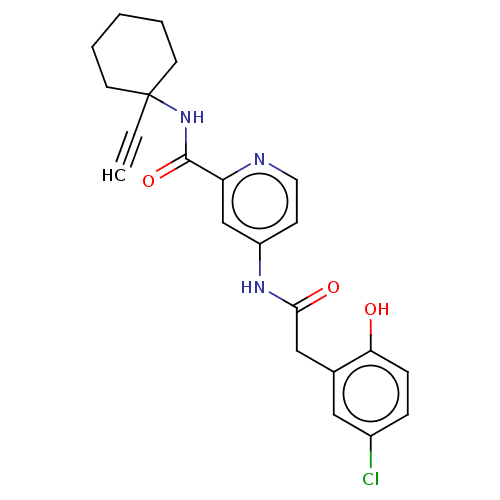
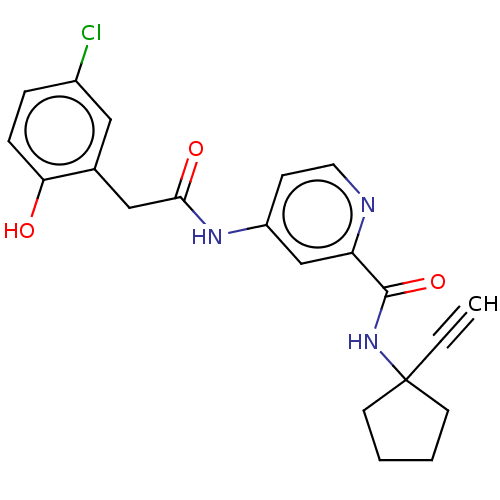
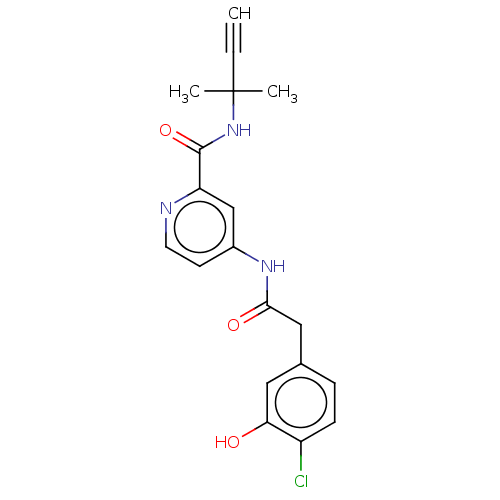
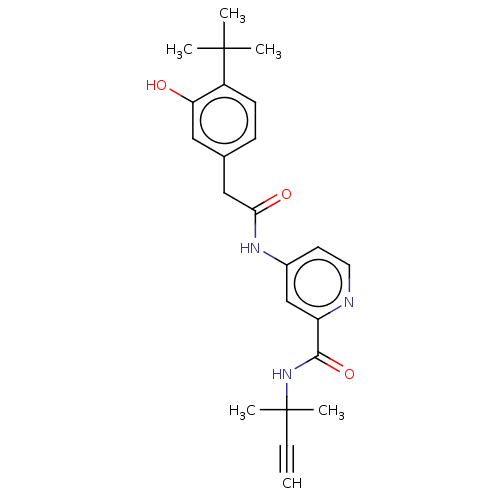
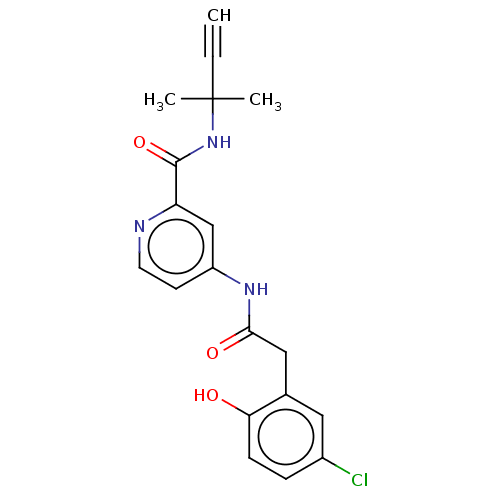
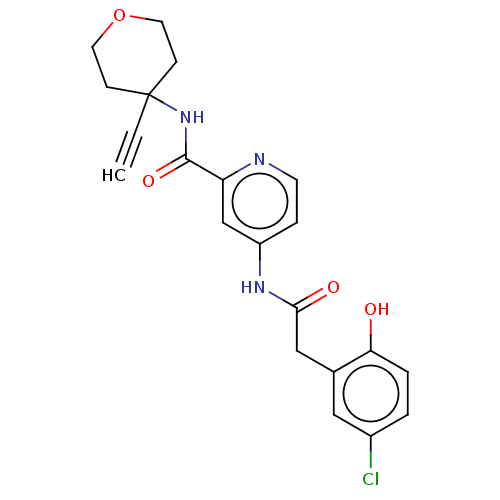
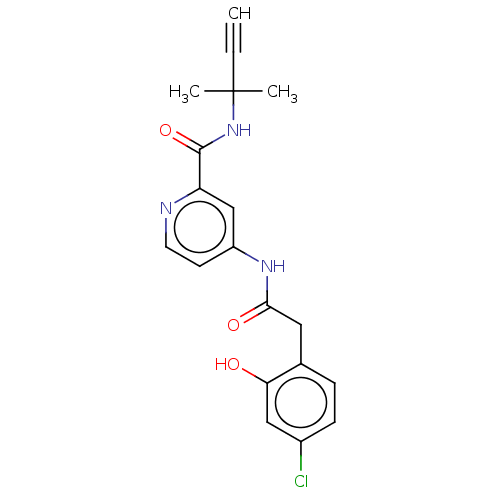
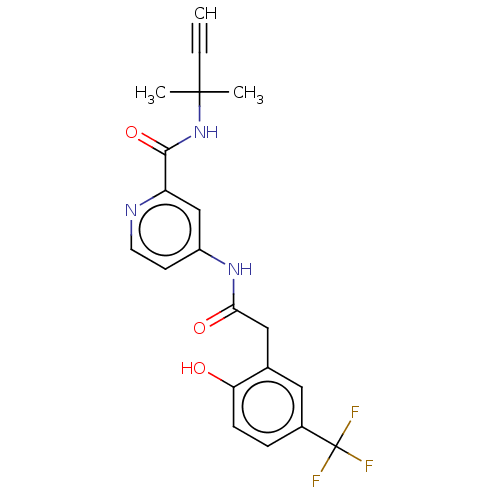
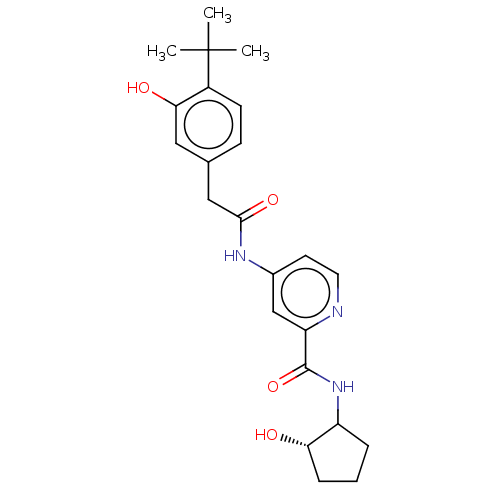
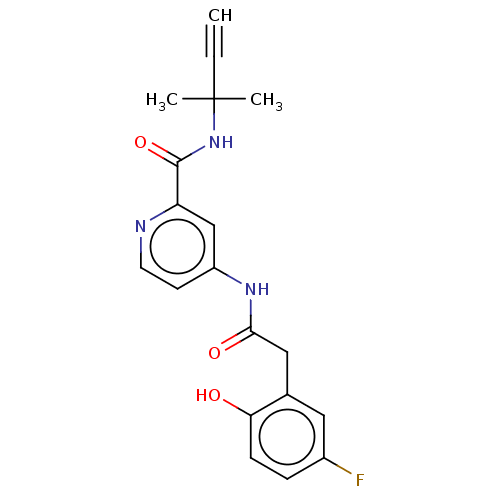
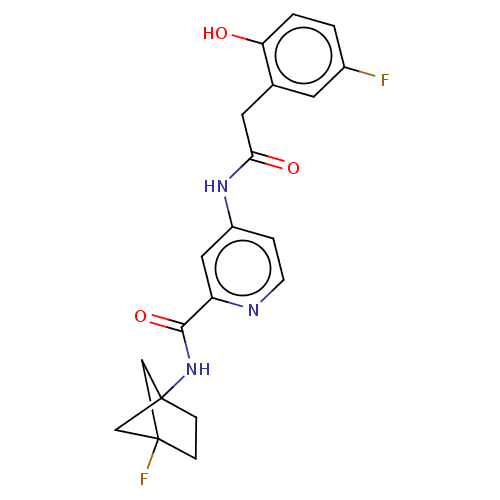
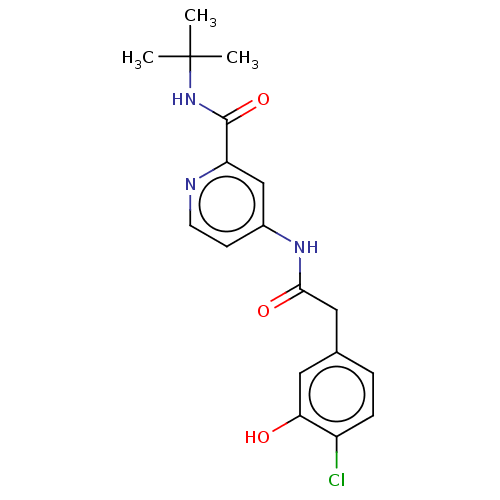
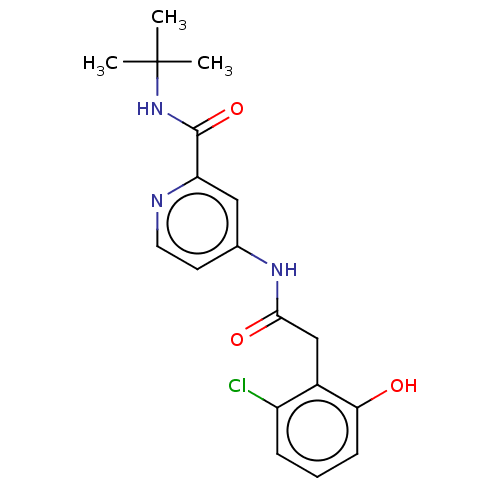
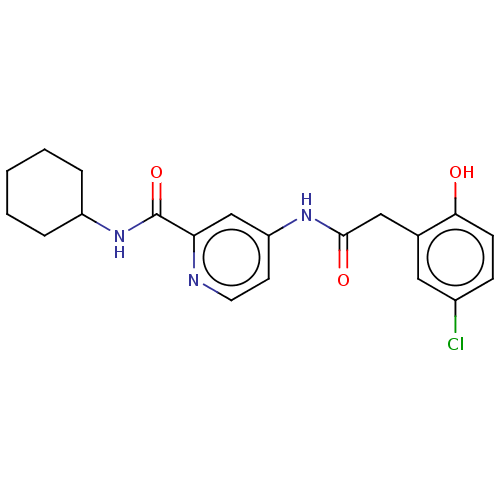
