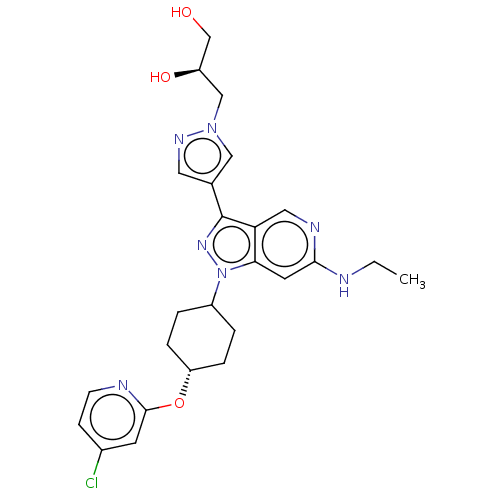
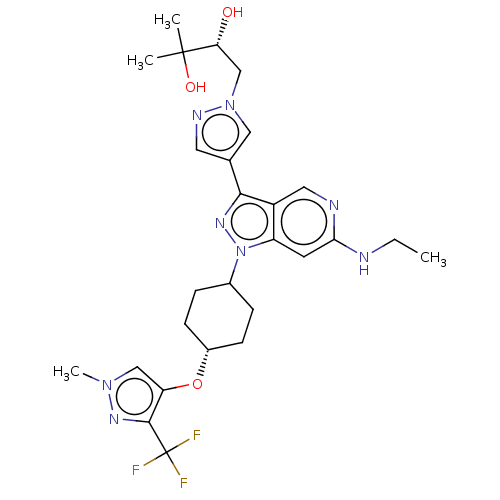
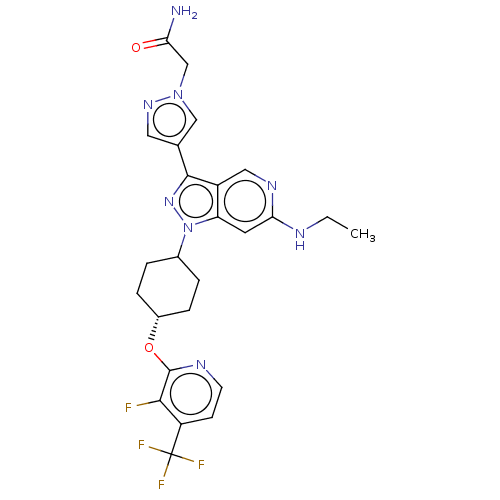
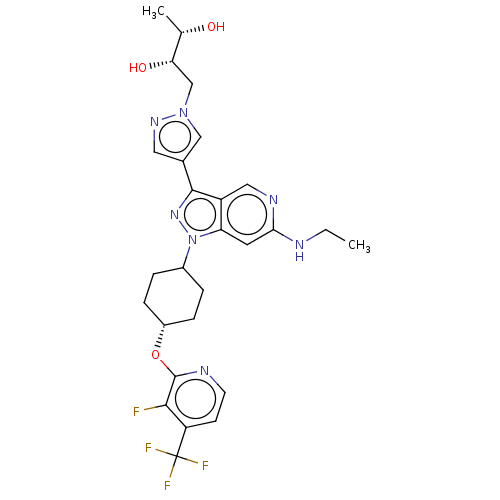
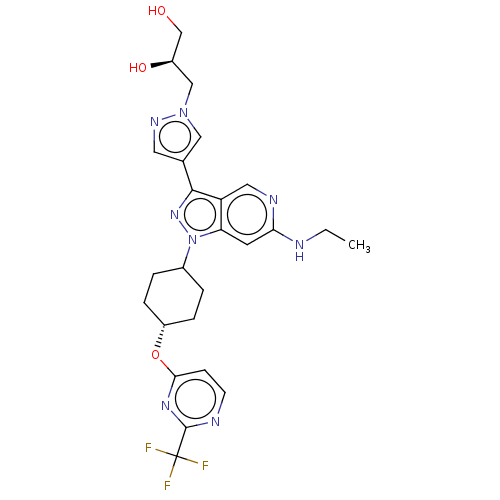
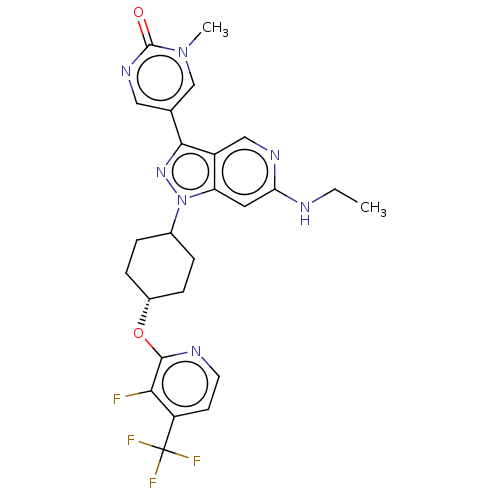
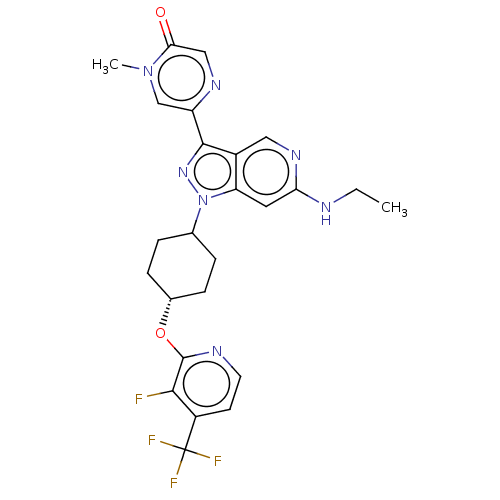
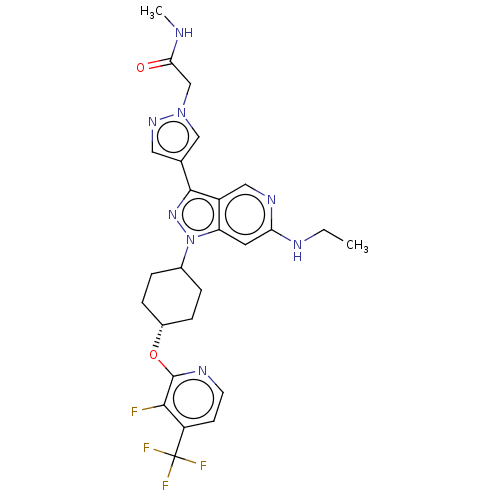
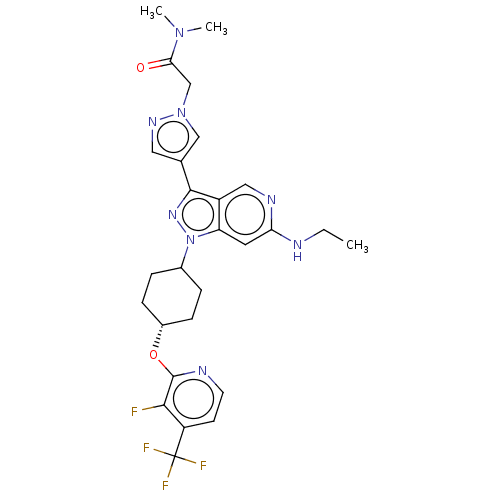
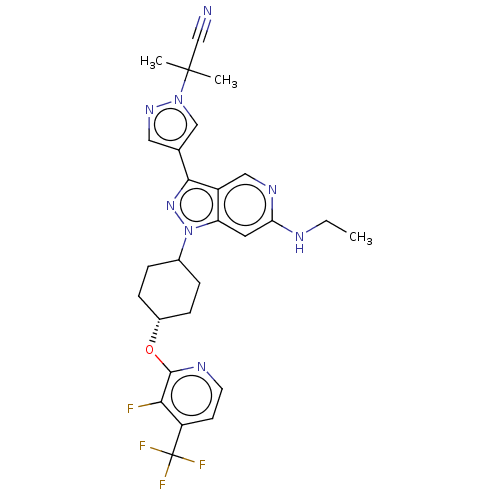
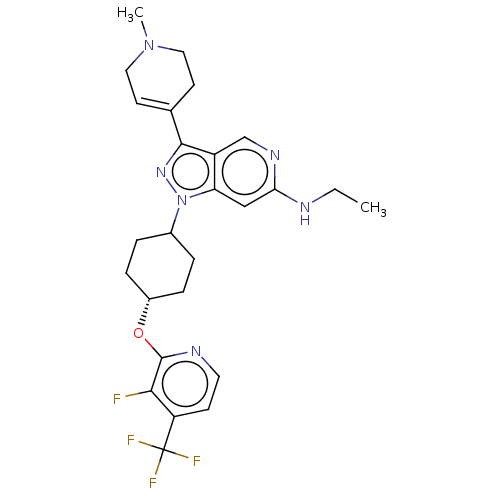
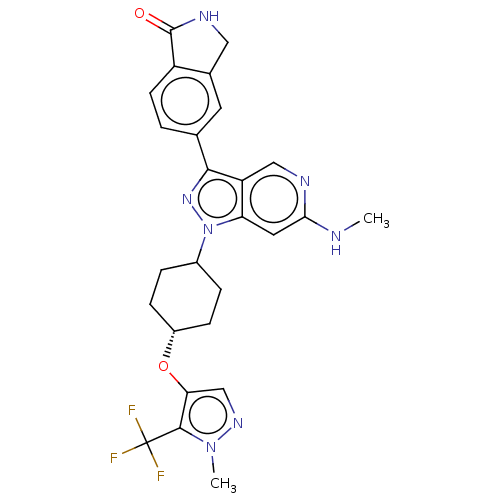
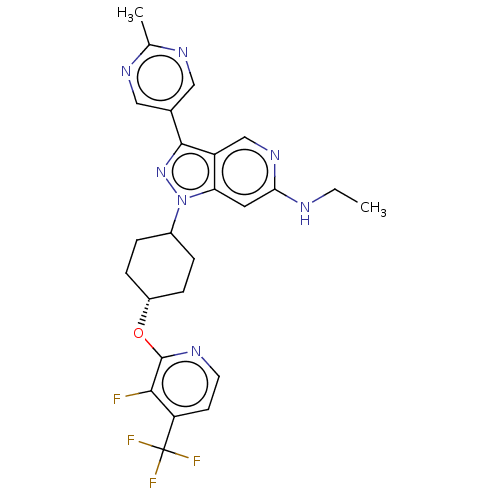
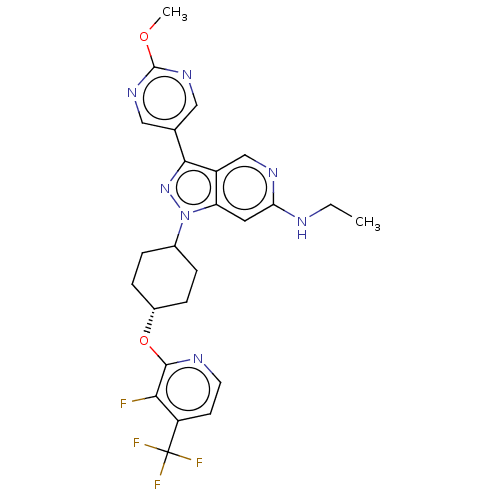
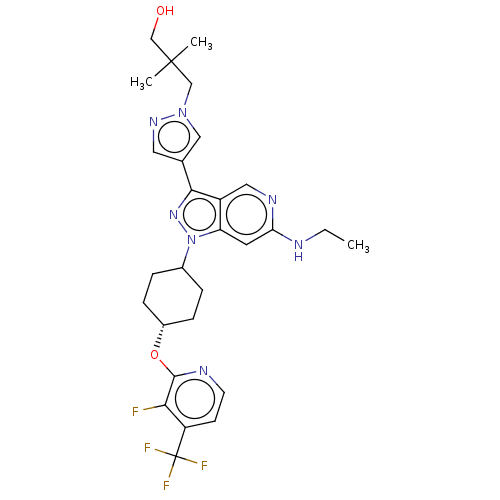
