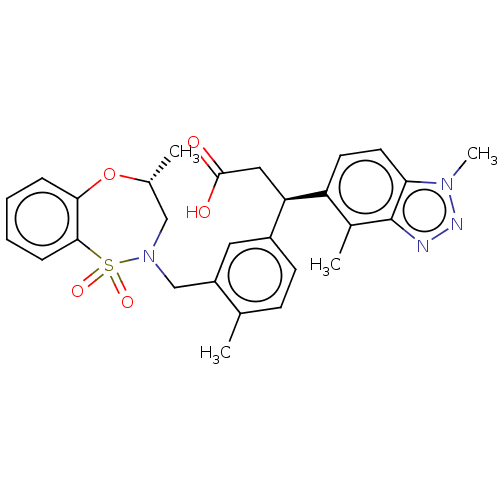
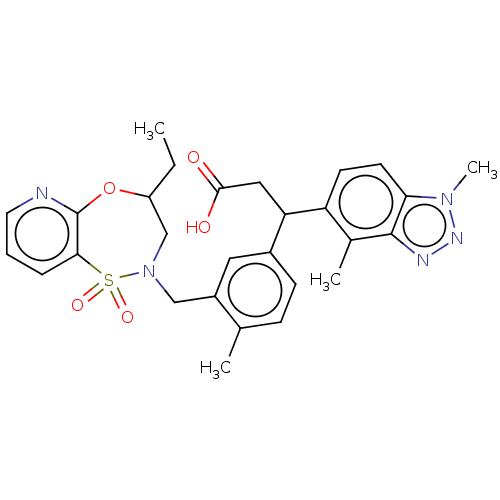
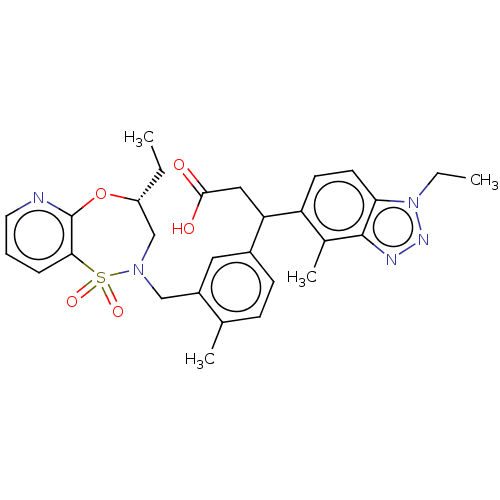
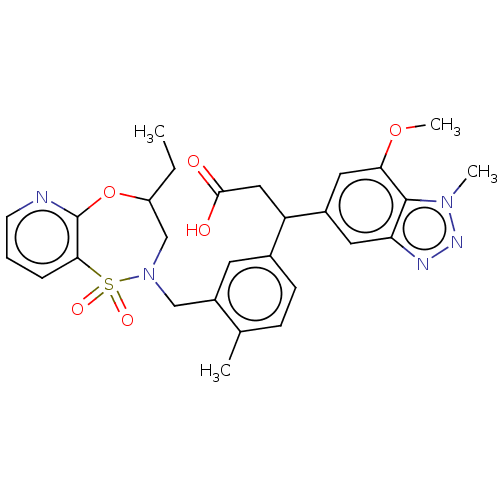
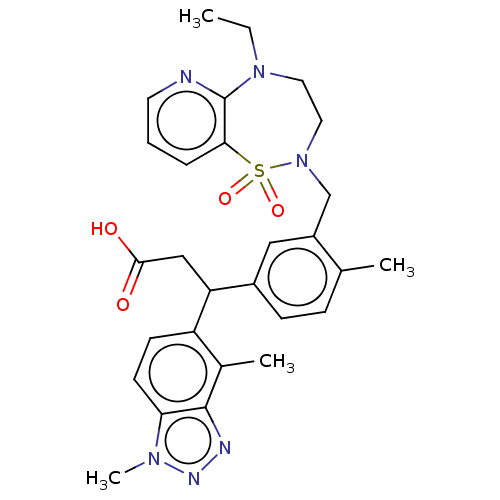
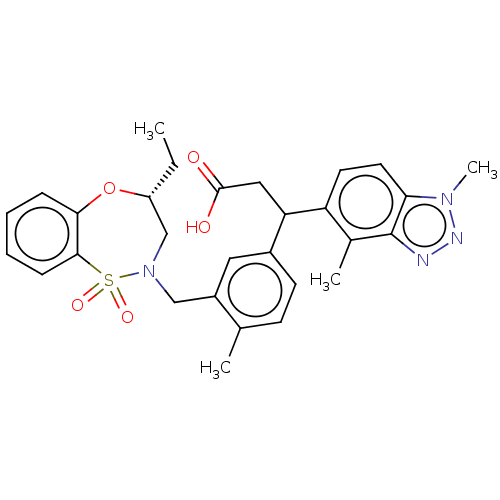
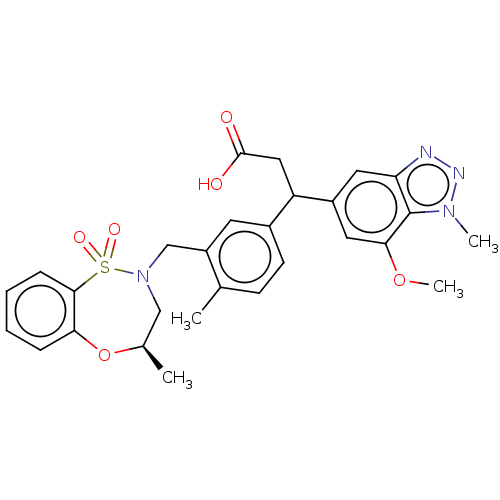
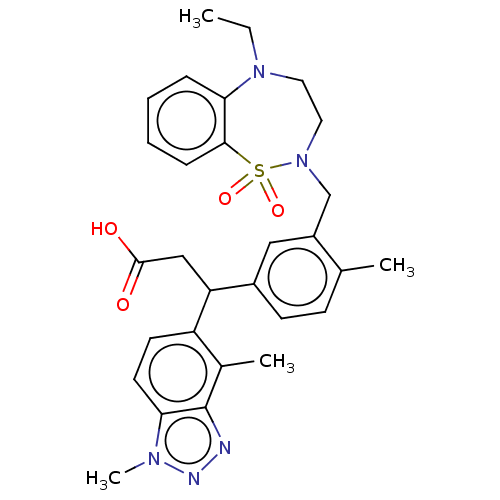
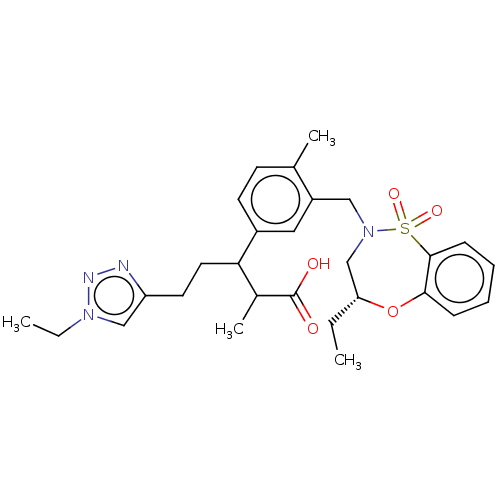
