Report error Found 65 Enz. Inhib. hit(s) with all data for entry = 1664
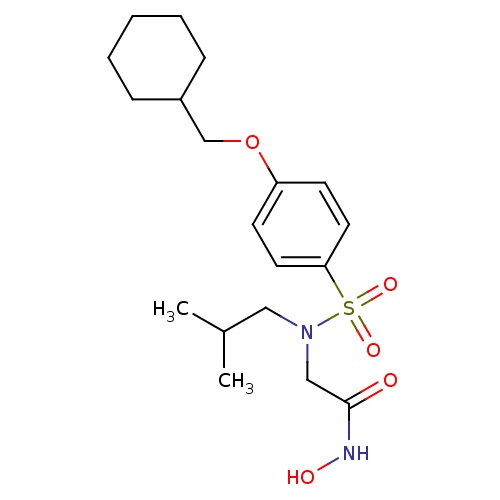
Affinity DataKi: 8nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
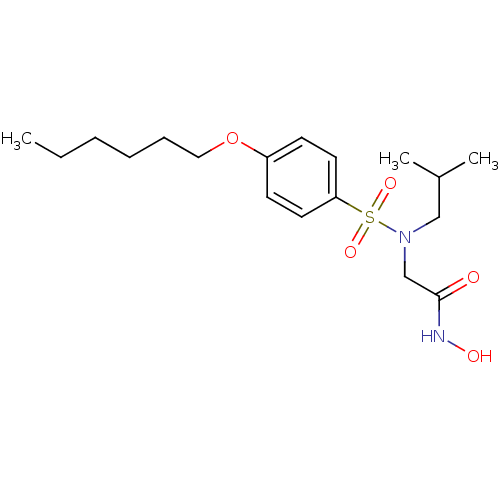
Affinity DataKi: 20nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
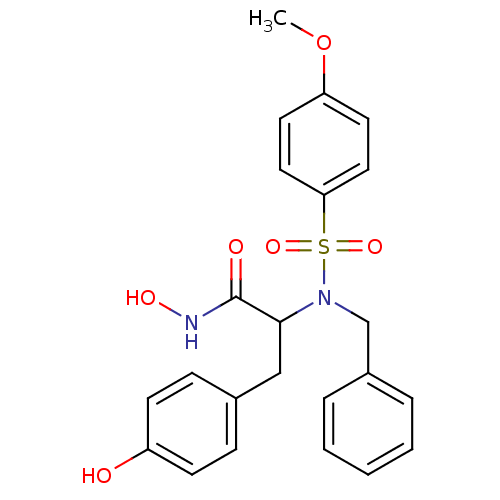
Affinity DataKi: 23nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
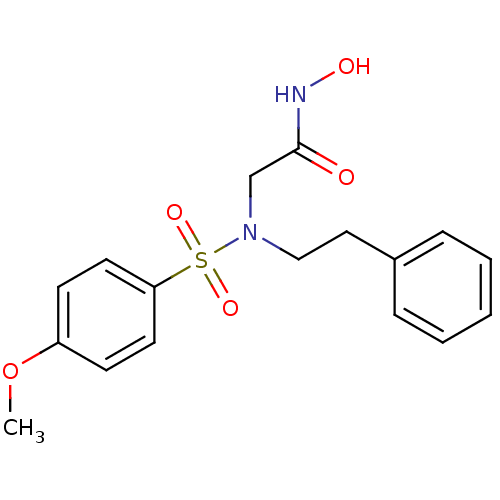
Affinity DataKi: 28nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 29nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 31nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 33nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 34nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 36nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 38nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 39nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 42nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 43nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 44nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 46nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 48nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 50nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 58nM ΔG°: -43.0kJ/molepH: 7.5 T: 2°CAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 61nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 61nM ΔG°: -42.8kJ/molepH: 7.5 T: 2°CAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 63nM ΔG°: -42.8kJ/molepH: 7.5 T: 2°CAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 65nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 66nM ΔG°: -42.6kJ/molepH: 7.5 T: 2°CAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 69nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 70nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 70nM ΔG°: -42.5kJ/molepH: 7.5 T: 2°CAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 70nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 71nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 78nM ΔG°: -42.2kJ/molepH: 7.5 T: 2°CAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 81nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 85nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 86nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 90nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 92nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 94nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 94nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 100nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 104nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 104nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 107nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 113nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 125nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 133nM ΔG°: -40.8kJ/molepH: 7.5 T: 2°CAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 134nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 141nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 151nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 192nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 236nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 305nMAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
Affinity DataKi: 336nM ΔG°: -38.4kJ/molepH: 7.5 T: 2°CAssay Description:Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ...More data for this Ligand-Target Pair
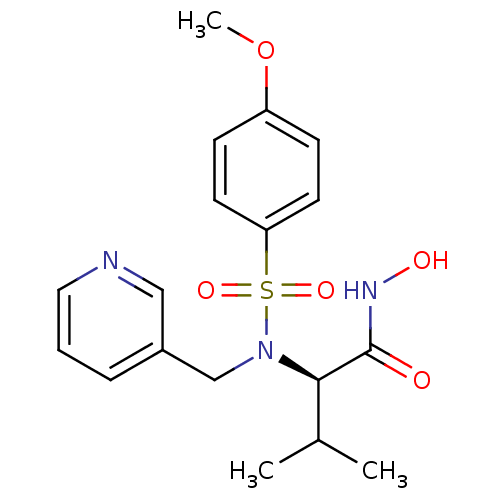



