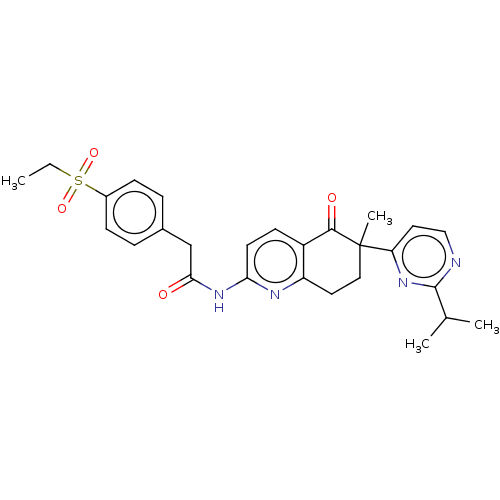
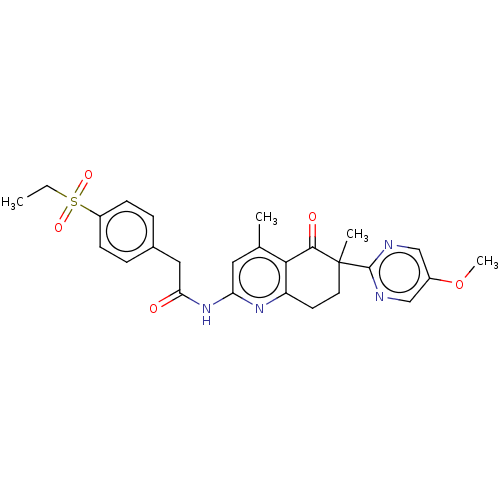
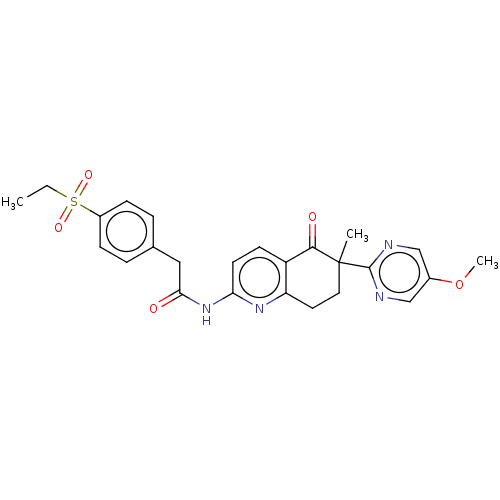
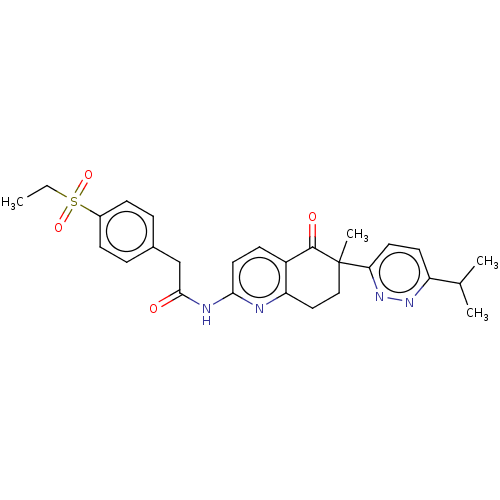
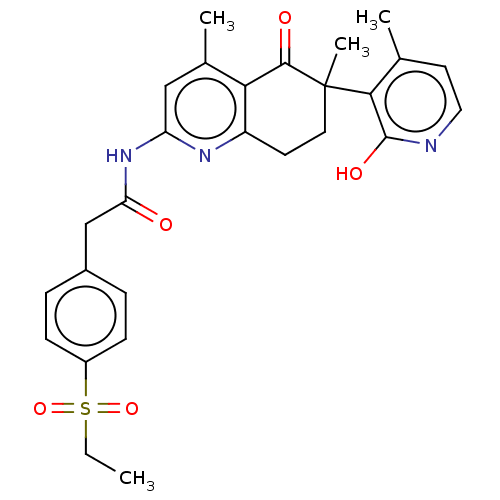
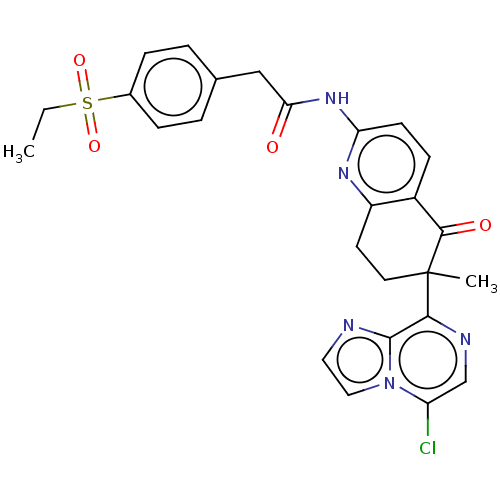
