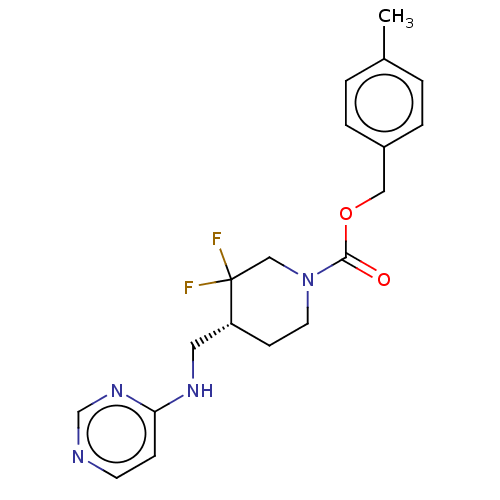
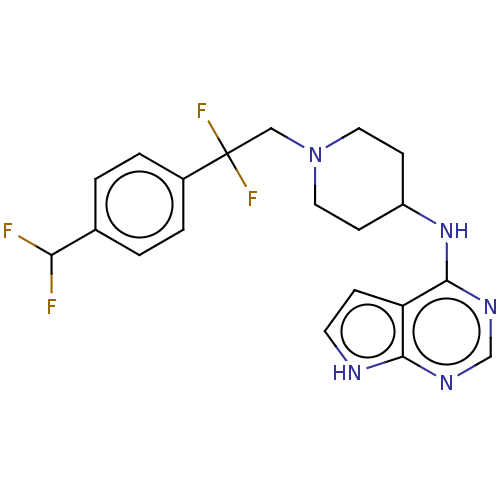
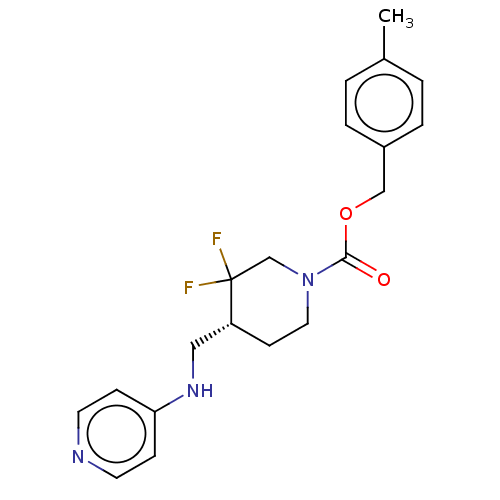
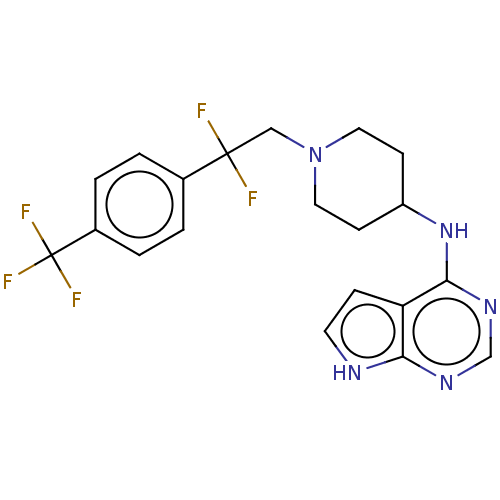
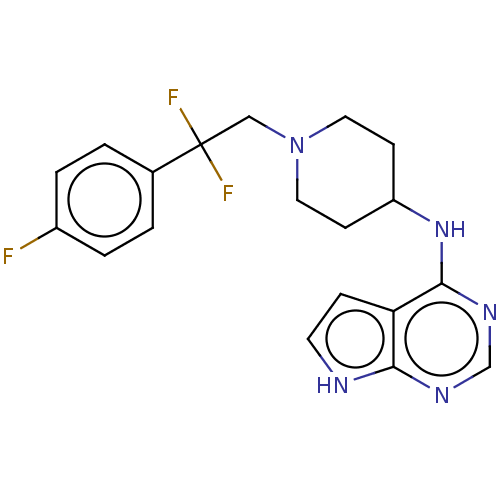
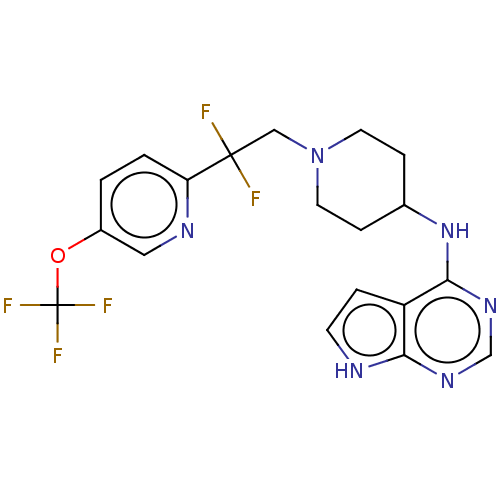
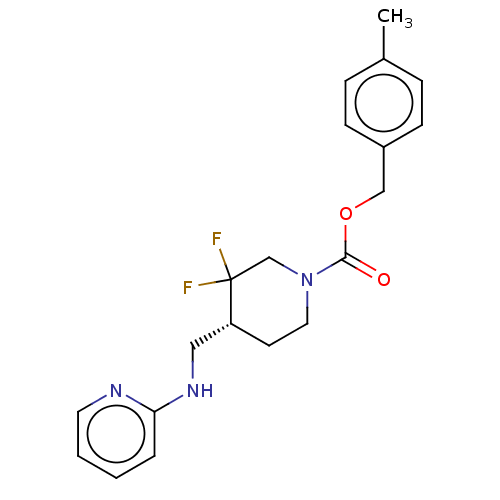
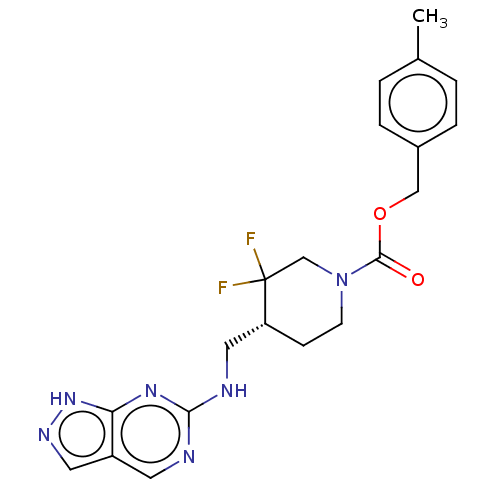
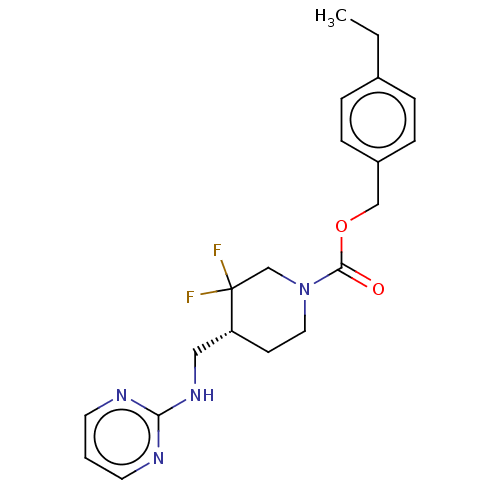
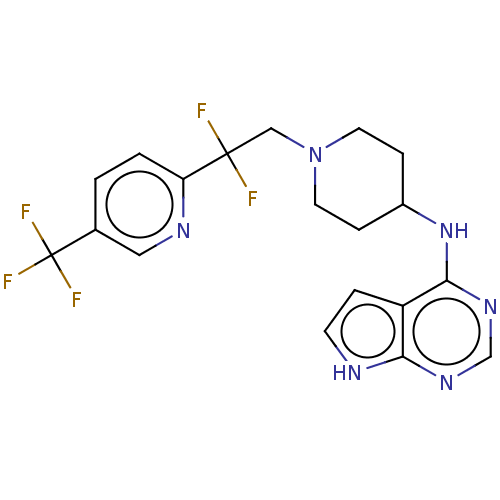
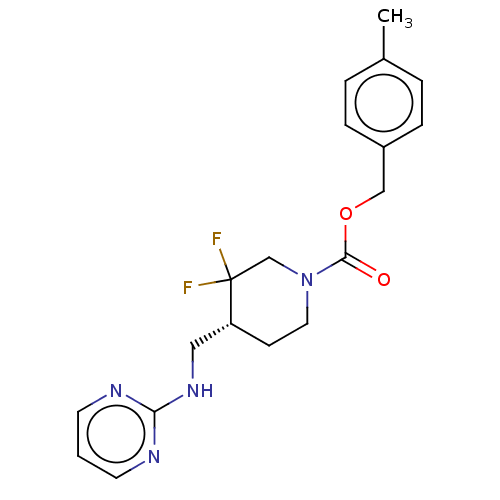
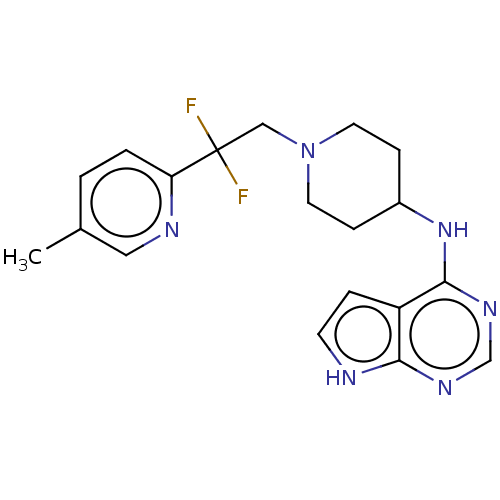
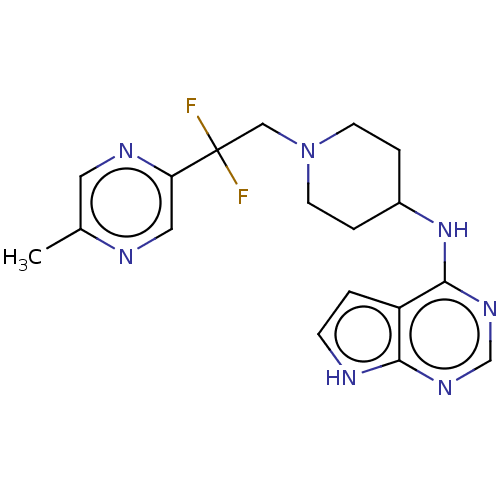
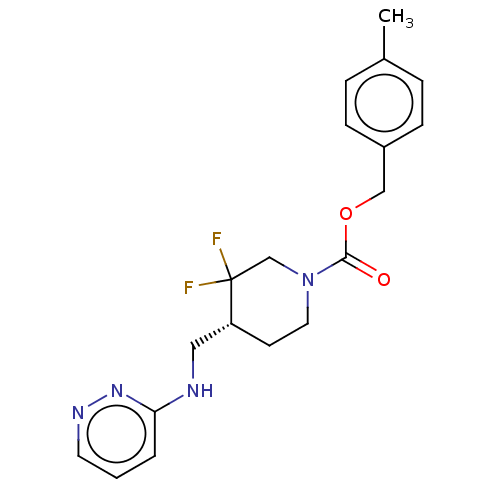
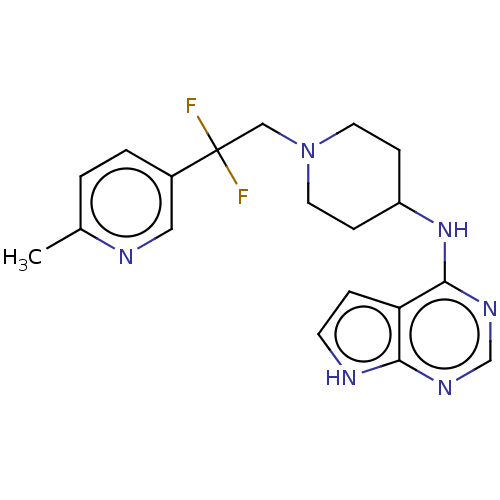
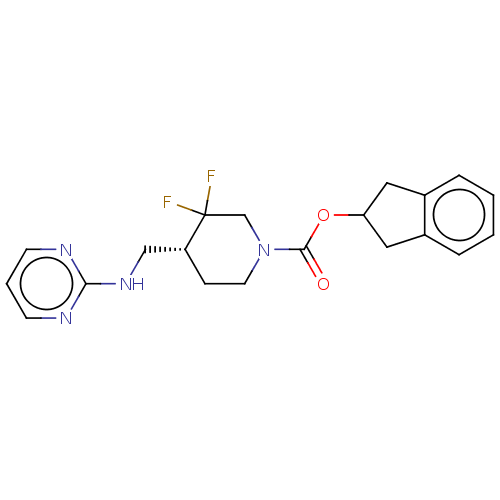
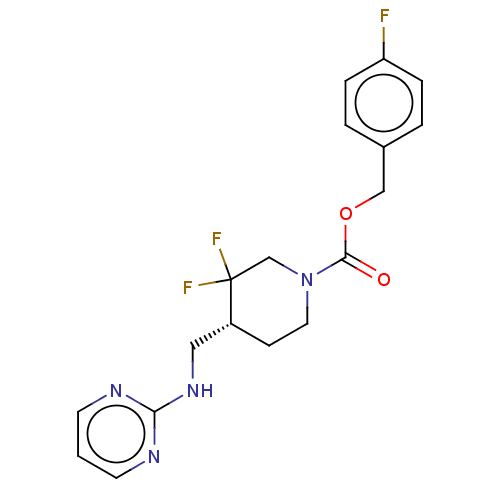
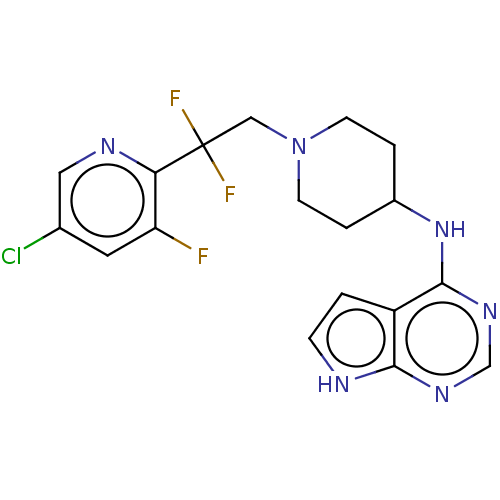
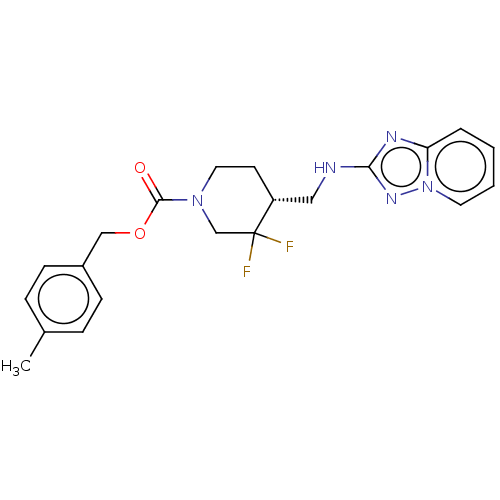
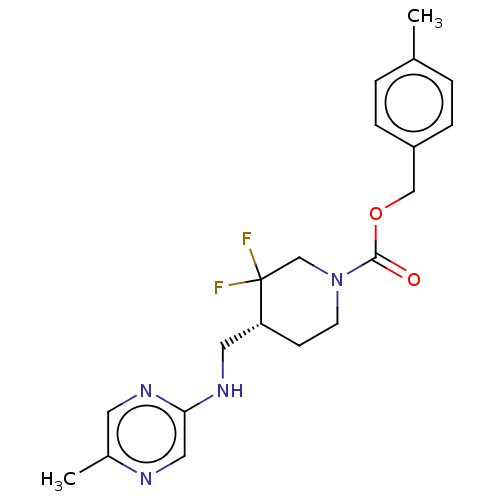
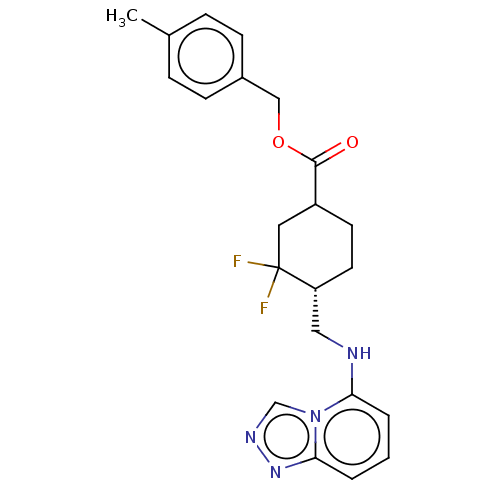
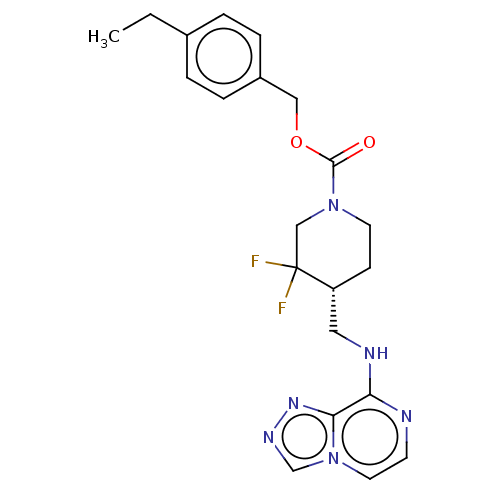
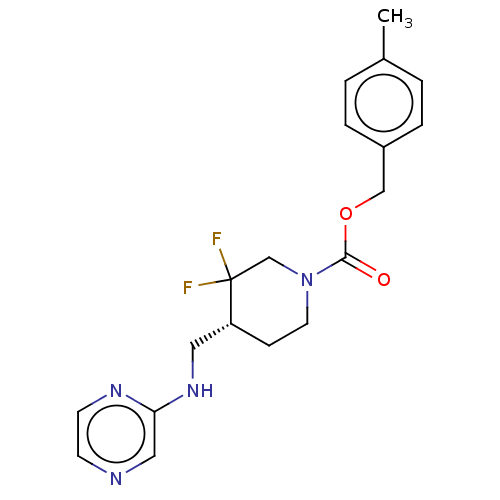
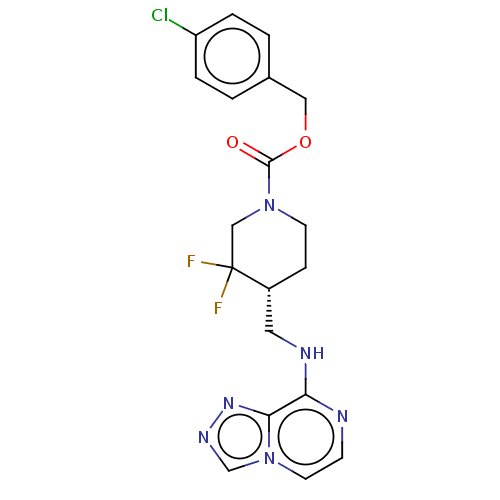
Report error Found 51 Enz. Inhib. hit(s) with all data for entry = 11449