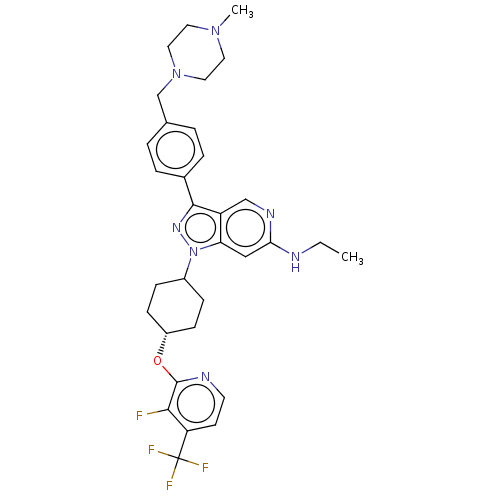
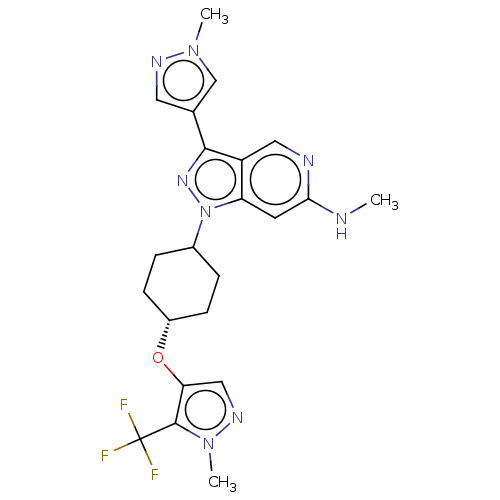
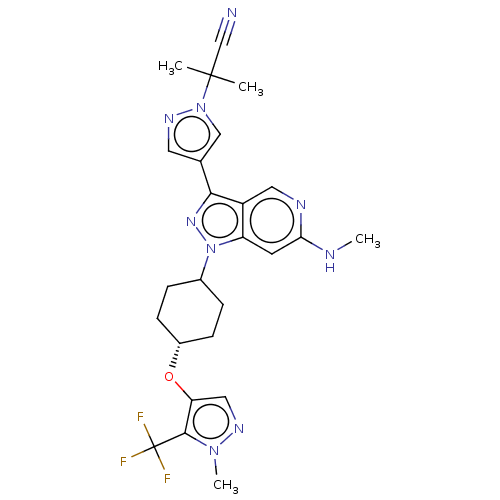
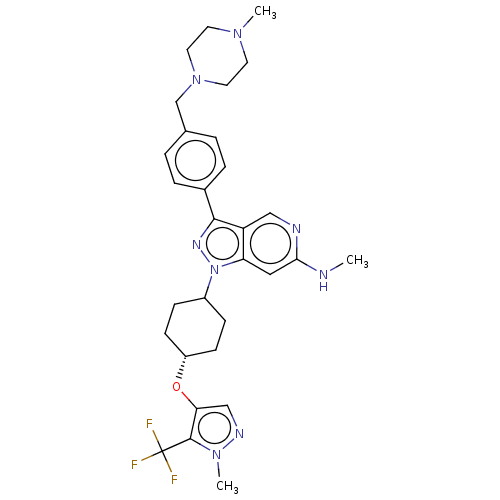
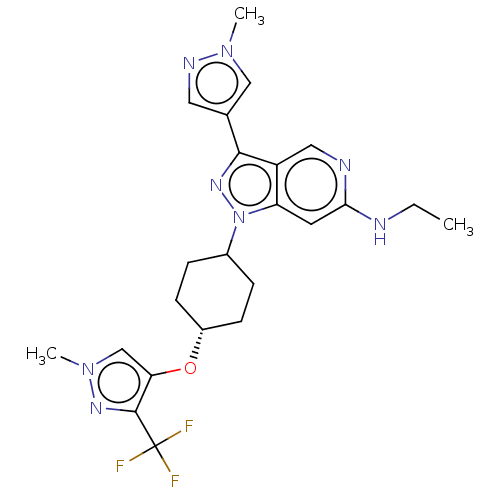
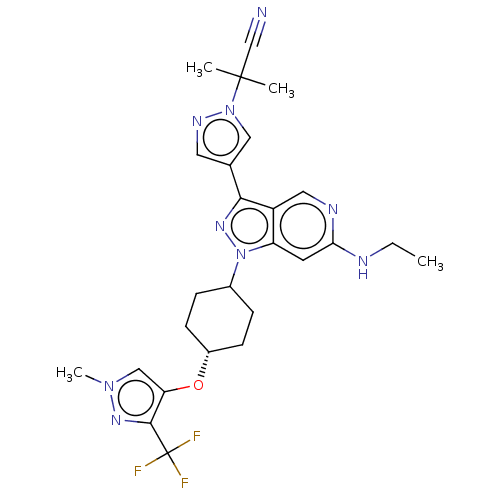
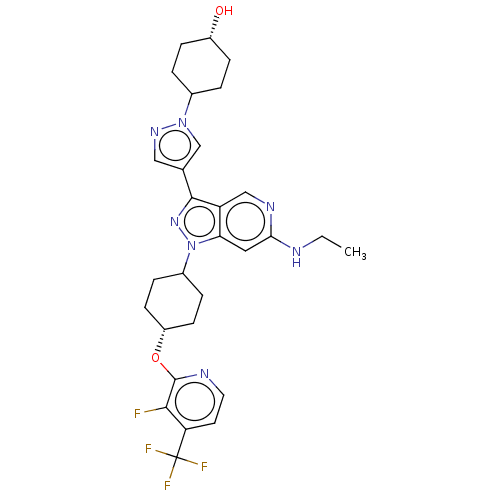
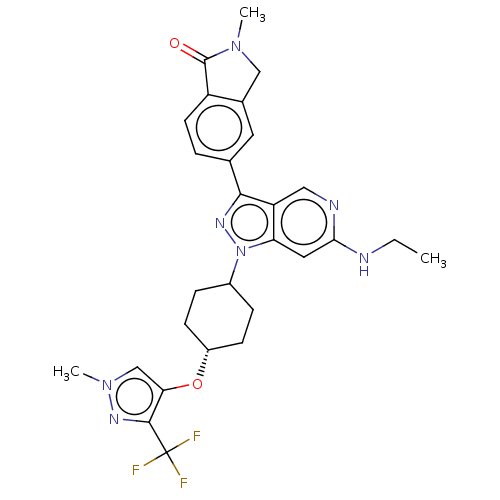
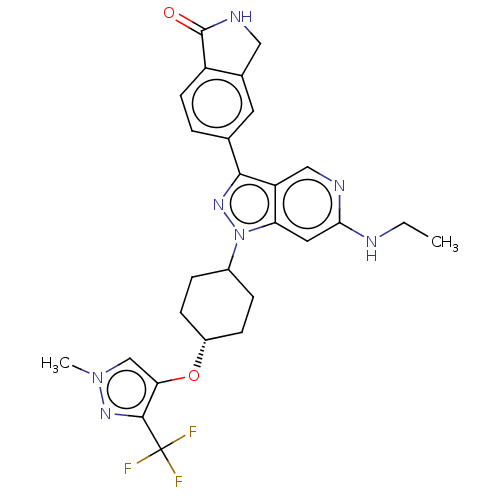
Report error Found 1533 Enz. Inhib. hit(s) with all data for entry = 10464
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:TYRO3: Compounds of Formula I were screened for their ability to inhibit TYRO3 kinase using Invitrogen's LanthaScreen Eu Kinase Binding technolog...