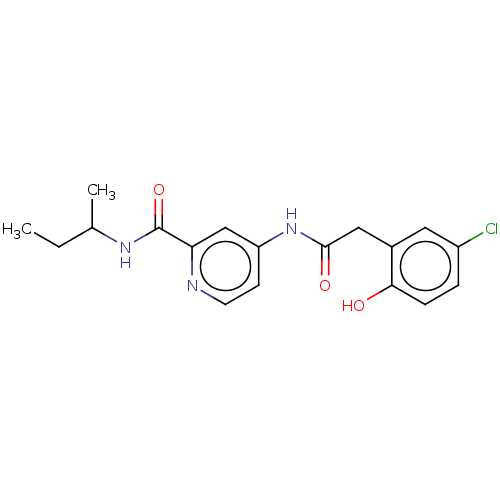
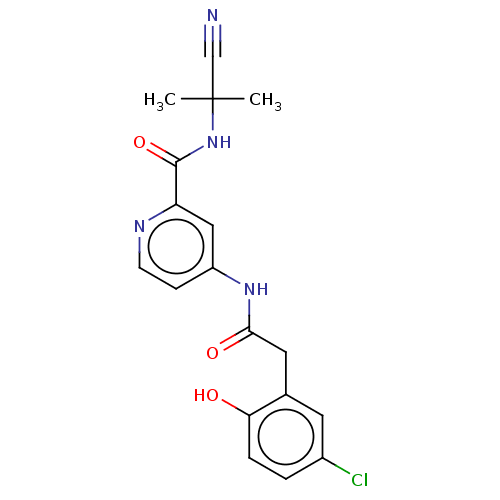
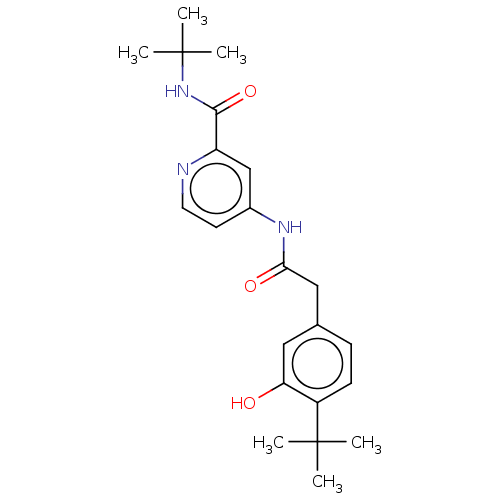
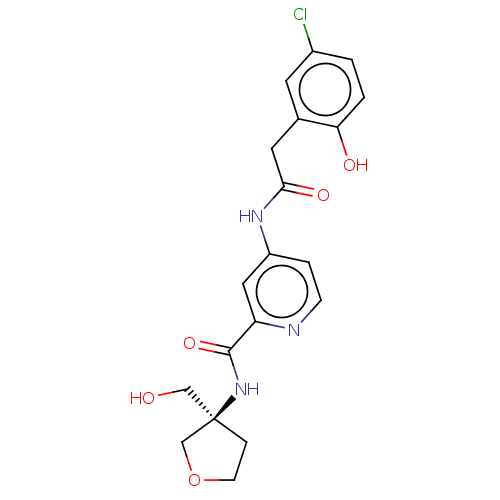
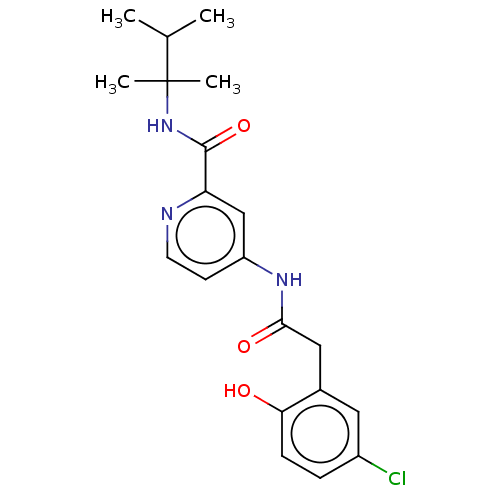
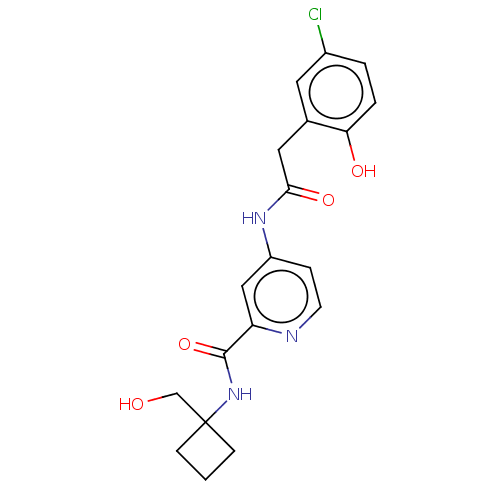
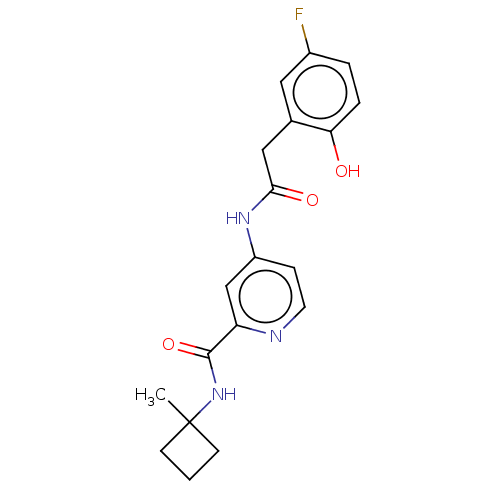
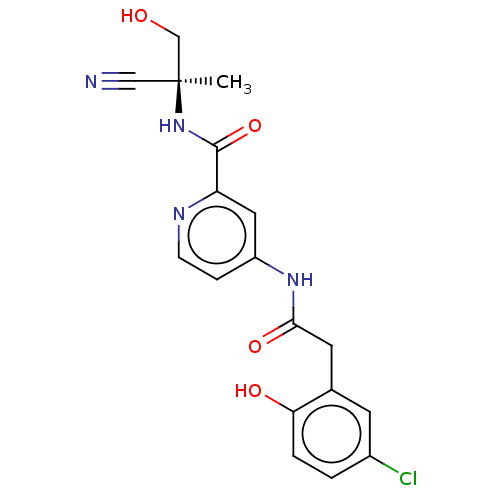
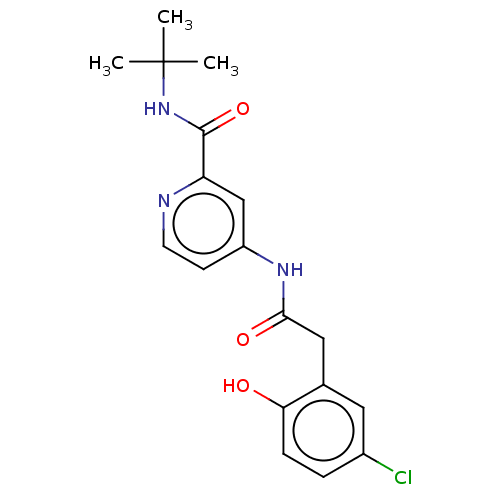
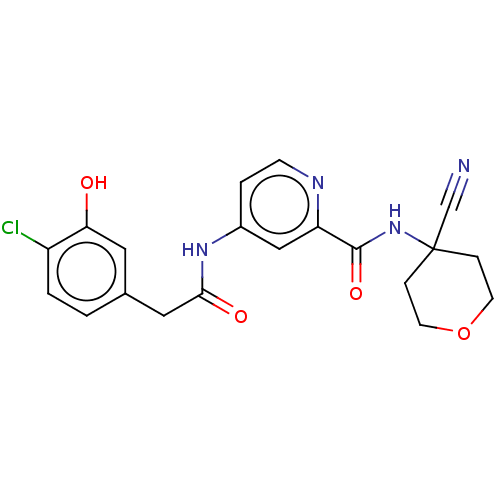
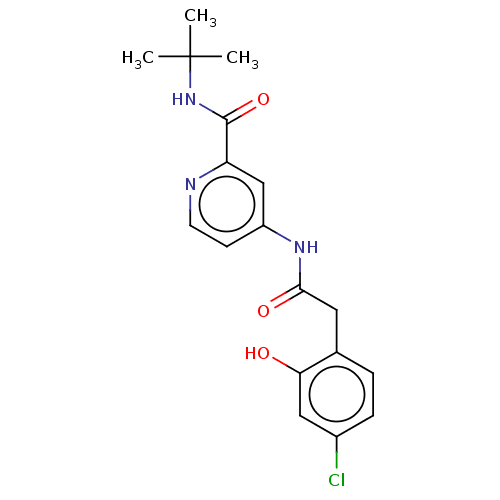
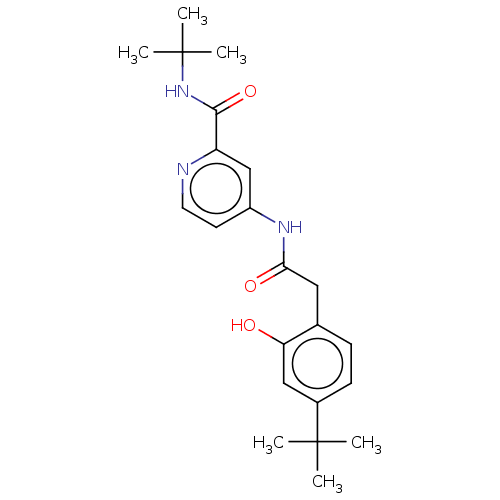
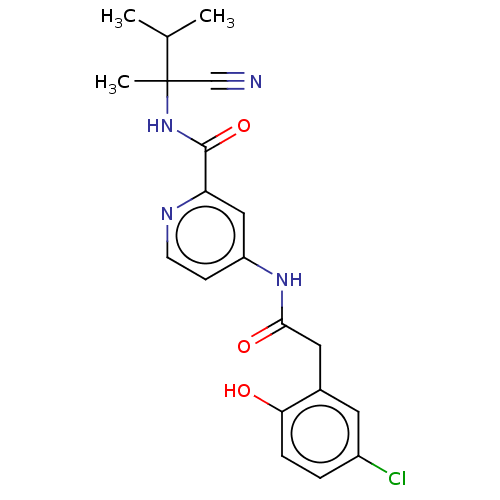
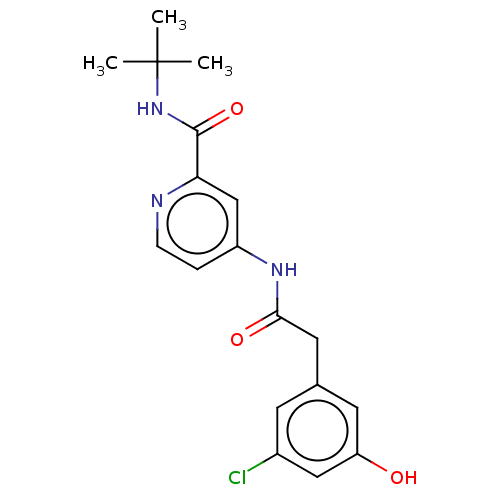