Affinity DataIC50: 200nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 1.49E+3nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30E+3nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.90E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 3.80E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 4.50E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 4.90E+3nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 5.60E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 5.90E+3nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 6.00E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 6.70E+3nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 6.80E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 6.90E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 7.00E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 7.30E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 9.20E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 9.50E+3nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 9.70E+3nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+4nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.14E+4nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.38E+4nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+4nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.70E+4nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80E+4nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 2.80E+4nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 2.90E+4nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 3.30E+4nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 3.50E+4nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 3.90E+4nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 4.00E+4nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 4.10E+4nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibitory activities of test compounds on 5 major isoforms of CYP P450 were evaluated by using pooled human liver microsome (HLM, purchased from BD ...More data for this Ligand-Target Pair
Affinity DataIC50: 6.00E+4nMAssay Description:The assay was performed on hERG channel stably expressed in HEK293 cells. The cells were cultured at 37° C. in a humidified CO2 incubator in the grow...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+7nMAssay Description:This example describes an NR2B receptor binding assay in rat brain using [3H] (E)-N1-(2-methoxybenzyl)-cinnamidine (see below). The binding assay met...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+7nMAssay Description:This example describes an NR2B receptor binding assay in rat brain using [3H] (E)-N1-(2-methoxybenzyl)-cinnamidine (see below). The binding assay met...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+7nMAssay Description:This example describes an NR2B receptor binding assay in rat brain using [3H] (E)-N1-(2-methoxybenzyl)-cinnamidine (see below). The binding assay met...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+7nMAssay Description:This example describes an NR2B receptor binding assay in rat brain using [3H] (E)-N1-(2-methoxybenzyl)-cinnamidine (see below). The binding assay met...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+7nMAssay Description:This example describes an NR2B receptor binding assay in rat brain using [3H] (E)-N1-(2-methoxybenzyl)-cinnamidine (see below). The binding assay met...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+7nMAssay Description:This example describes an NR2B receptor binding assay in rat brain using [3H] (E)-N1-(2-methoxybenzyl)-cinnamidine (see below). The binding assay met...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+7nMAssay Description:This example describes an NR2B receptor binding assay in rat brain using [3H] (E)-N1-(2-methoxybenzyl)-cinnamidine (see below). The binding assay met...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+7nMAssay Description:This example describes an NR2B receptor binding assay in rat brain using [3H] (E)-N1-(2-methoxybenzyl)-cinnamidine (see below). The binding assay met...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+7nMAssay Description:This example describes an NR2B receptor binding assay in rat brain using [3H] (E)-N1-(2-methoxybenzyl)-cinnamidine (see below). The binding assay met...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+7nMAssay Description:This example describes an NR2B receptor binding assay in rat brain using [3H] (E)-N1-(2-methoxybenzyl)-cinnamidine (see below). The binding assay met...More data for this Ligand-Target Pair