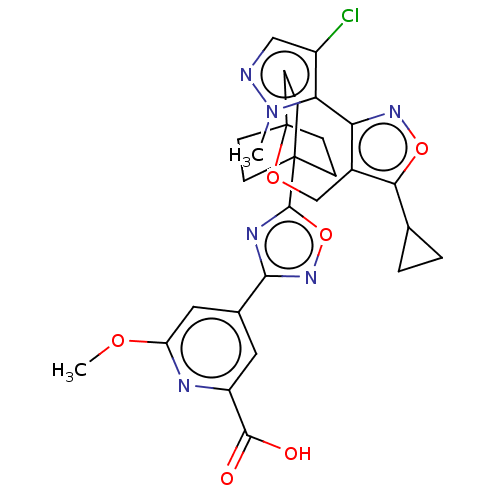
Report error Found 435 Enz. Inhib. hit(s) with all data for entry = 9265
Affinity DataEC50: 1nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 1nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 1nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 1nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 1nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 1nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 1nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 3nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 4nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 4nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 4nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 5nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 5nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 6nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 6nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 7nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 7nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 7nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 7.20nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 8nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 9nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 10nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 10nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 10nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 12nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 13nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 13nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 14nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 15nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 15nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 16nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 16nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 16nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 17nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 17nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 17nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair