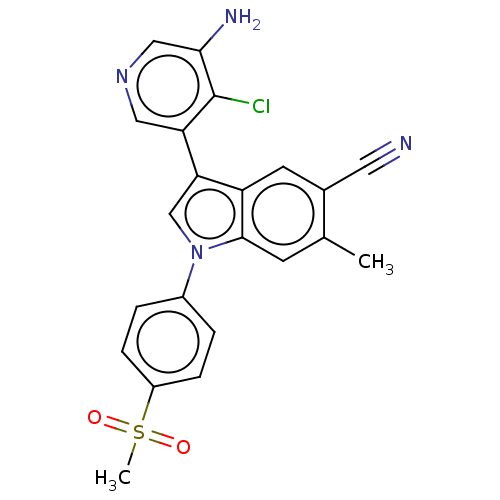
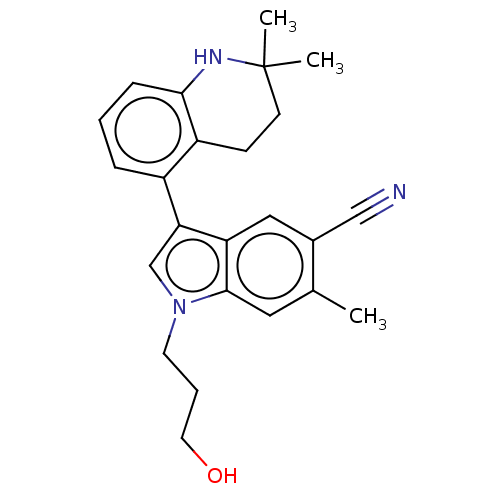
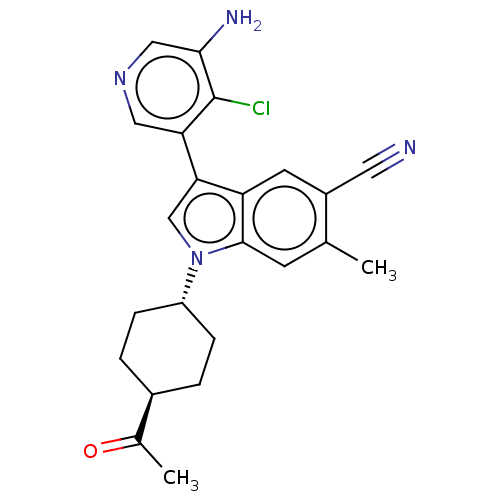
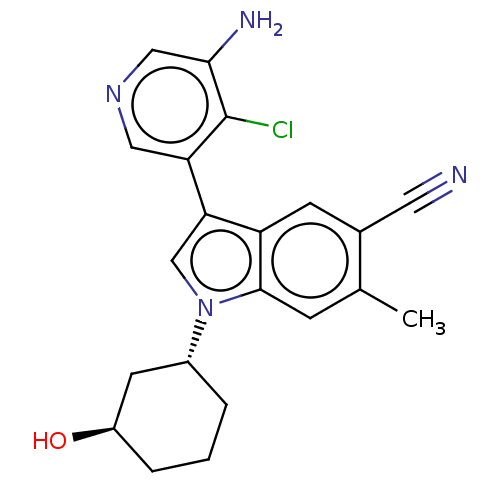
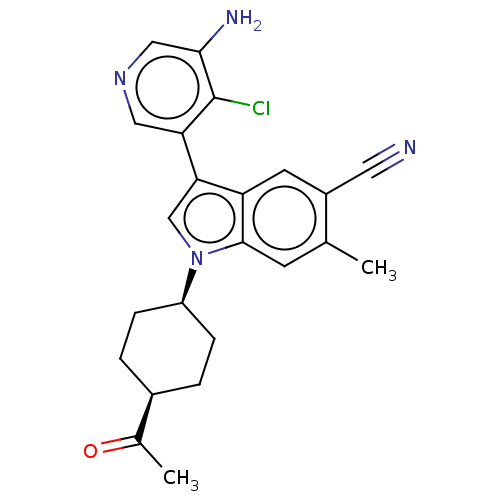
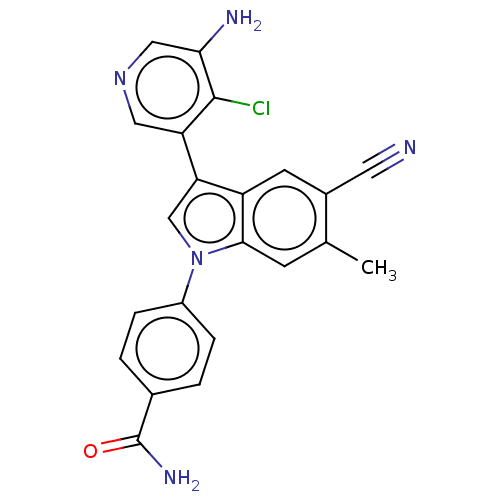
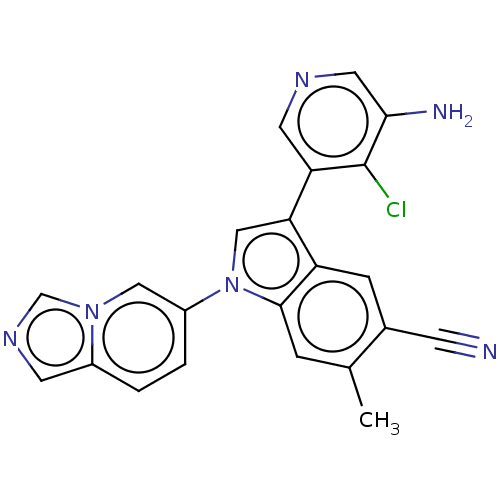
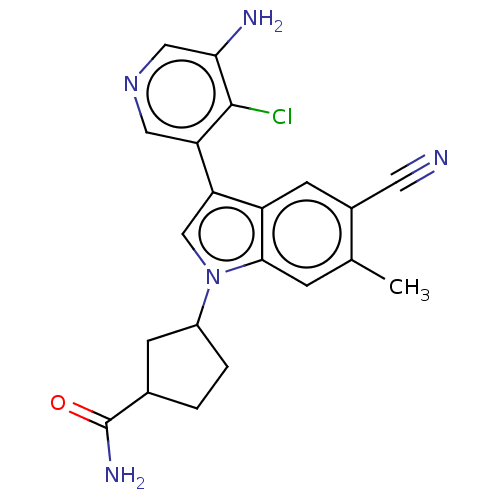
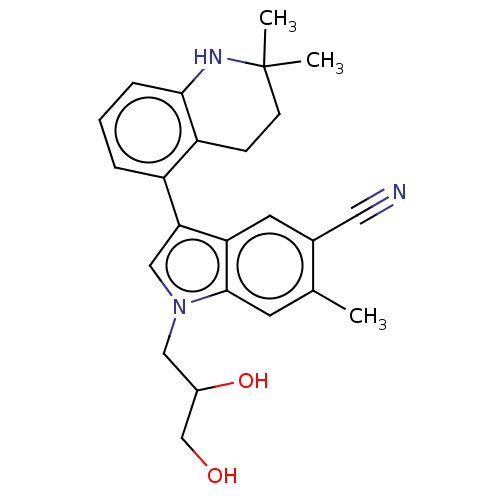
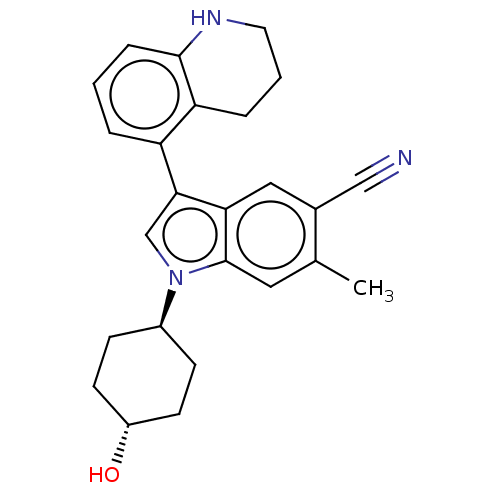
Report error Found 112 Enz. Inhib. hit(s) with all data for entry = 9072
Affinity DataIC50: 1nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 23nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 23nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 26nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 26nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 27nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:Representative compounds of the present invention were serially and separately diluted 3-fold in DMSO to obtain a total of twelve concentrations. The...More data for this Ligand-Target Pair