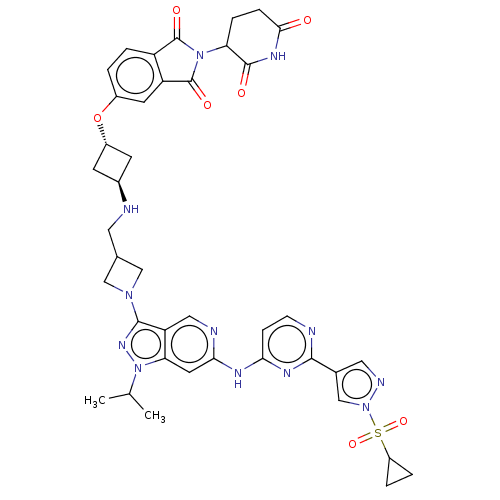
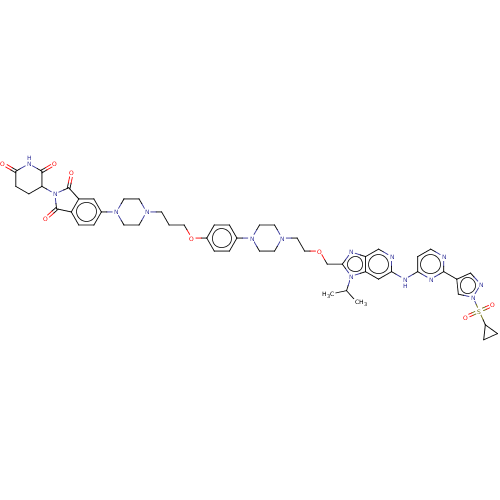
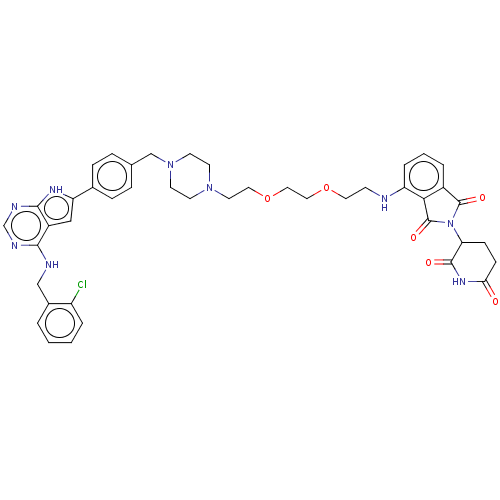
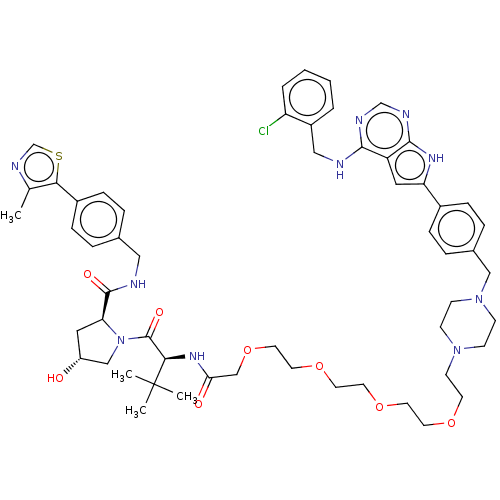
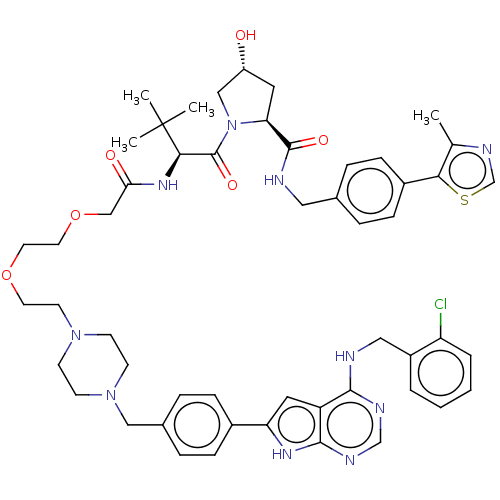
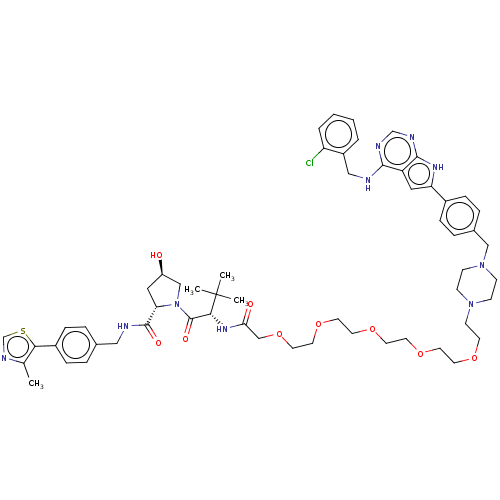
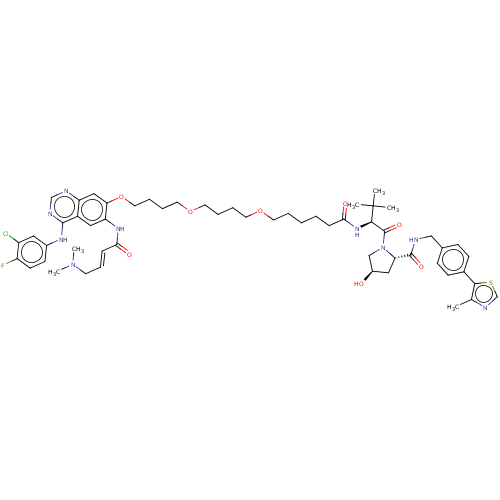
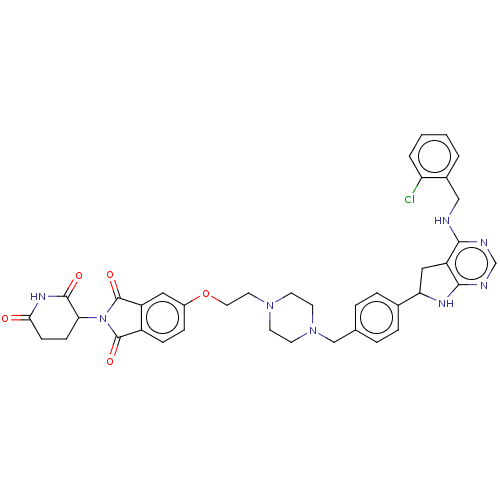
Report error Found 521 Enz. Inhib. hit(s) with all data for entry = 9869
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 0.140nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 0.260nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 0.270nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 0.290nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210,T790M](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 0.300nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 0.350nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 0.400nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 0.670nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 0.910nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 1nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 1.30nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210,T790M](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 1.60nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 1.70nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 1.90nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210,T790M](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 2.10nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 2.10nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 2.90nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 4nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 4.10nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 4.20nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 4.20nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 4.70nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210,T790M](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 4.90nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 4.90nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 5.5nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 7.20nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 7.5nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 8.30nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 8.40nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 9.80nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor [1-18,20-745,747-749,751-1210,T790M](Human)
Arvinas Operations
US Patent
Arvinas Operations
US Patent
Affinity DataIC50: 21nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:All compounds and PROTACs were serially diluted in three-fold increments using 100% DMSO, followed by an intermediate 10-fold dilution using Buffer A...More data for this Ligand-Target Pair