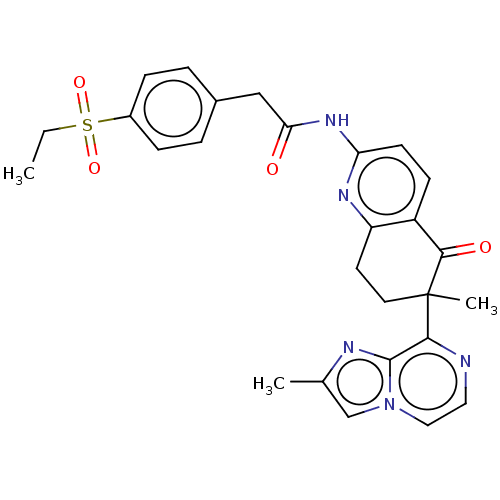
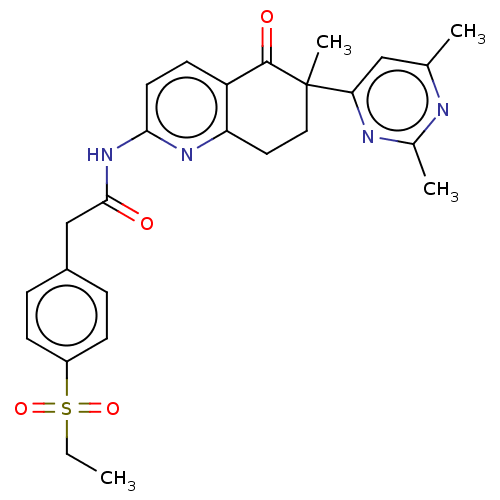
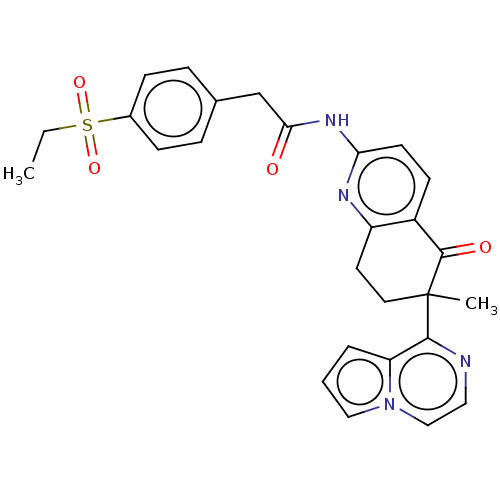
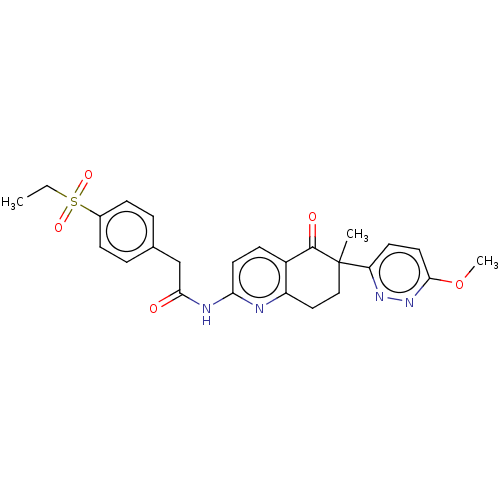
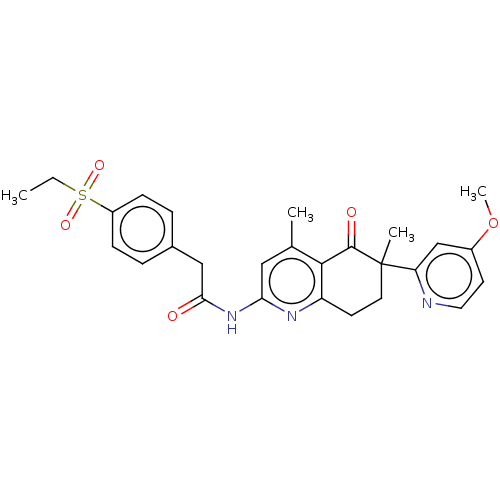
