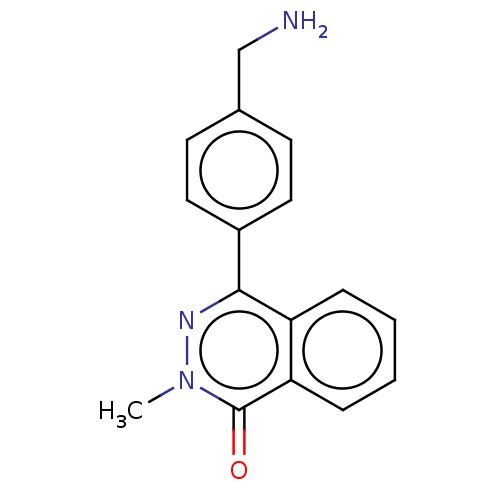
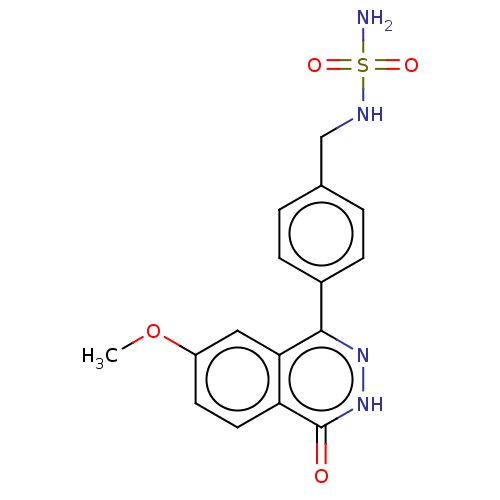
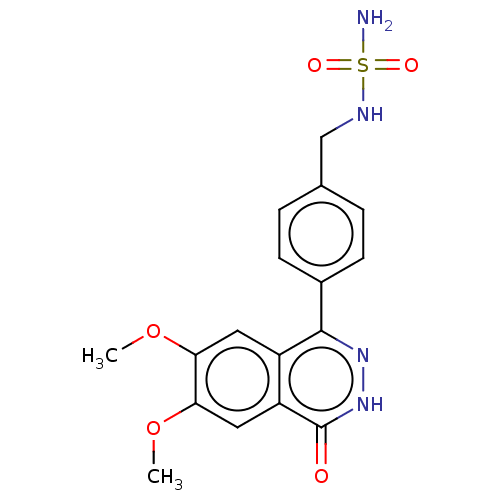
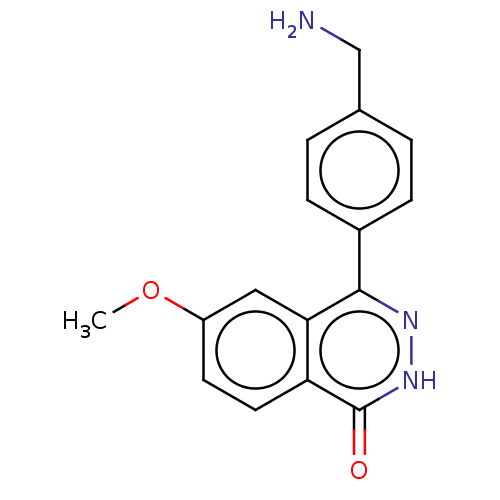
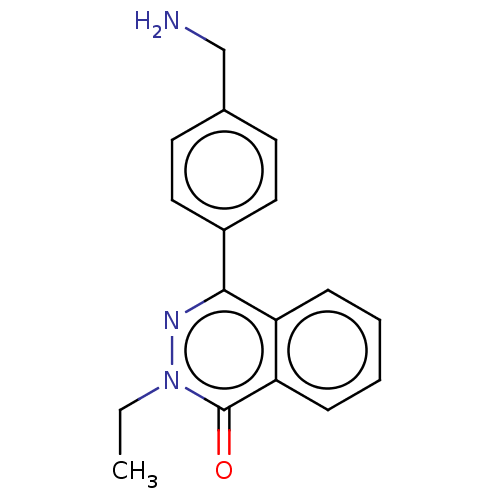
