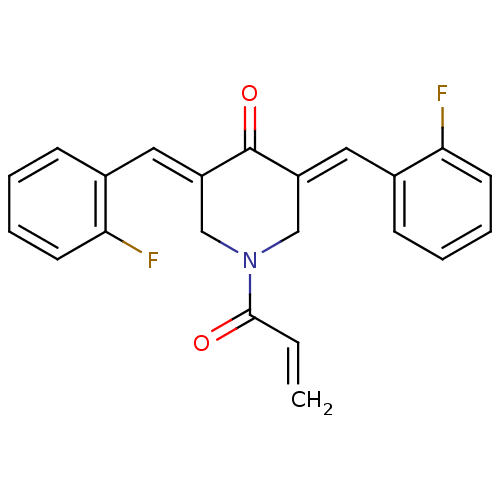
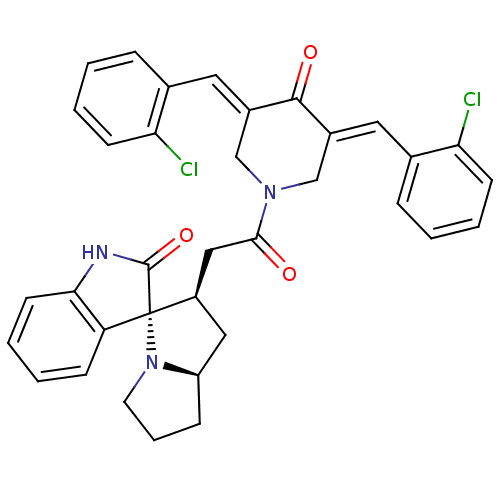
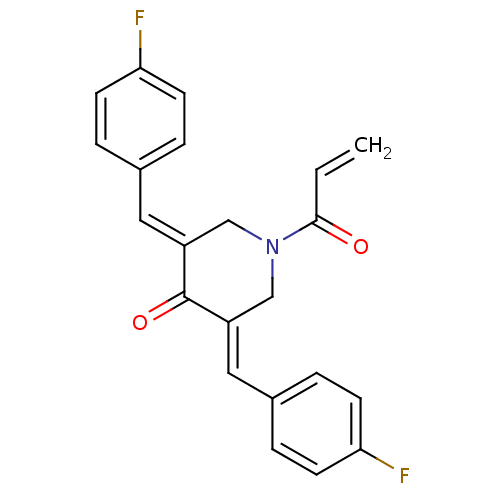
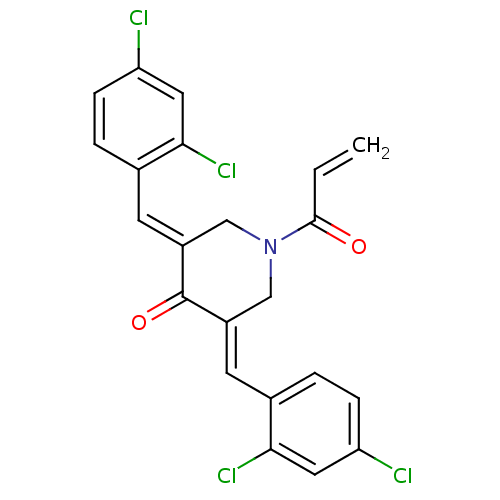
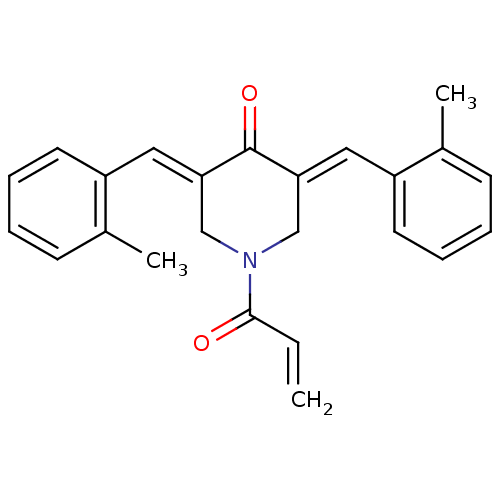
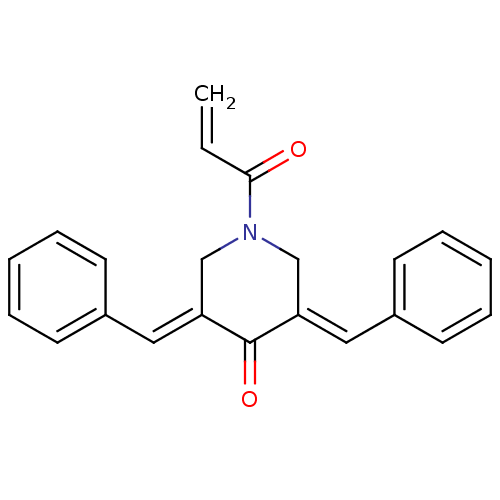
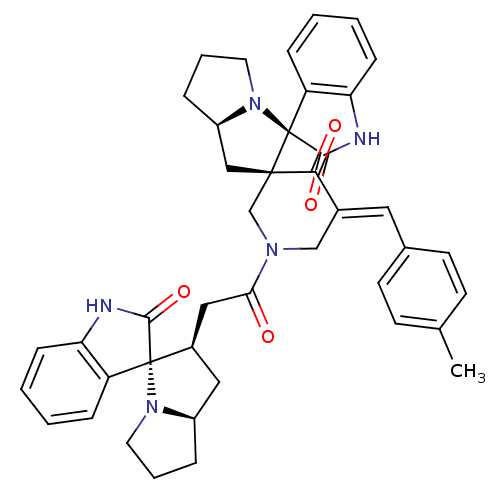
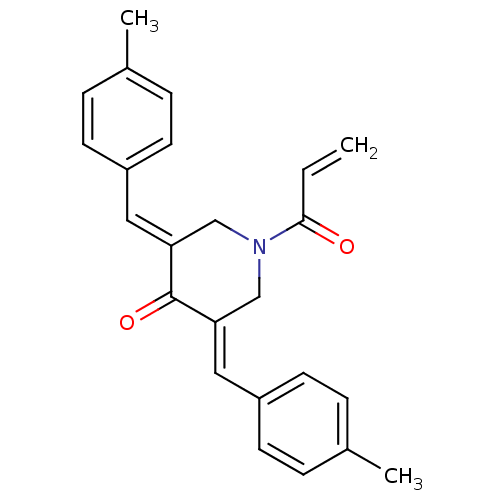
Report error Found 68 Enz. Inhib. hit(s) with all data for entry = 50042765
Affinity DataIC50: 2.09E+3nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 3.36E+3nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 3.50E+3nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 3.99E+3nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 4.11E+3nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 5.99E+3nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 6.42E+3nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 6.76E+3nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 6.96E+3nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 7.92E+3nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 8.29E+3nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 8.72E+3nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 9.09E+3nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 9.41E+3nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 9.45E+3nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 9.76E+3nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.09E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.14E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.17E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.18E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.18E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.19E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.25E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.36E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.38E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.51E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.58E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.59E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.63E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.63E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.74E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.75E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.77E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.78E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.79E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.81E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.81E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.82E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.92E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.93E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.96E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.97E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.97E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.98E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.01E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.06E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.12E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.20E+4nMAssay Description:Inhibition of AChE (unknown origin) using acetylthiocholine iodide substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.21E+4nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine chloride substrate by Ellman's method based spectrophotometryMore data for this Ligand-Target Pair