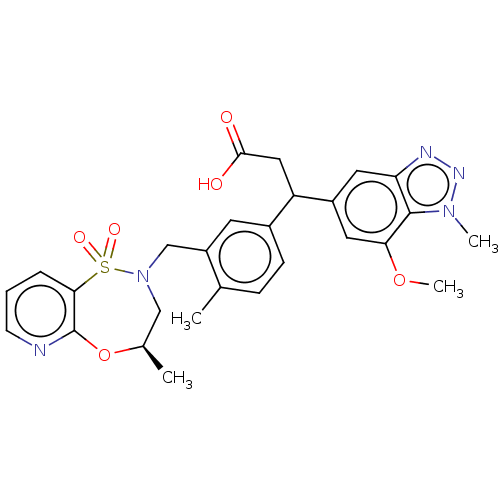
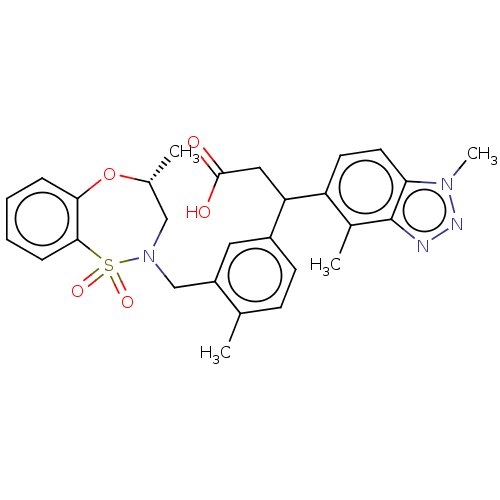
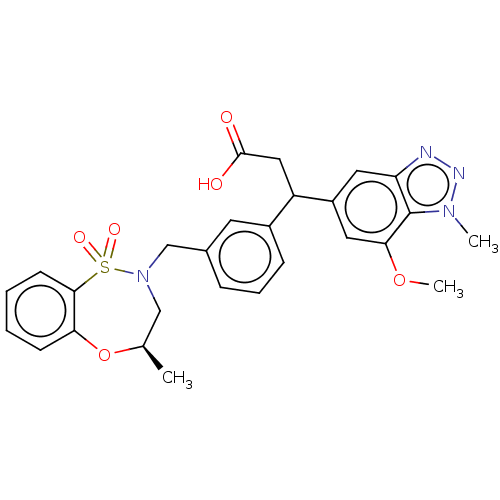
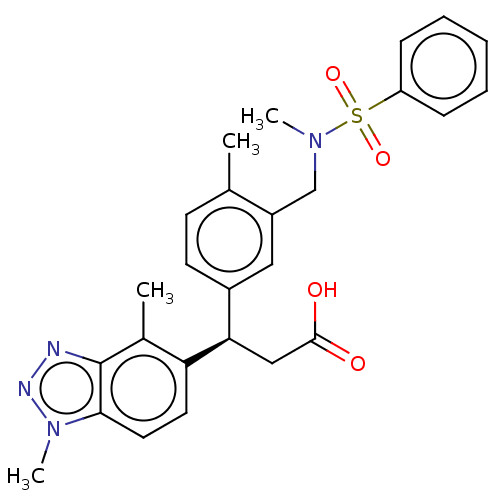
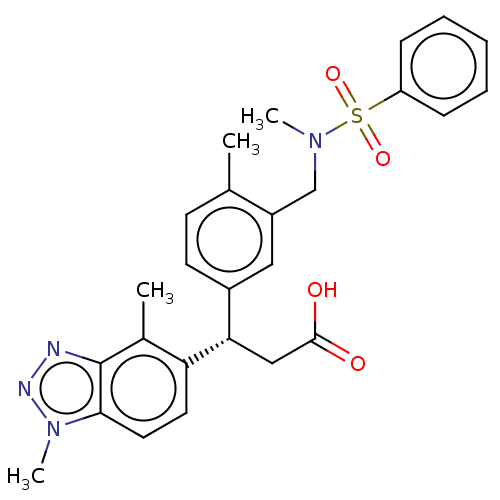
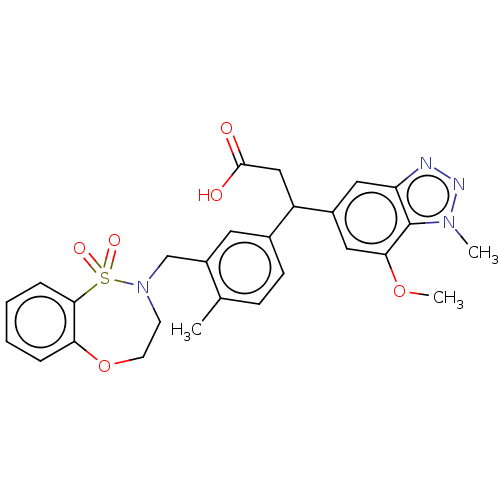
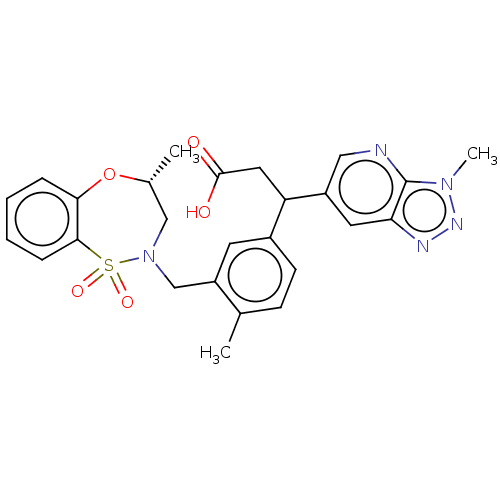
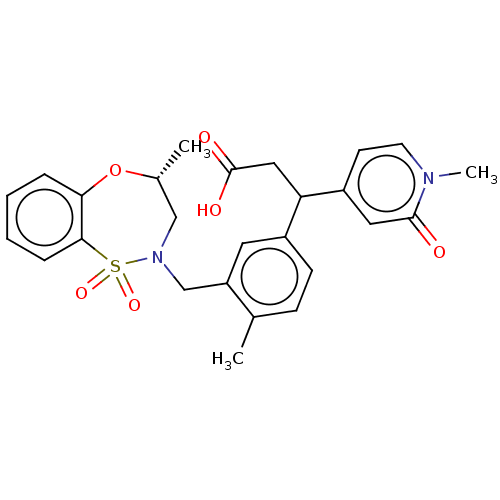