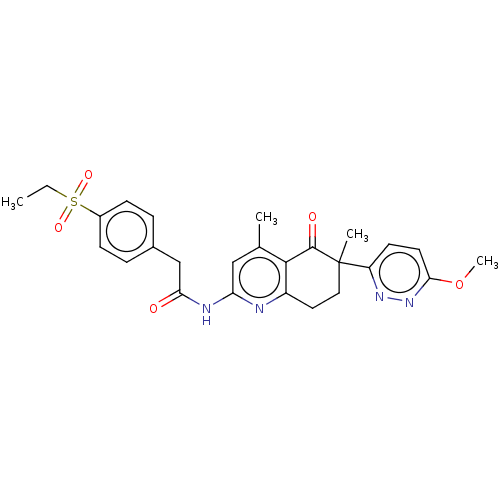
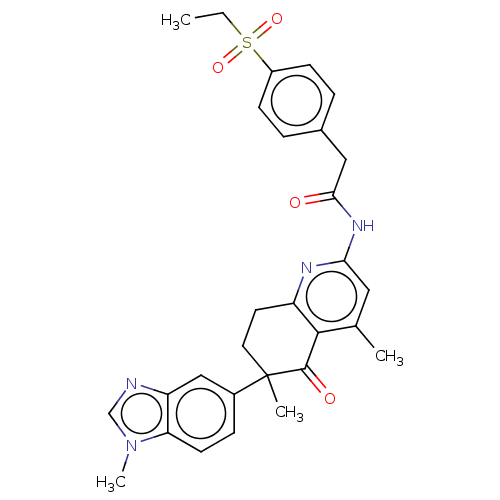
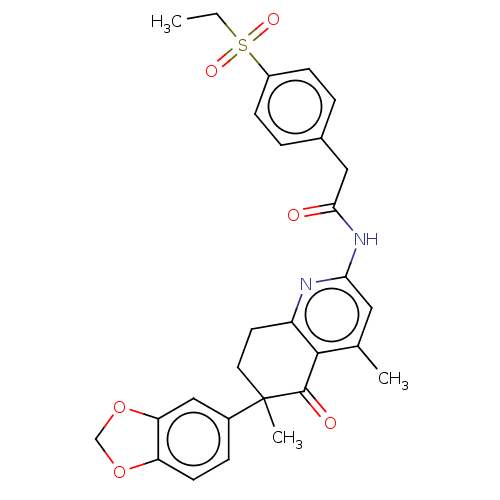
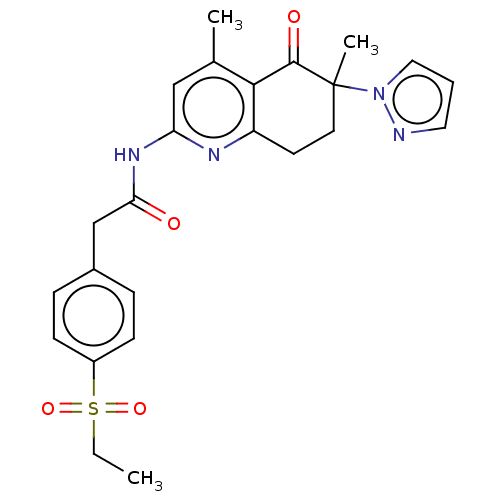
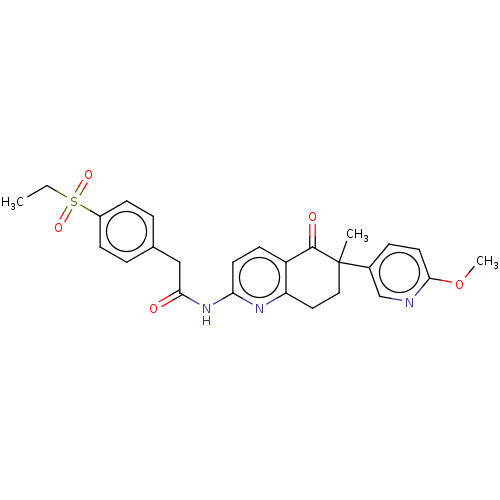
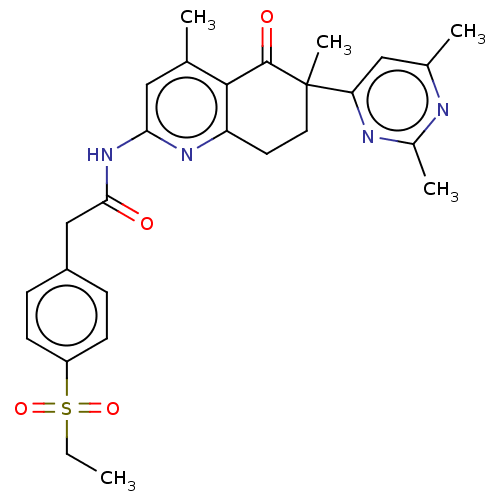
