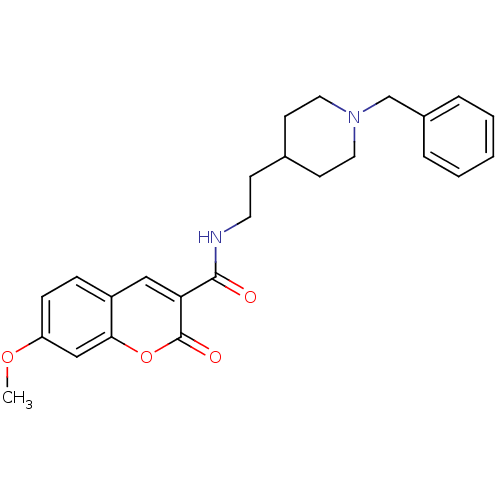
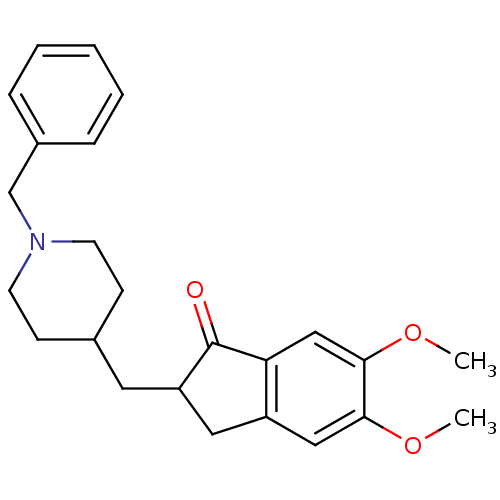
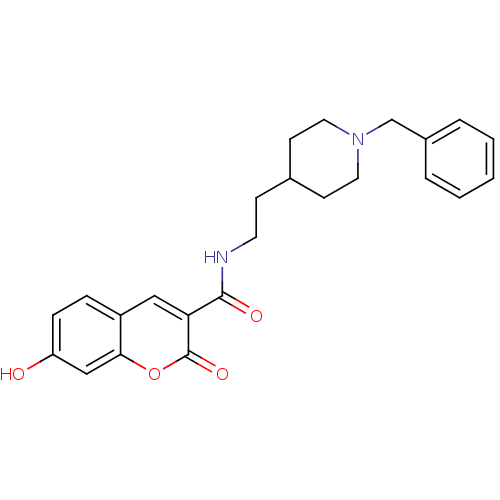
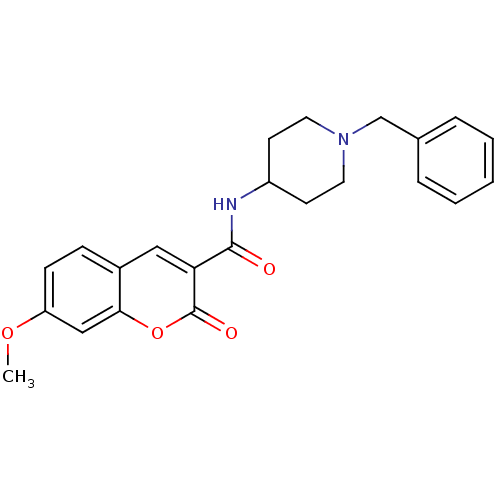
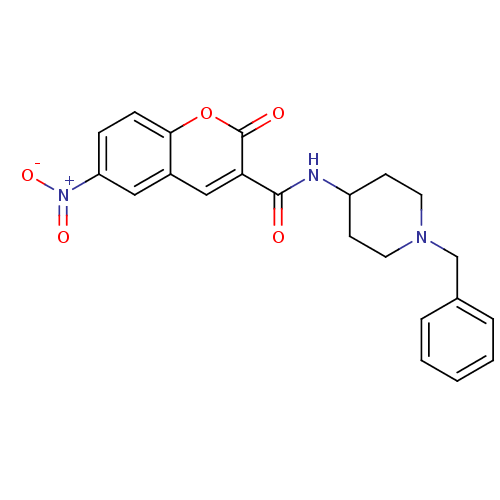
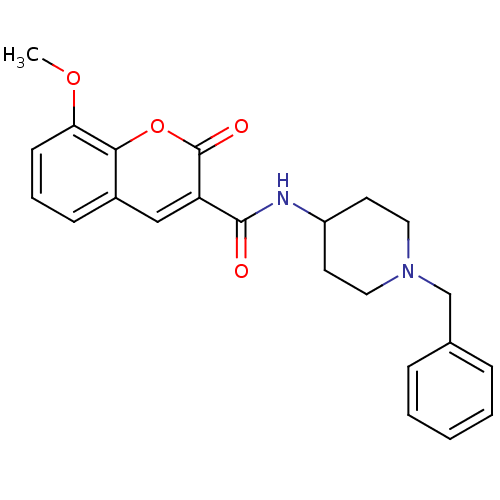
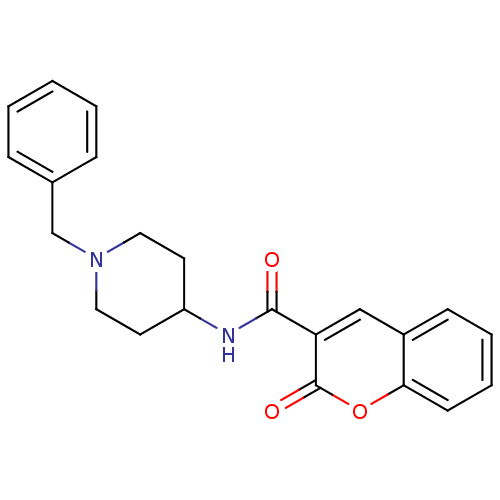
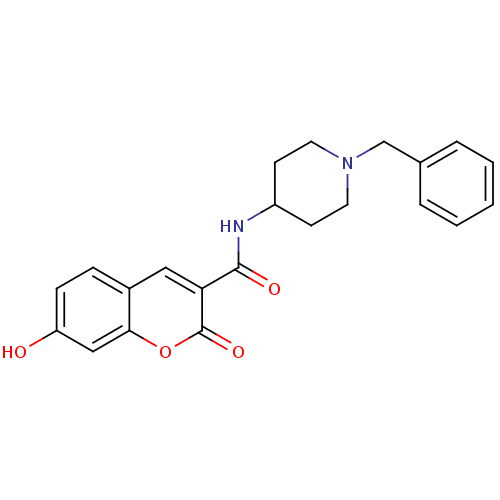
Report error Found 28 Enz. Inhib. hit(s) with all data for entry = 50043603
Affinity DataIC50: 0.300nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 26nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 30nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 70nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 143nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 183nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 357nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 371nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 420nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 430nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 633nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+3nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.70E+3nMAssay Description:Inhibition of electric eel AChE using acetylcholine as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.38E+3nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 7.90E+3nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.56E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.70E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.80E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.20E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.70E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 4.20E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 4.60E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 5.50E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair