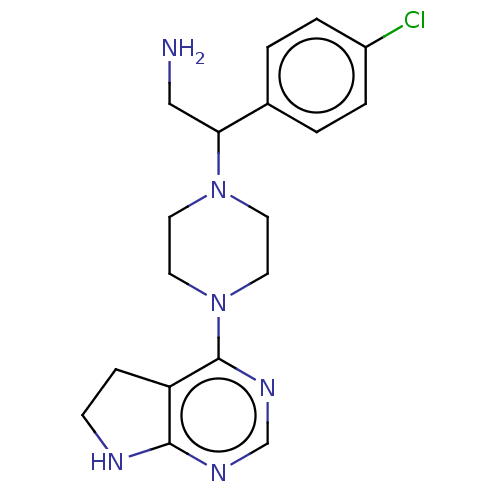
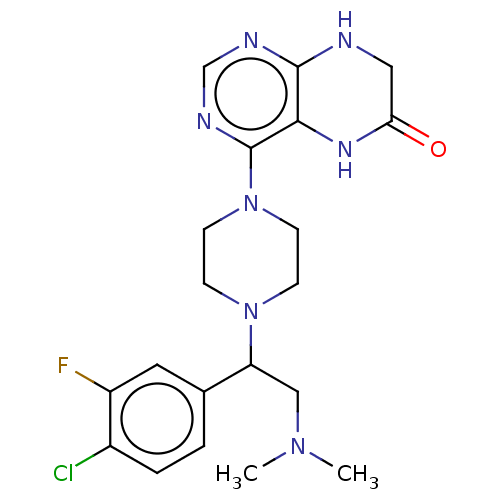
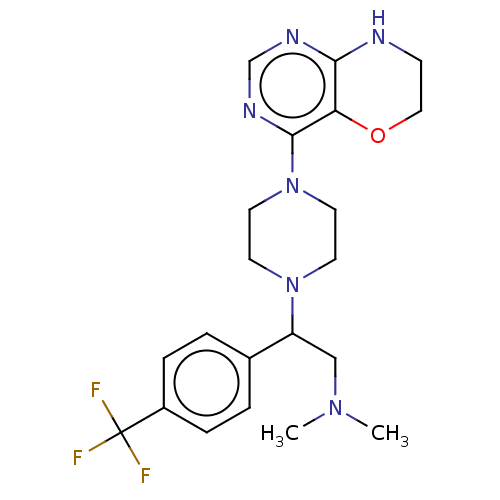
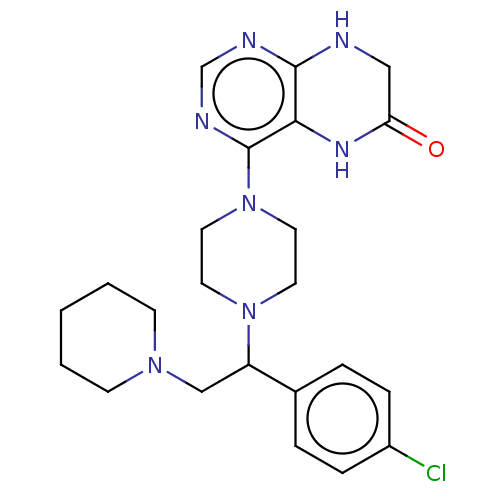
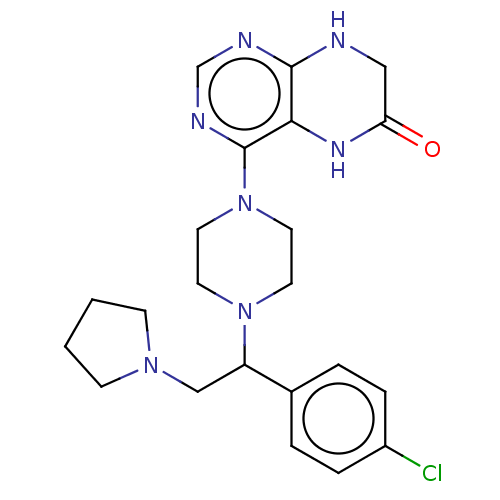
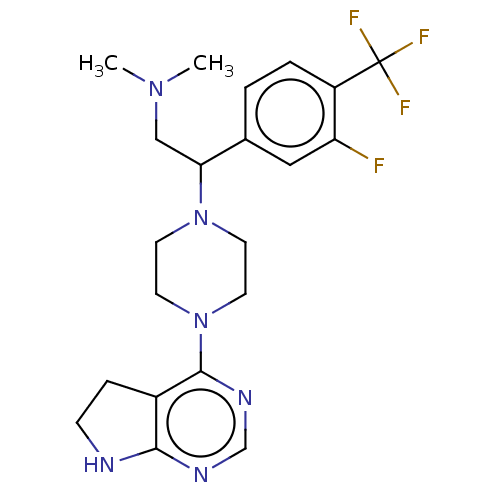
Report error Found 130 Enz. Inhib. hit(s) with all data for entry = 2948
Affinity DataIC50: 62nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:P70S6K inhibitor compounds were diluted and plated in 96 well plates. A reaction mixture including the following components was then added to the com...More data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:A TTP Mosquito liquid handling instrument was used to place 125 nl of the appropriate concentration of inhibitor in 100% DMSO (for a dose response cu...More data for this Ligand-Target Pair