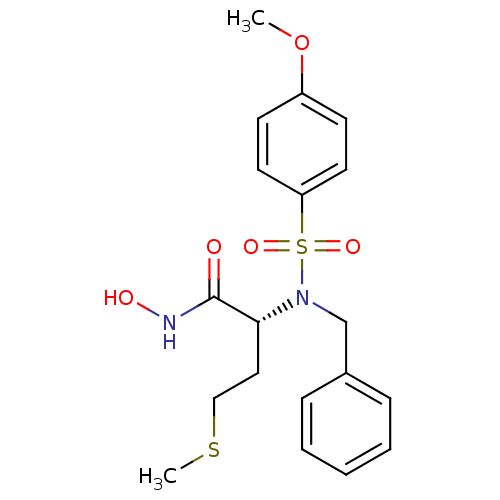

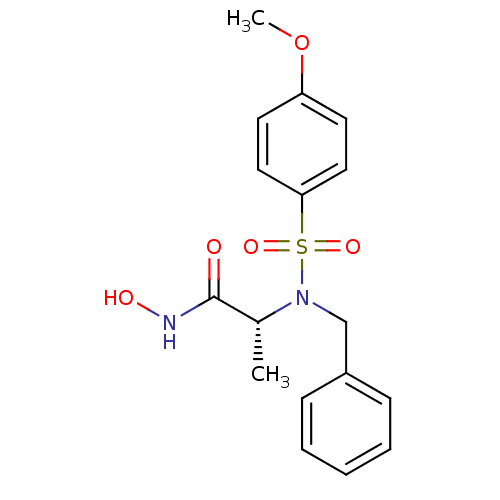
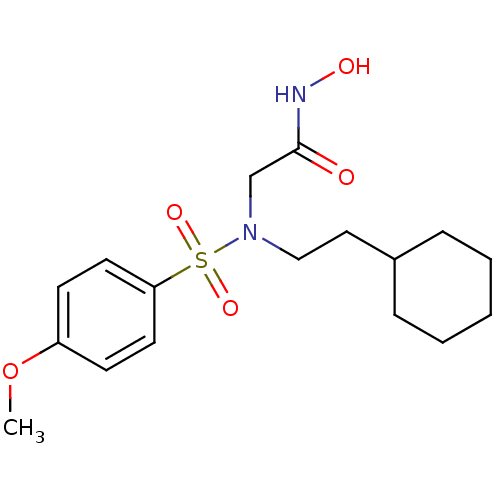
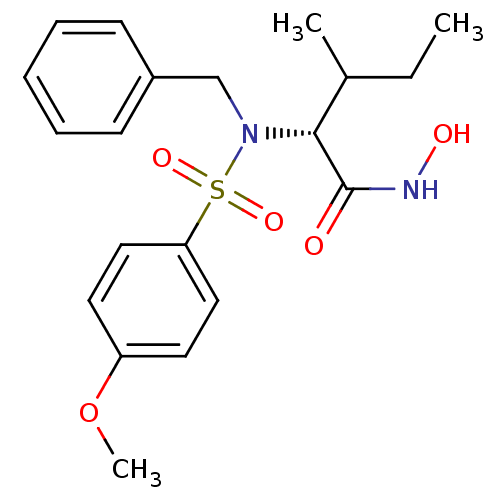
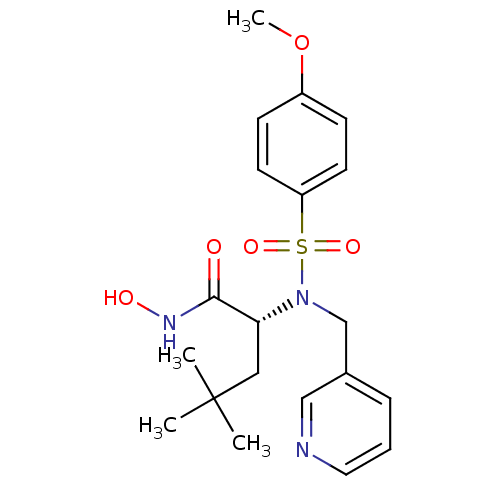