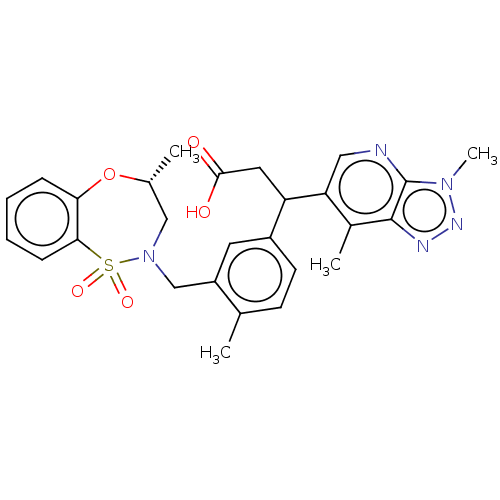
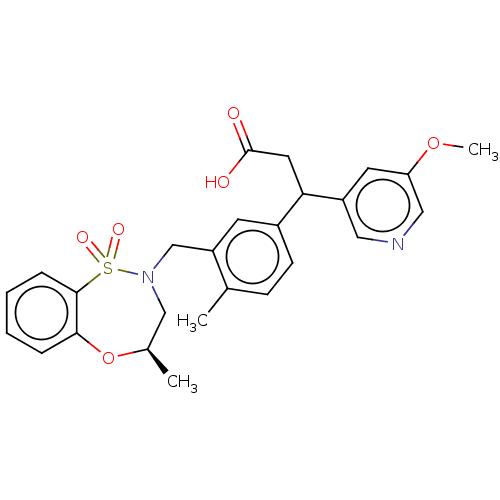
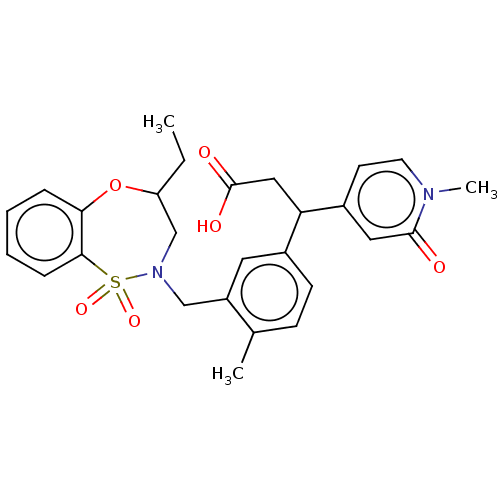
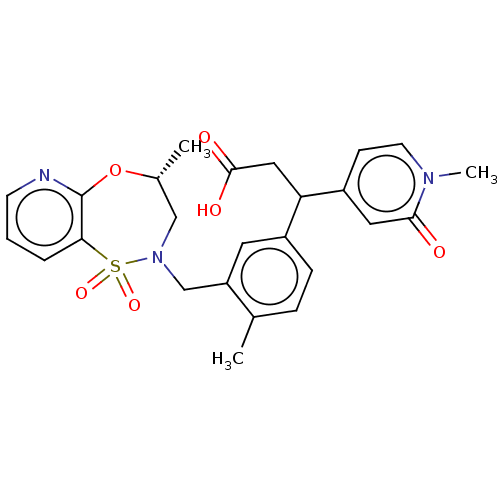
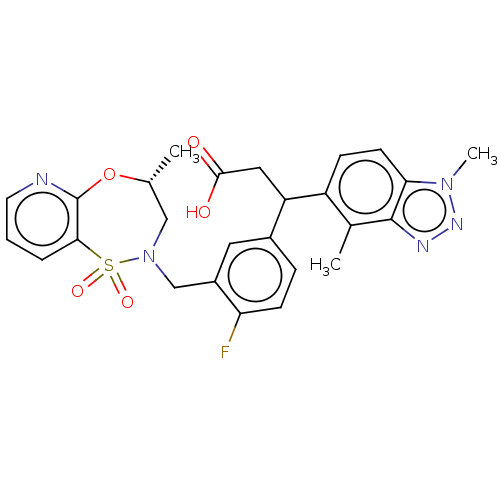
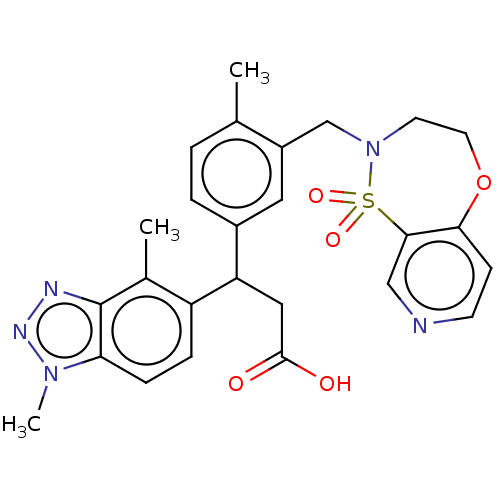
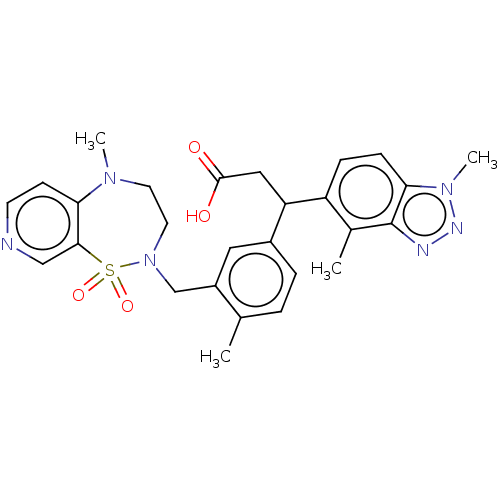
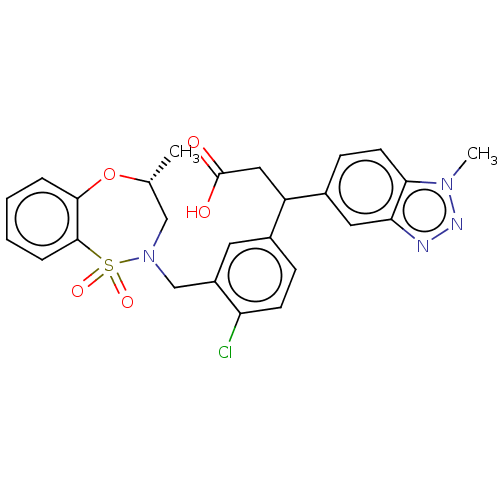
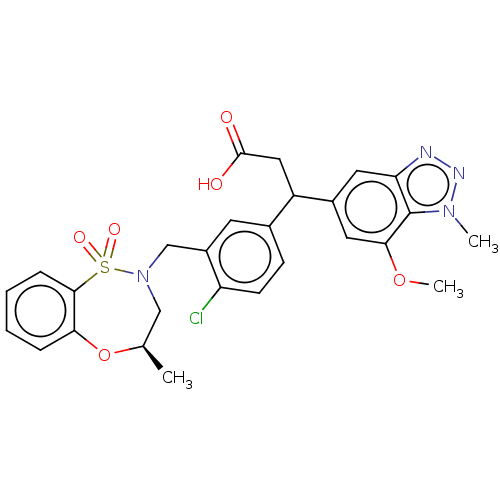