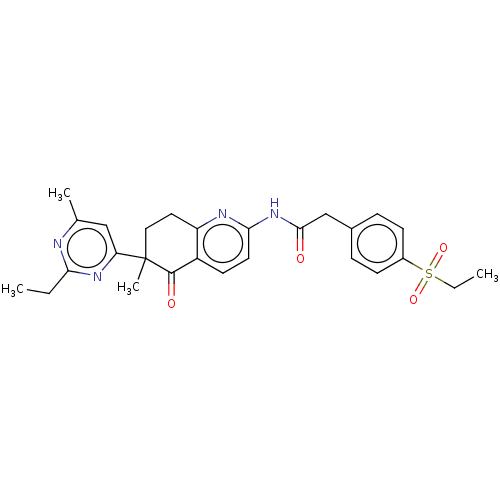
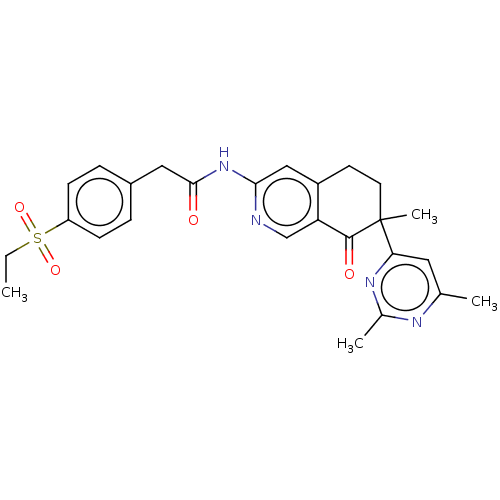
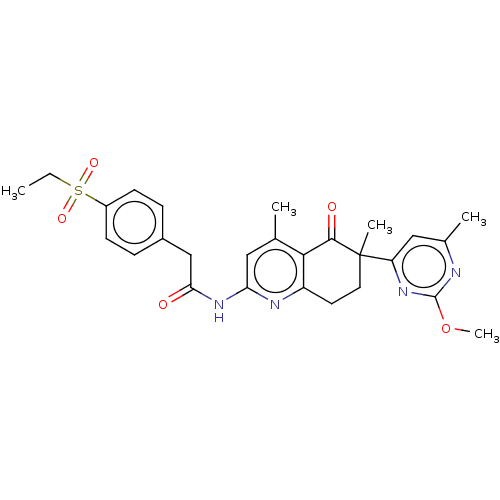
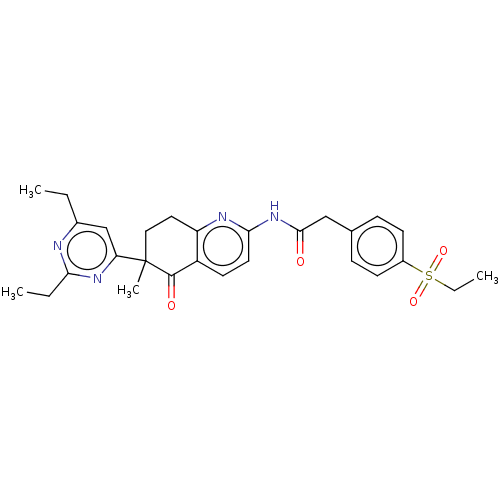
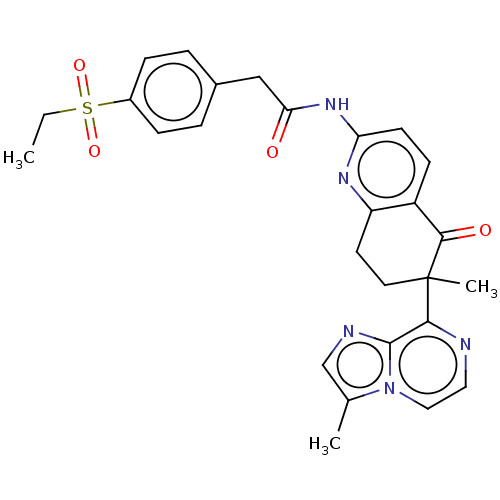
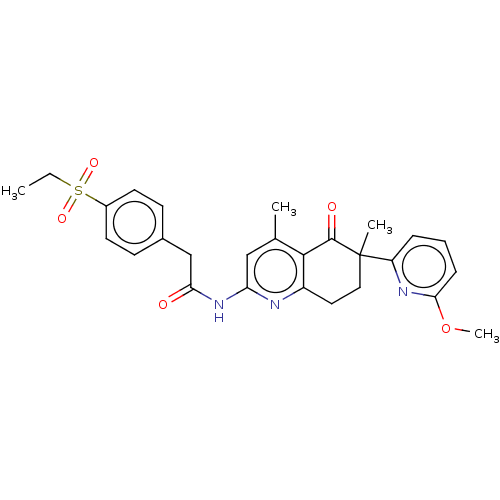
