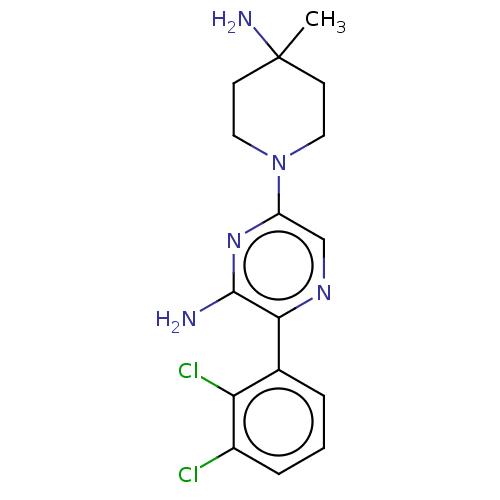
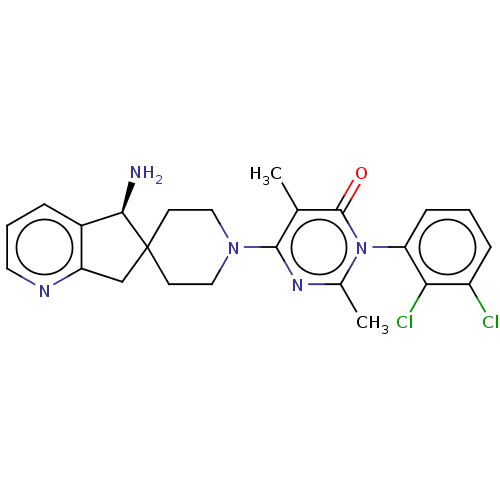
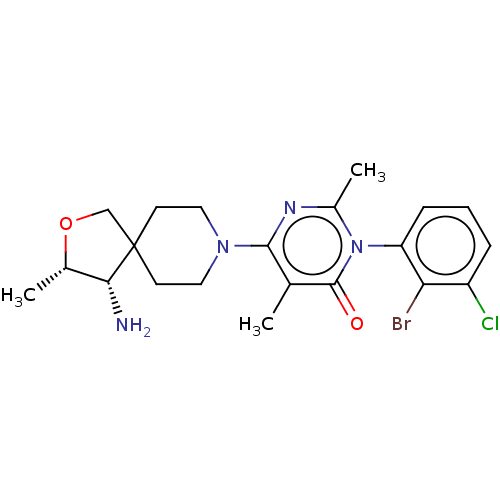
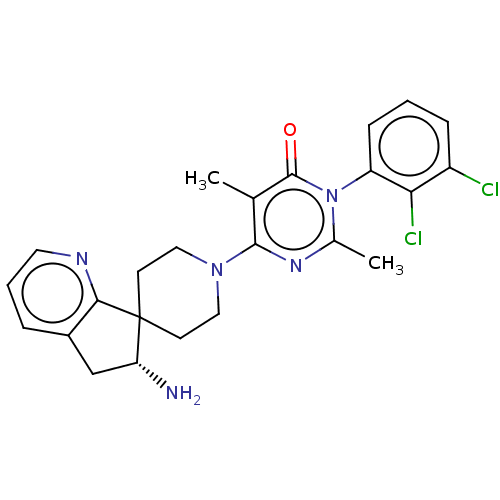
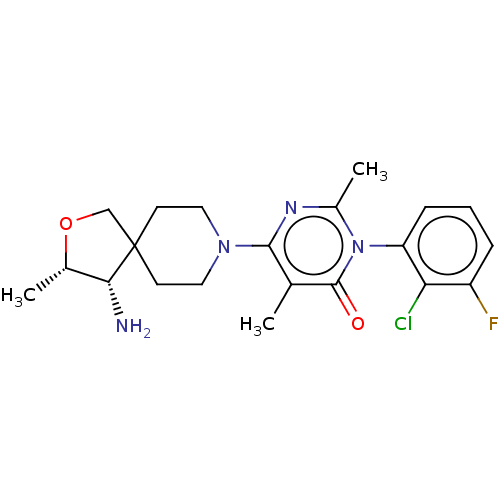
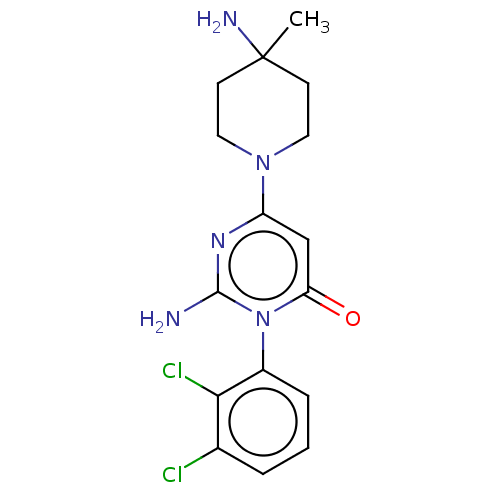
Report error Found 174 Enz. Inhib. hit(s) with all data for entry = 11348
Affinity DataIC50: 0.300nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 1.70nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 3.5nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 6.40nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 21nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 26nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 27nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 37nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 38nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 39nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 41nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 47nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 48nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 51nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 67nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 73nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 78nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 81nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 93nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 93nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 97nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 120nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
Affinity DataIC50: 180nMAssay Description:The inhibition of SHP2 by compounds of the invention was monitored using the surrogate substrate DiFMUP after protein activation by a peptide bearing...More data for this Ligand-Target Pair
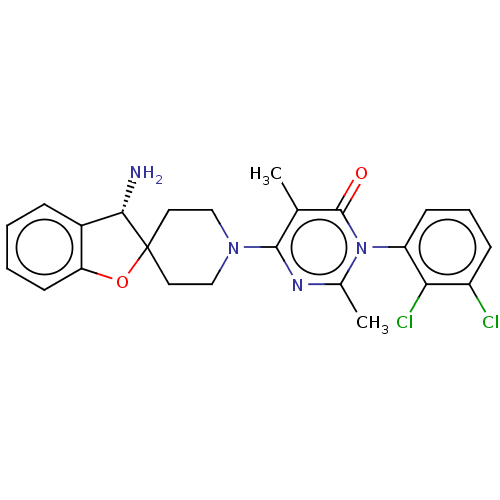
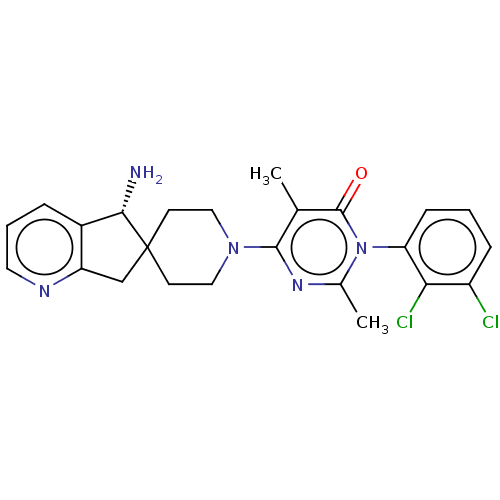