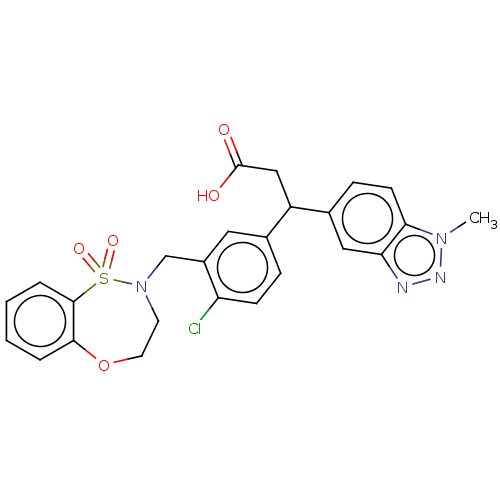
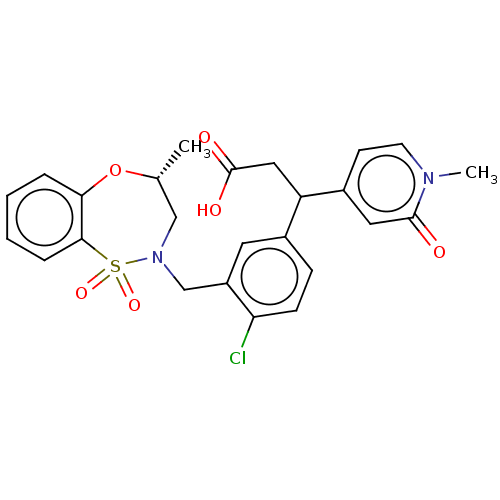
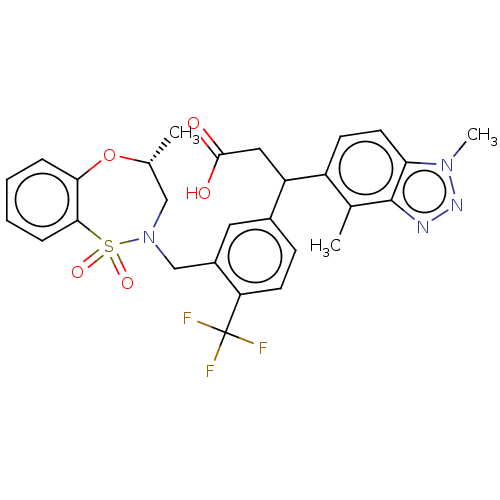
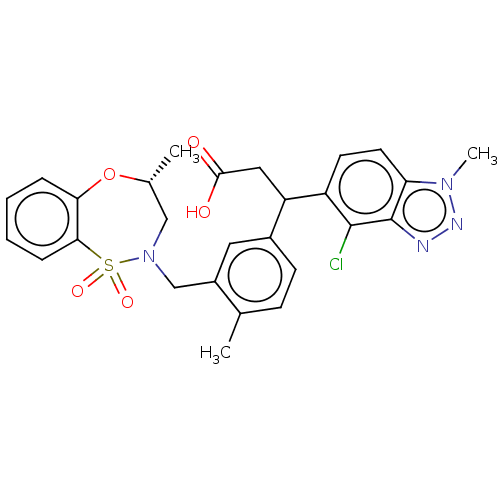
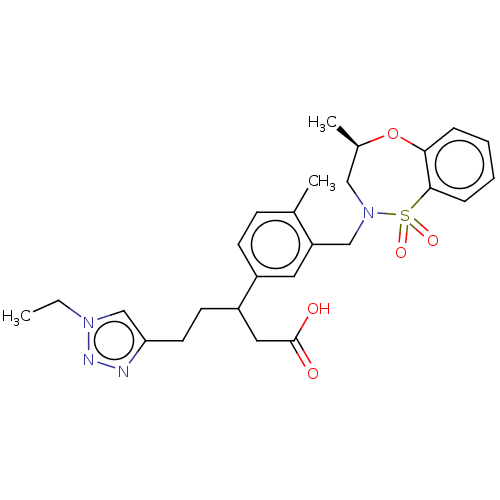
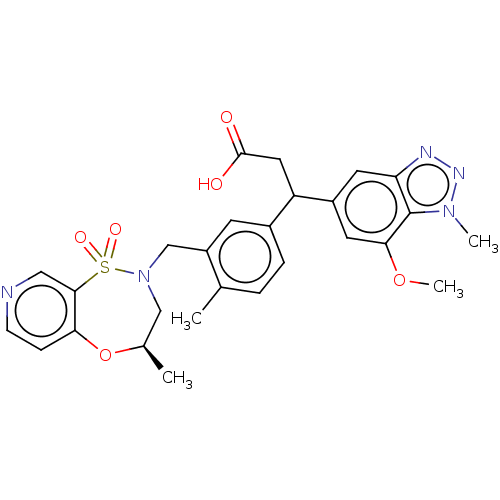
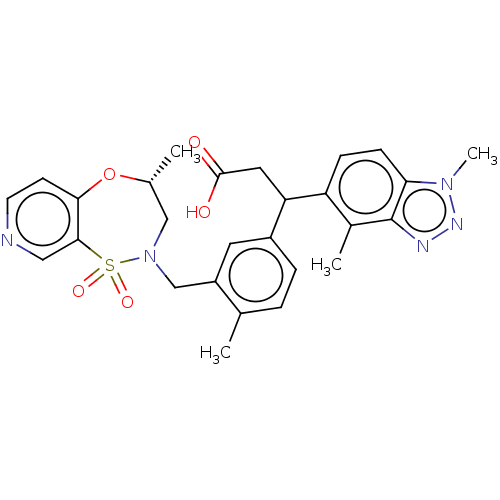
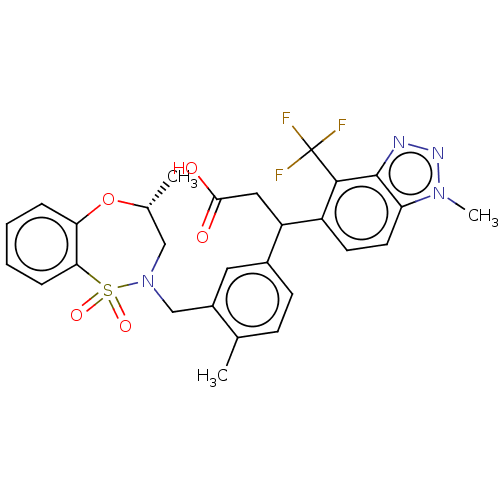
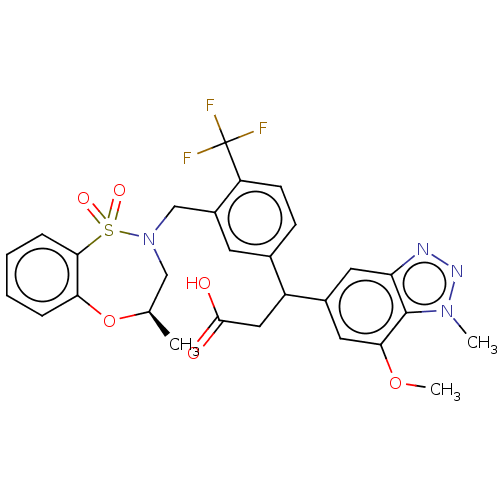
Report error Found 519 Enz. Inhib. hit(s) with all data for entry = 1051
Affinity DataIC50: 10nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataEC50: <10nMAssay Description:NAD(P)H:quinone oxidoreductase 1 (NQO1), also called DT diaphorase, is a homodimeric FAD-containing enzyme that catalyzes obligatory NAD(P)H-dependen...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataEC50: <10nMAssay Description:NAD(P)H:quinone oxidoreductase 1 (NQO1), also called DT diaphorase, is a homodimeric FAD-containing enzyme that catalyzes obligatory NAD(P)H-dependen...More data for this Ligand-Target Pair
Affinity DataEC50: <10nMAssay Description:NAD(P)H:quinone oxidoreductase 1 (NQO1), also called DT diaphorase, is a homodimeric FAD-containing enzyme that catalyzes obligatory NAD(P)H-dependen...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:100 nL of 100× compound dose response curves (serial 3-fold dilutions) in DMSO are stamped using an Echo liquid handling system (Labcyte) into 384-we...More data for this Ligand-Target Pair