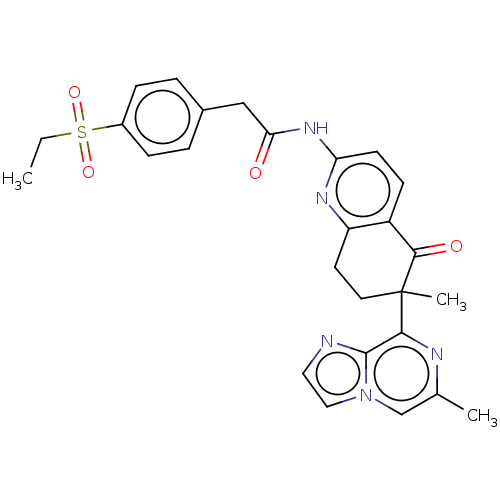
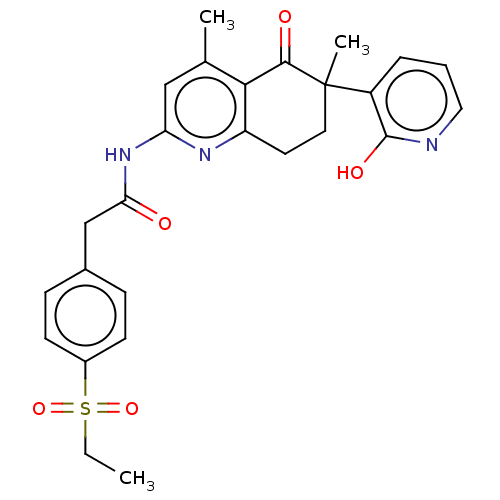
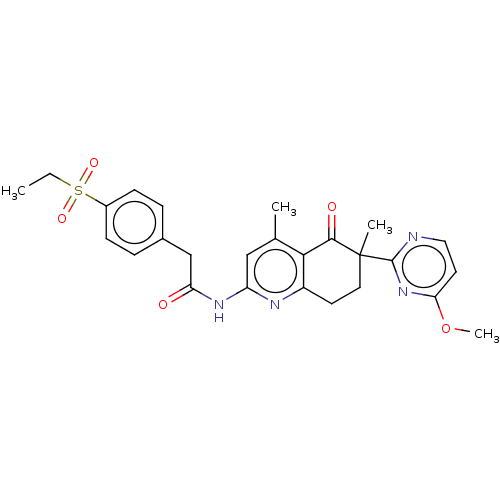
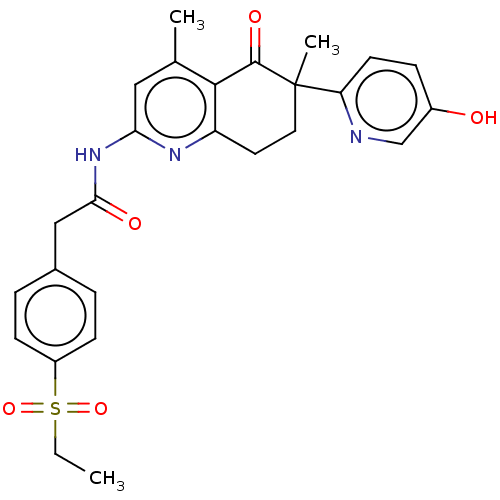
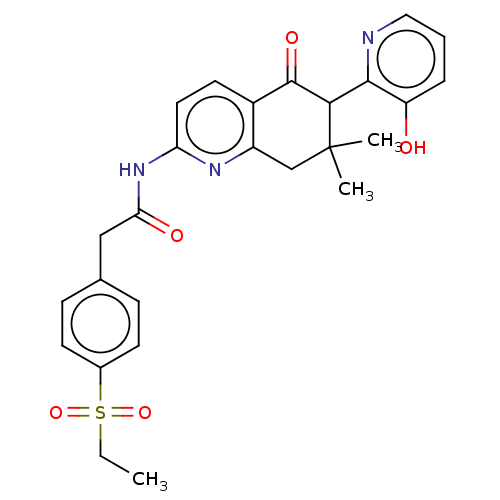
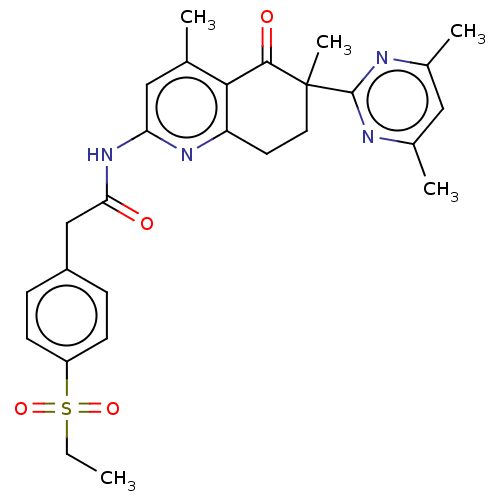