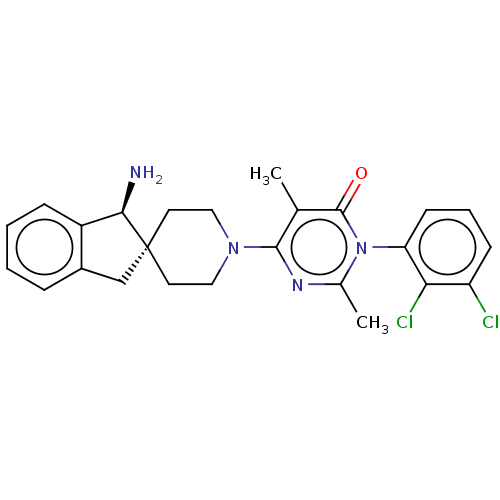
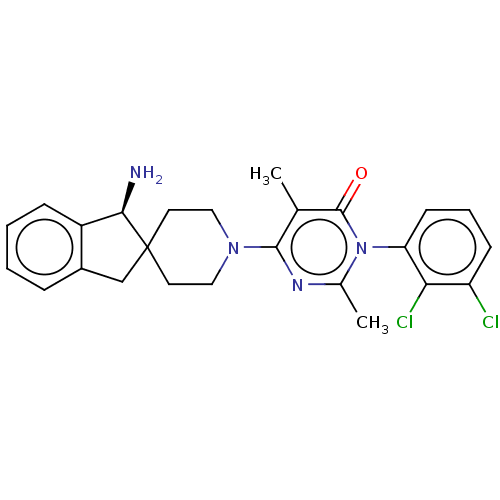