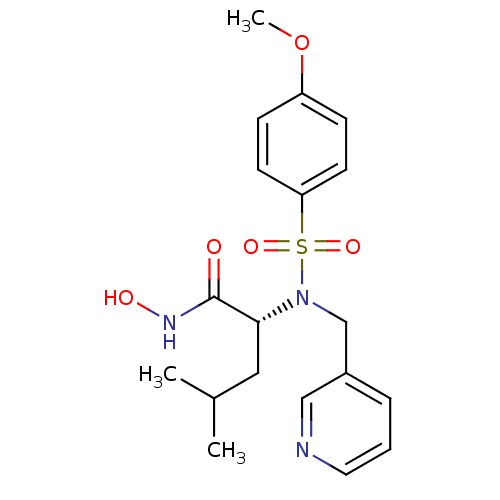

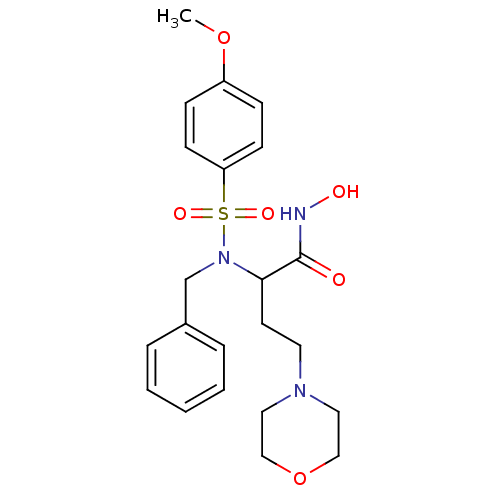


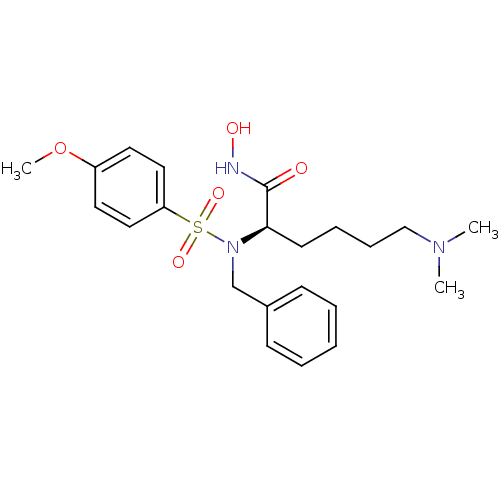
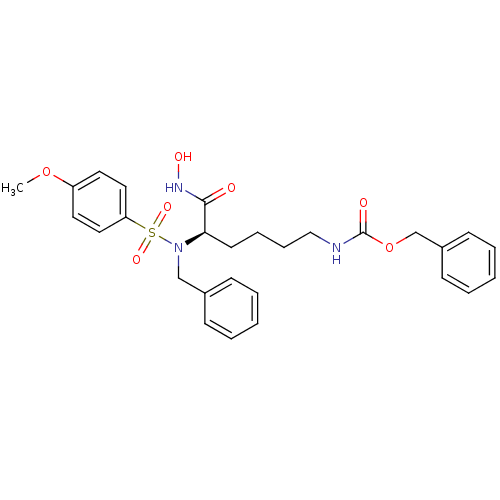