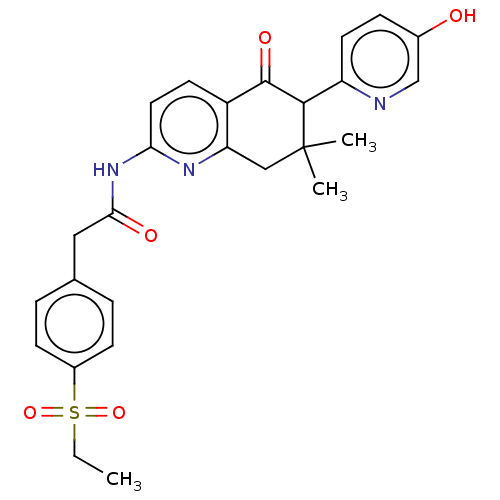
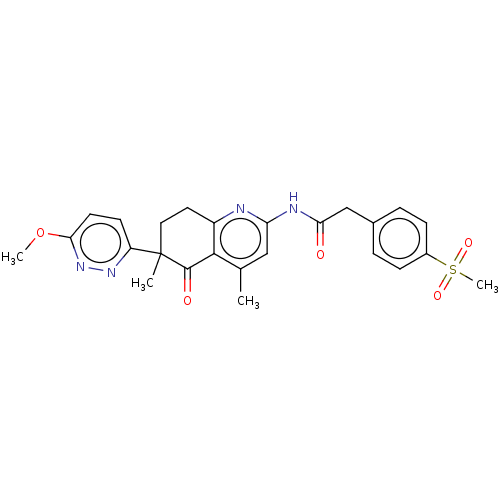
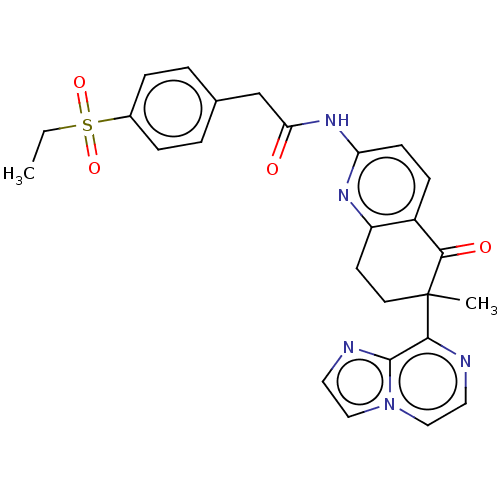
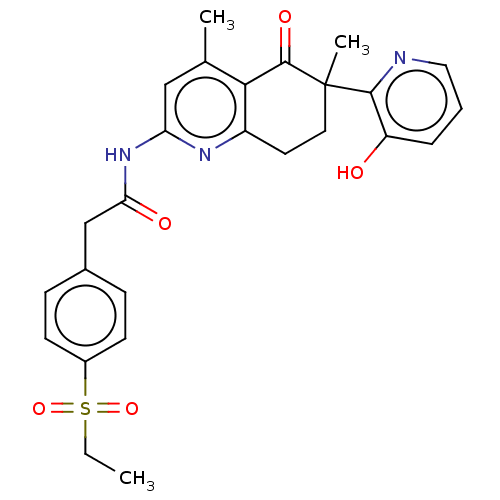
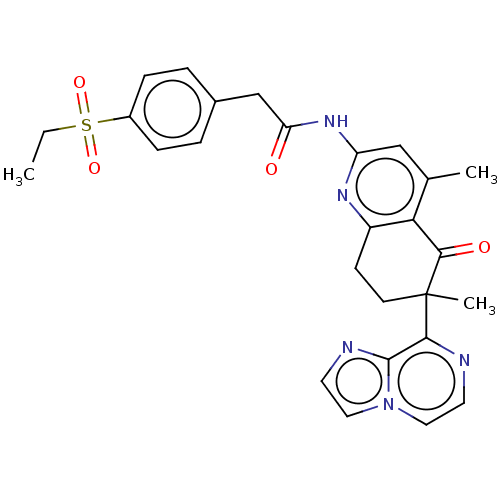