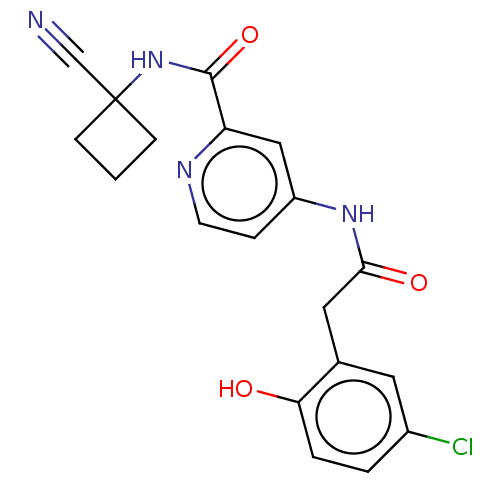
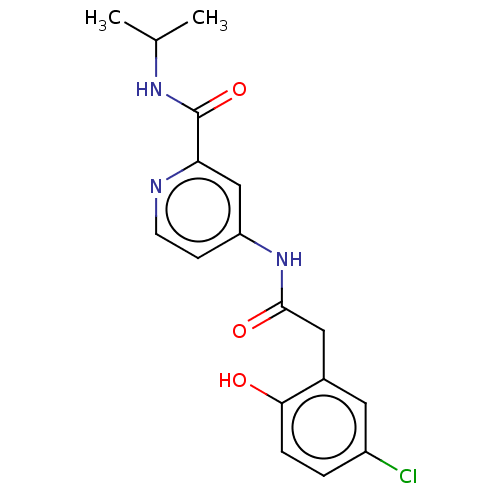
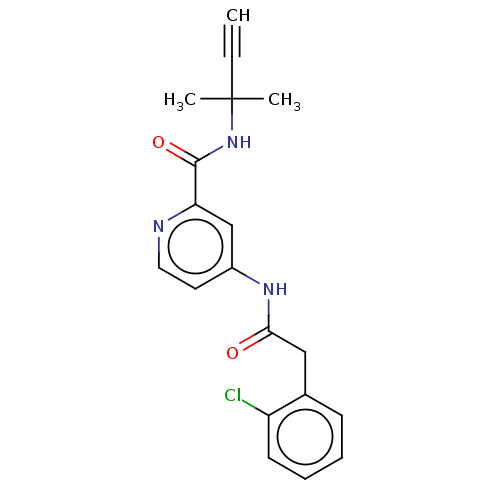
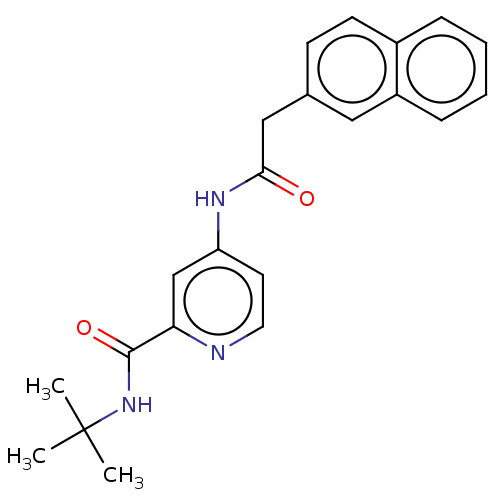
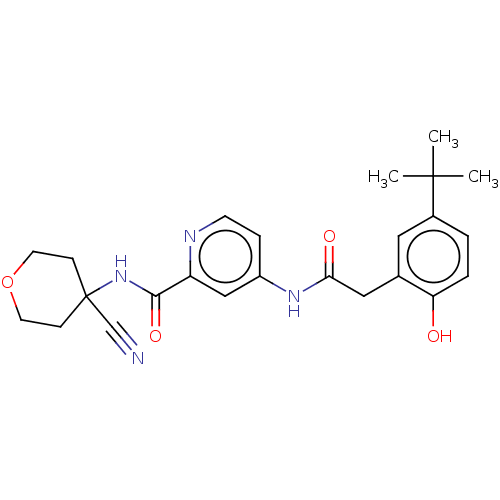
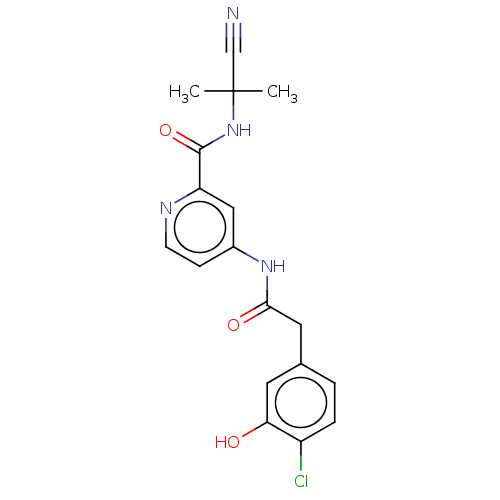
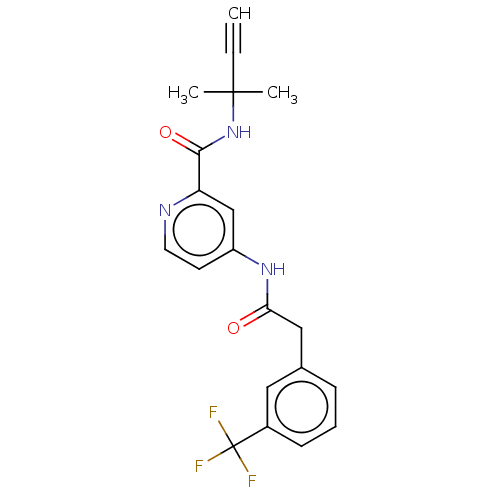
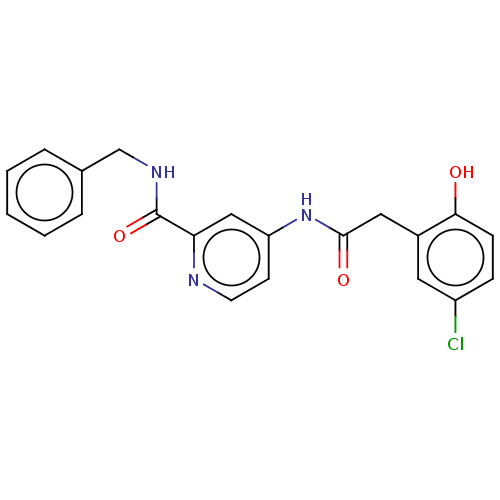
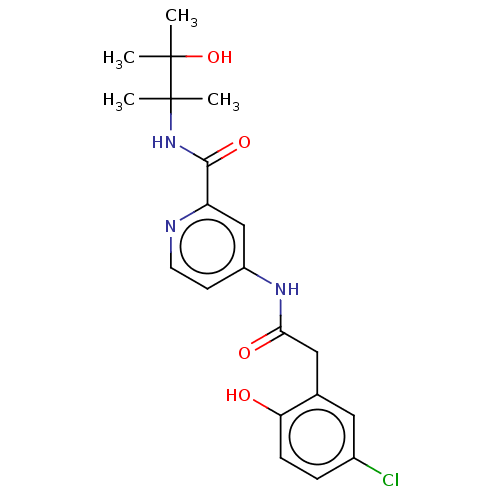
Report error Found 233 Enz. Inhib. hit(s) with all data for entry = 10032
Affinity DataEC50: 10nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 10nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 10nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 20nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 20nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 20nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 20nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 20nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 40nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 40nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 40nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 40nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 50nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 50nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 50nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 50nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 50nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 60nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 60nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 60nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 60nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 70nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 70nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 80nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 90nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 90nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 90nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 100nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 100nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 100nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 100nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 110nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 110nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 110nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 120nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 120nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 120nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 120nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 120nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 130nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 130nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 130nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 140nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 140nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 140nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 140nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 140nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 150nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 150nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair
Affinity DataEC50: 150nMAssay Description:Compound activity was quantified by measuring the increase in current upon compound addition and expressing this as a percentage increase of baseline...More data for this Ligand-Target Pair