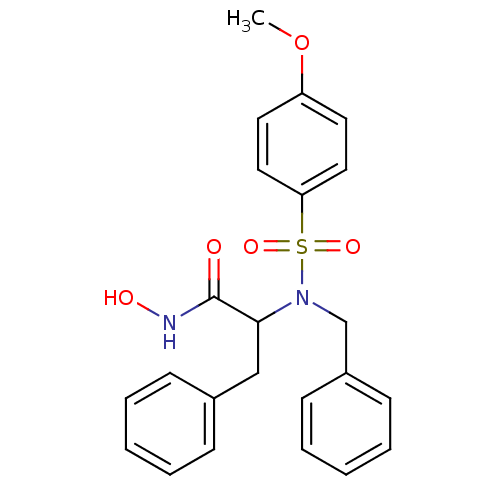

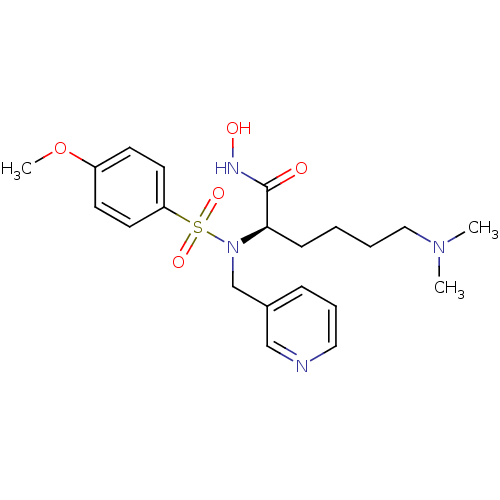
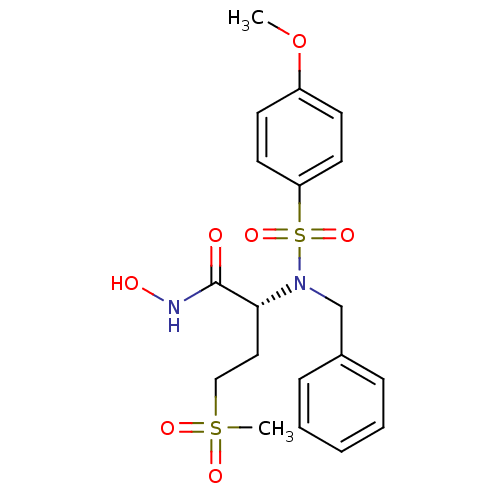

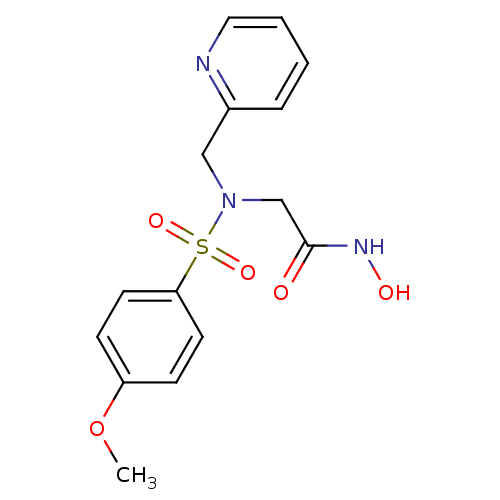

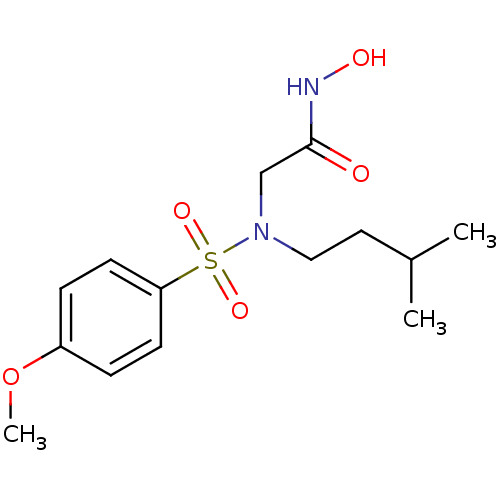
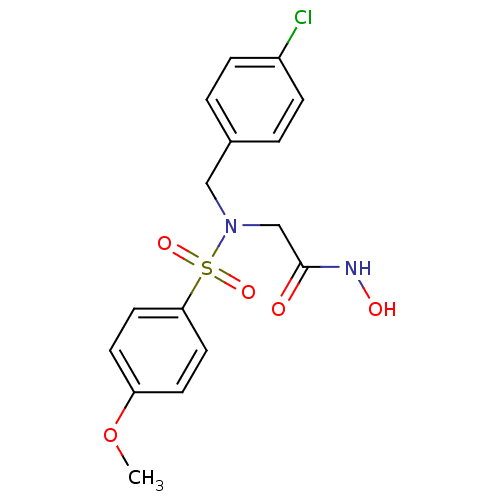
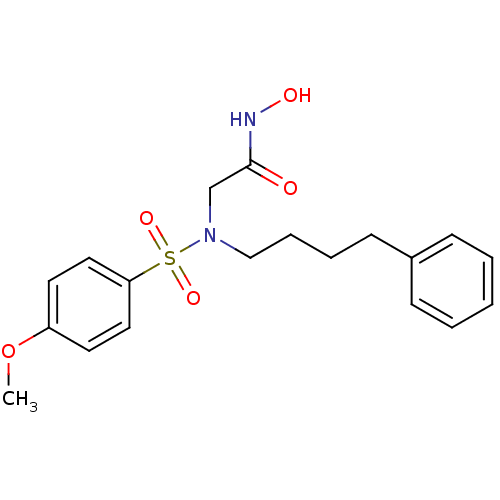