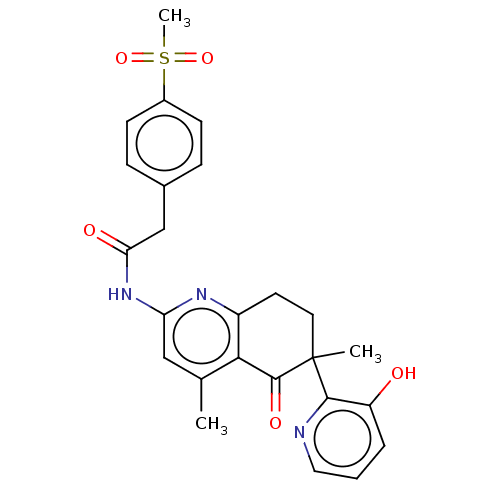
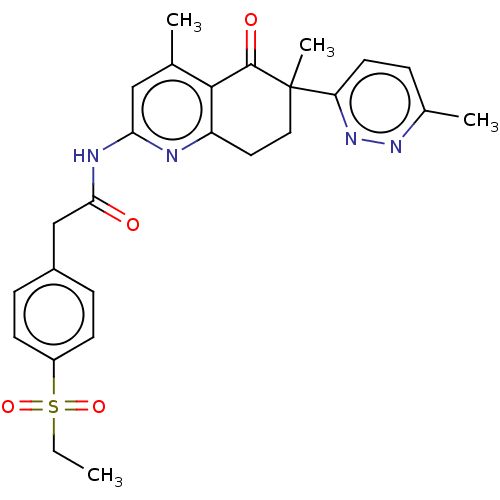
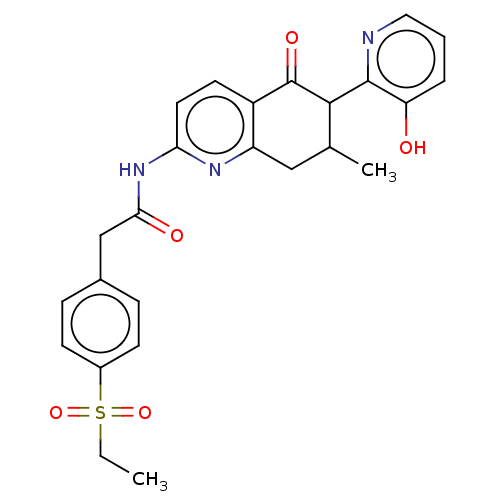
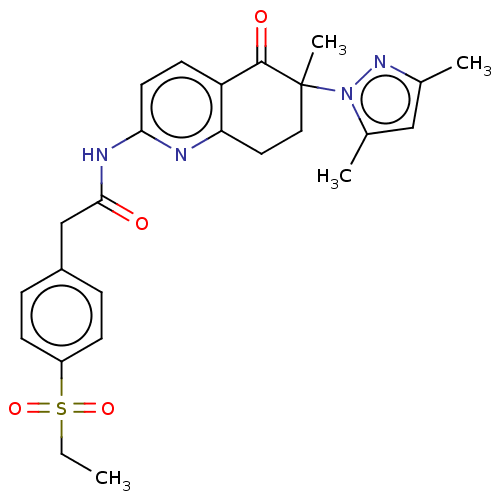
Report error Found 46 Enz. Inhib. hit(s) with all data for entry = 10421
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:ROR gamma radioligand binding was performed using 3H 25-Hydroxycholesterol in a competitive displacement assay using dextran charcoal method. Using 5...More data for this Ligand-Target Pair