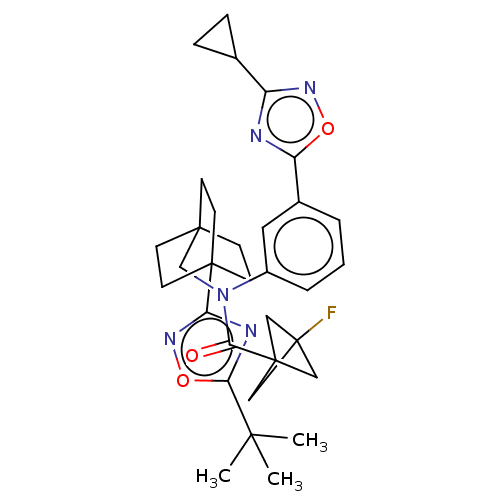
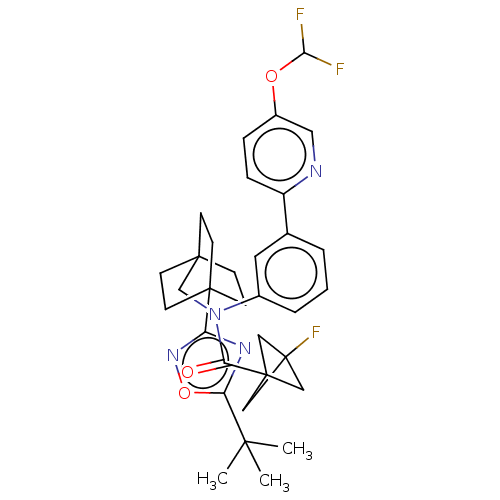
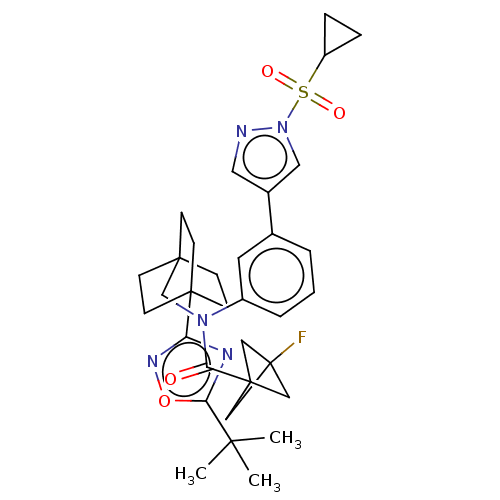
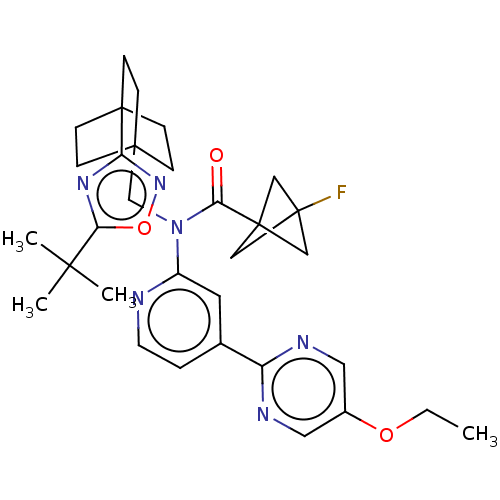
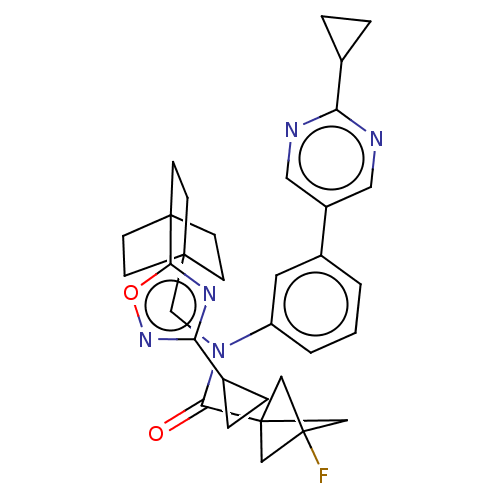
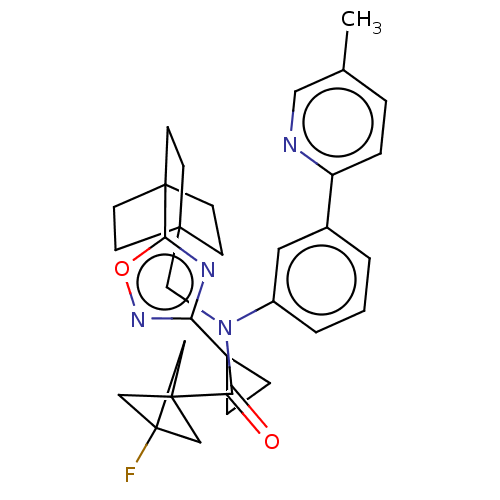
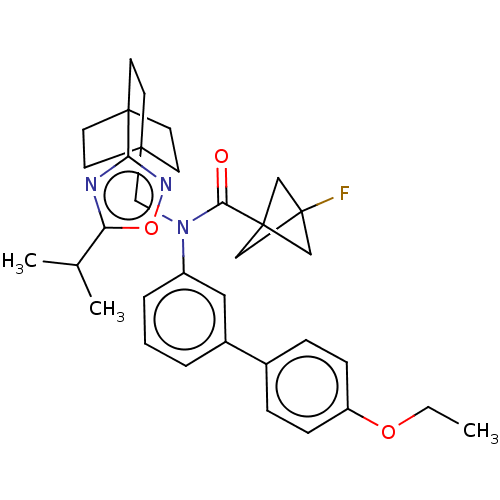
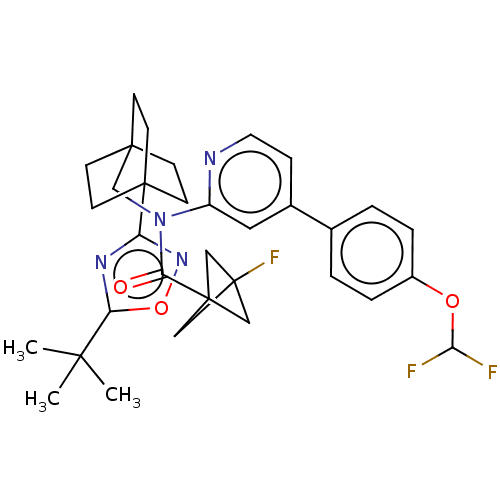
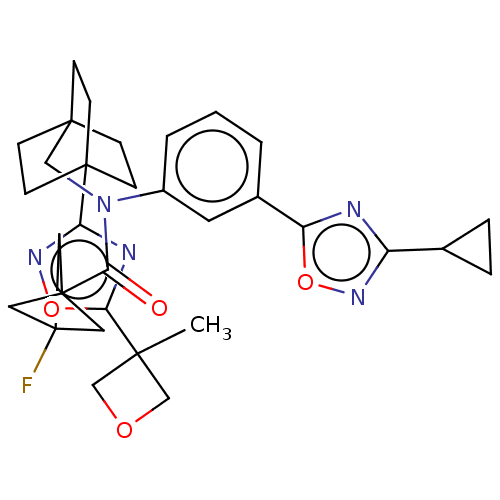
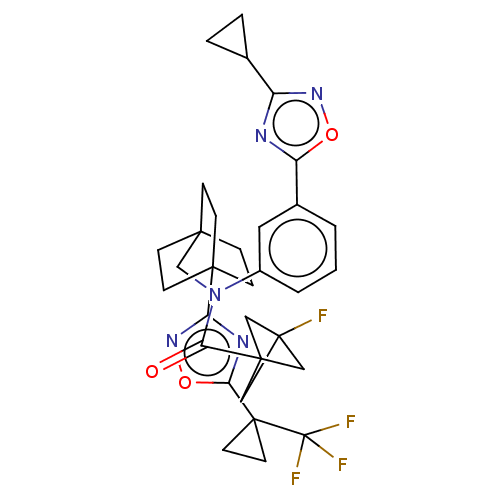
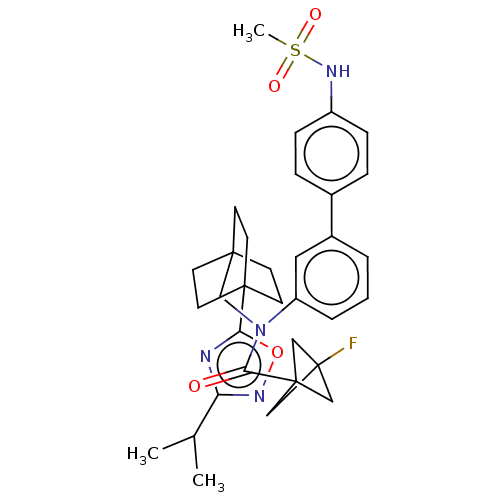
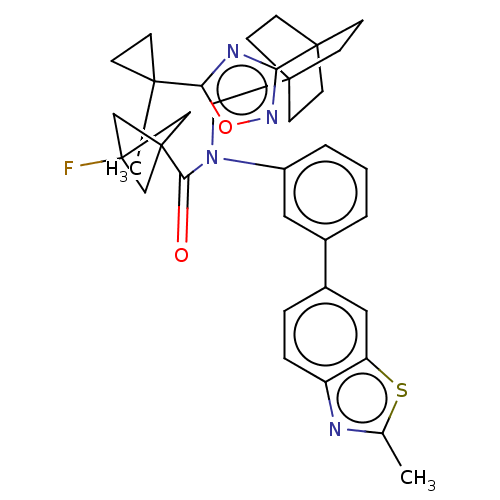
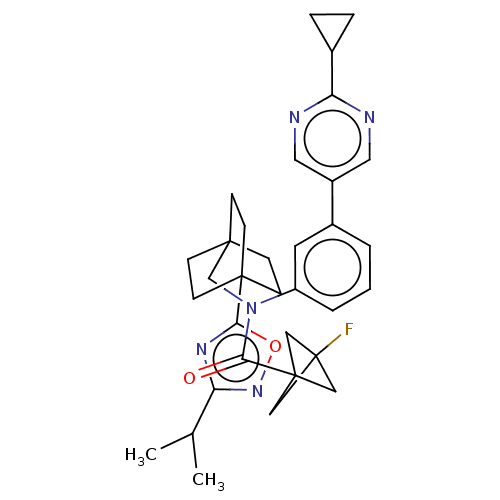
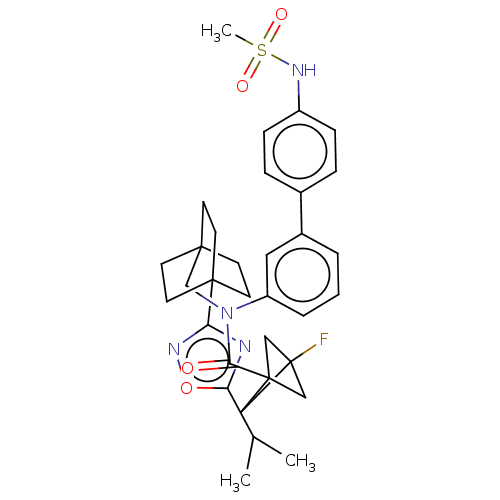
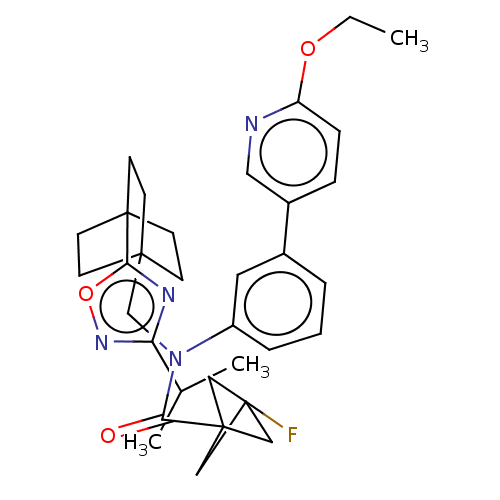
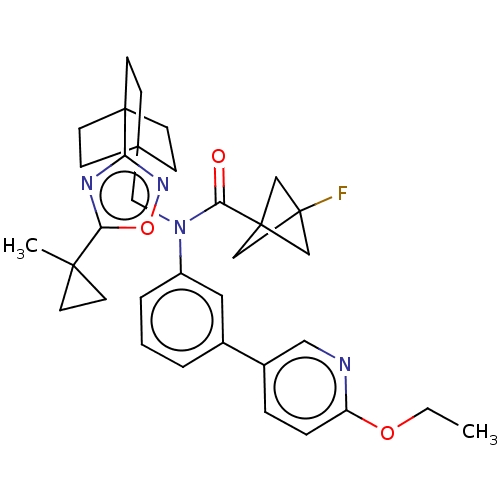
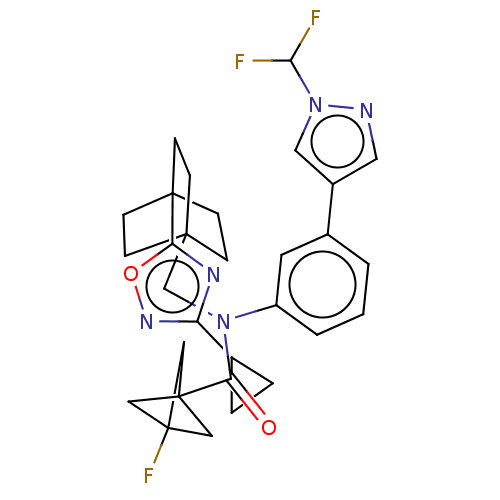
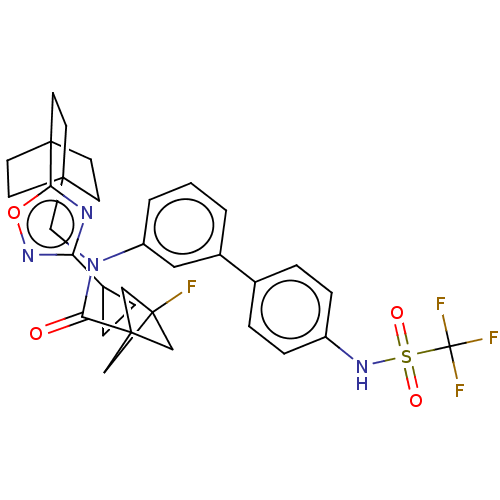
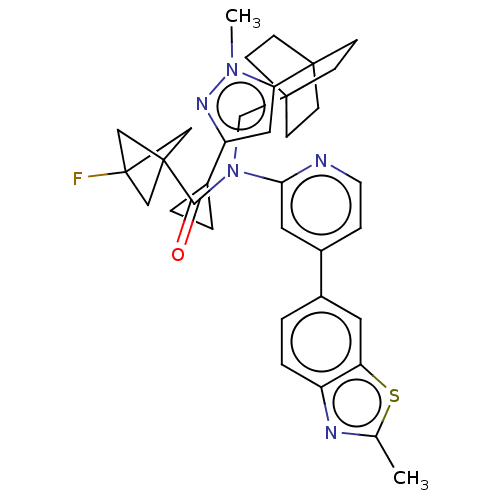
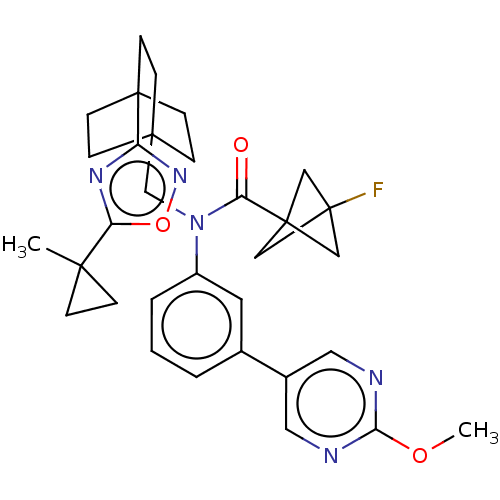
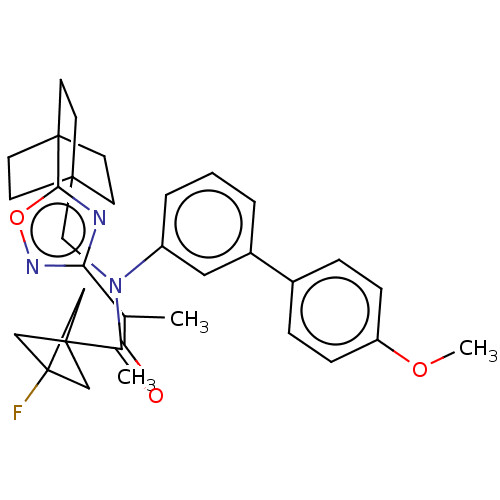
Report error Found 410 Enz. Inhib. hit(s) with all data for entry = 10475
Affinity DataEC50: 12nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 14nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 15nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 15nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 15nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 17nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 17nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 19nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 20nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 20nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 22nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 22nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 23nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 24nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 24nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 25nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 26nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 26nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 28nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 29nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 30nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 31nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 32nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 34nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 35nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 35nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 35nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 35nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 37nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 39nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 40nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 40nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 42nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 43nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 43nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 45nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 46nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 46nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 46nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 47nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 47nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 47nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 48nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 48nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 48nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 49nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 51nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 51nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair
Affinity DataEC50: 52nMAssay Description:A Gal4-hFXR fusion construct reporter system was used as the primary assay to characterize compound activity. A construct including 5 copies of the G...More data for this Ligand-Target Pair