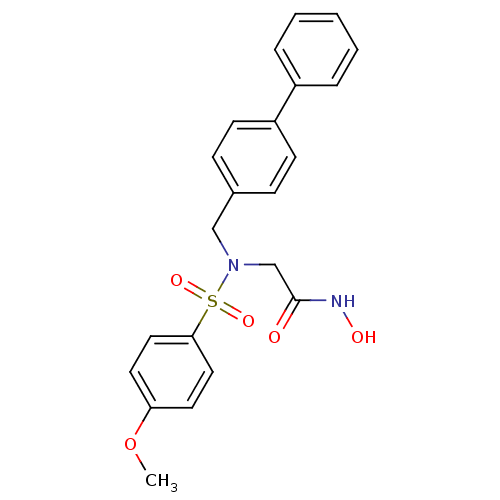
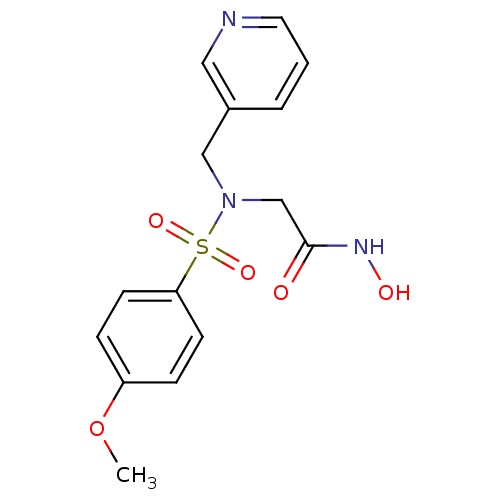
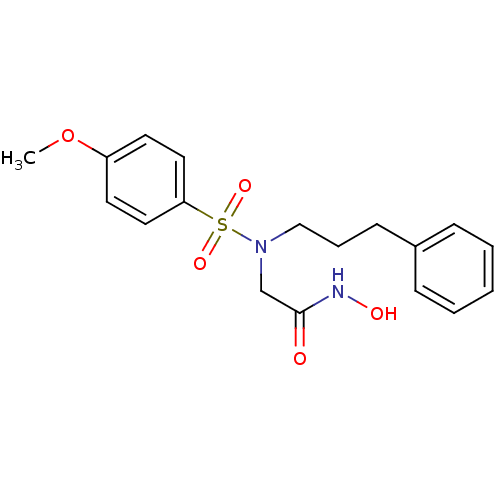
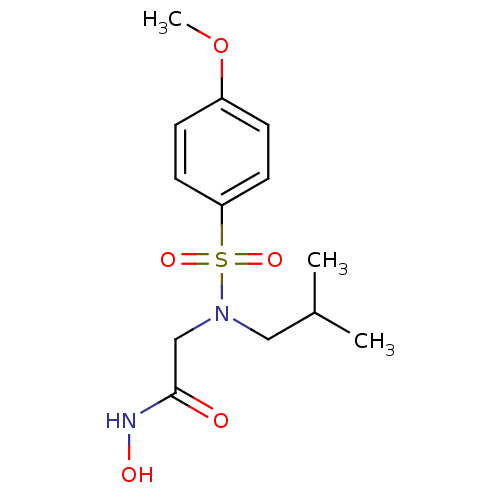
 3D Structure (crystal)
3D Structure (crystal)