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:TYRO3: Compounds of Formula I were screened for their ability to inhibit TYRO3 kinase using Invitrogen's LanthaScreen Eu Kinase Binding technolog...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:TYRO3: Compounds of Formula I were screened for their ability to inhibit TYRO3 kinase using Invitrogen's LanthaScreen Eu Kinase Binding technolog...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:TYRO3: Compounds of Formula I were screened for their ability to inhibit TYRO3 kinase using Invitrogen's LanthaScreen Eu Kinase Binding technolog...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:TYRO3: Compounds of Formula I were screened for their ability to inhibit TYRO3 kinase using Invitrogen's LanthaScreen Eu Kinase Binding technolog...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:TYRO3: Compounds of Formula I were screened for their ability to inhibit TYRO3 kinase using Invitrogen's LanthaScreen Eu Kinase Binding technolog...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:AXL: Compounds of Formula I were screened for their ability to inhibit AXL kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:TYRO3: Compounds of Formula I were screened for their ability to inhibit TYRO3 kinase using Invitrogen's LanthaScreen Eu Kinase Binding technolog...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:TYRO3: Compounds of Formula I were screened for their ability to inhibit TYRO3 kinase using Invitrogen's LanthaScreen Eu Kinase Binding technolog...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:MER: Compounds of Formula I were screened for their ability to inhibit MER kinase using Invitrogen's LanthaScreen Eu Kinase Binding technology. H...More data for this Ligand-Target Pair