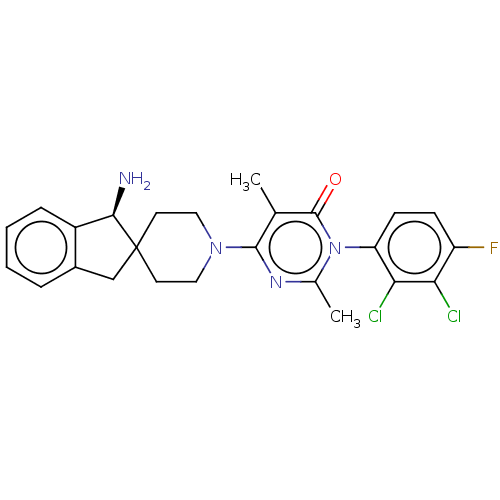



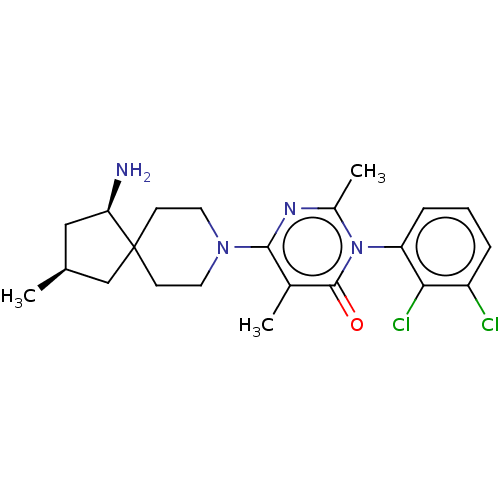

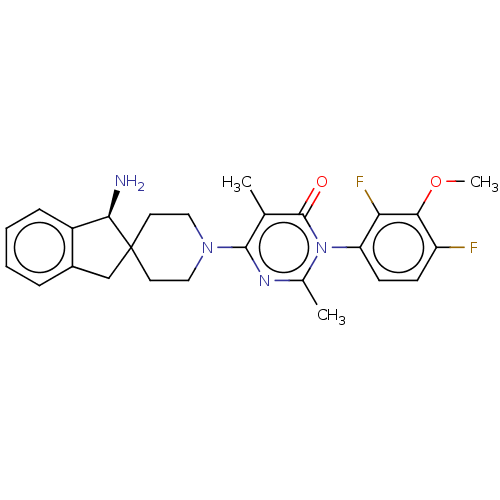


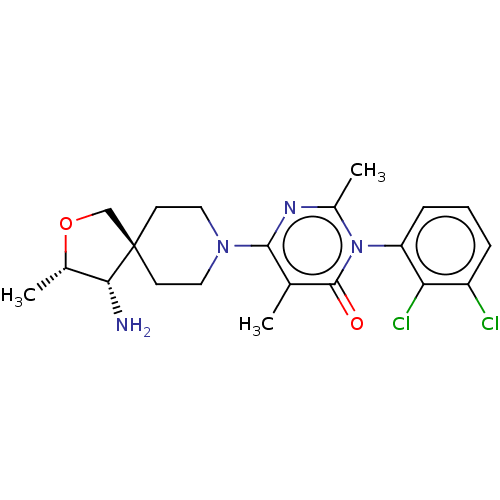



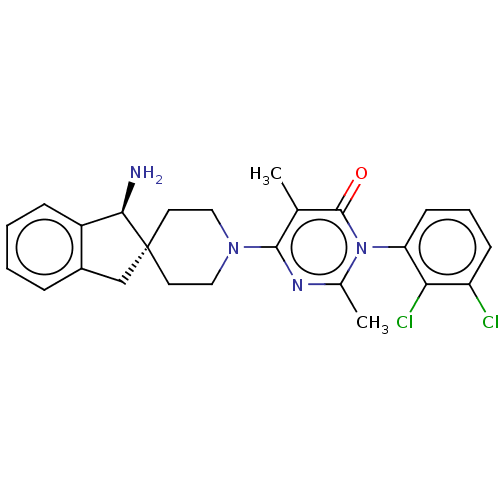
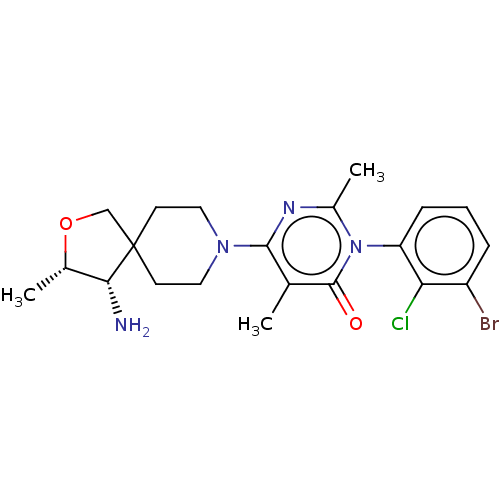


 3D Structure (crystal)
3D Structure (crystal)