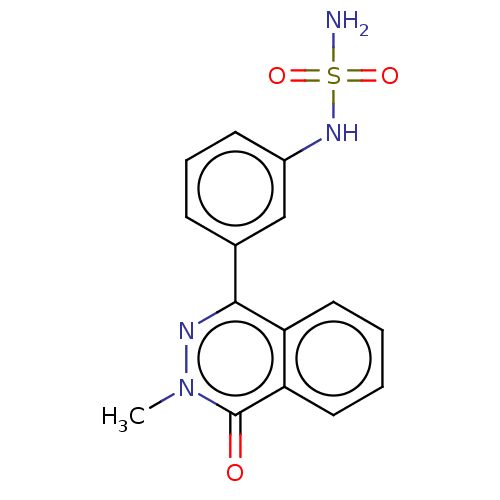
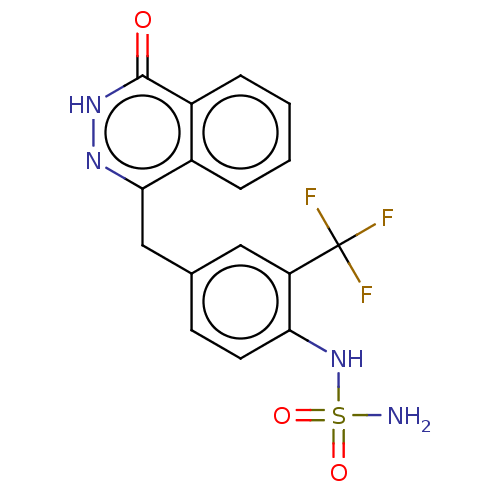
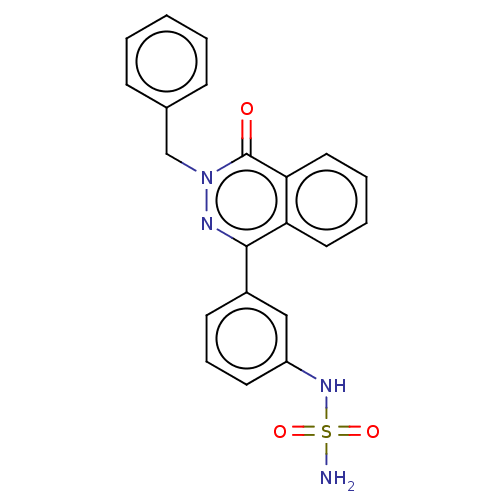
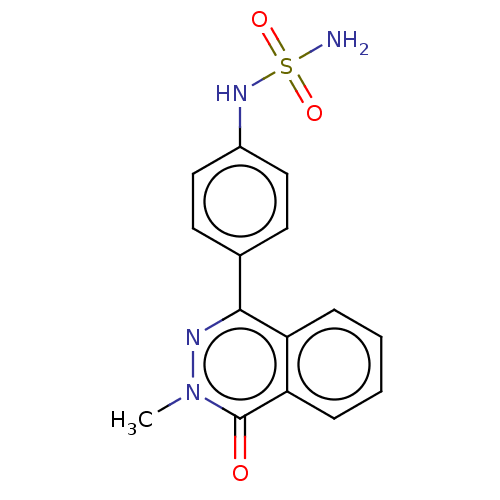
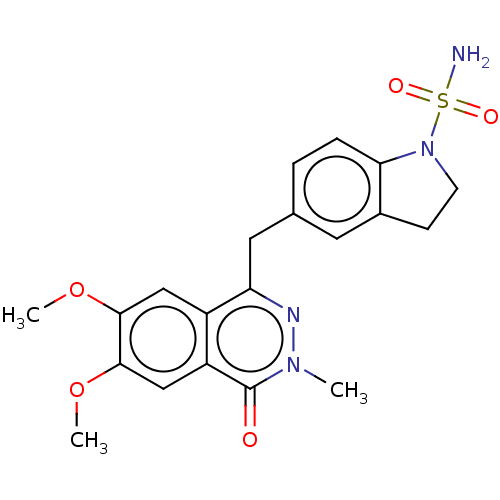
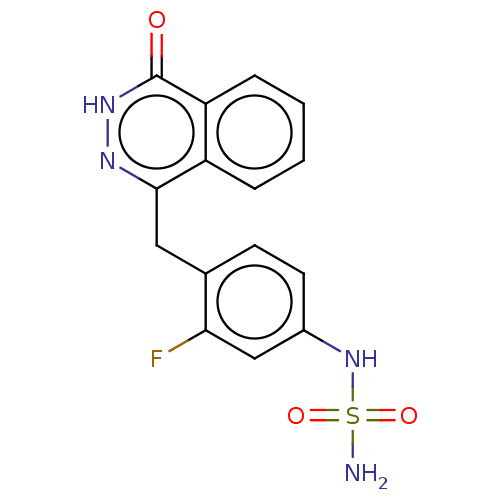
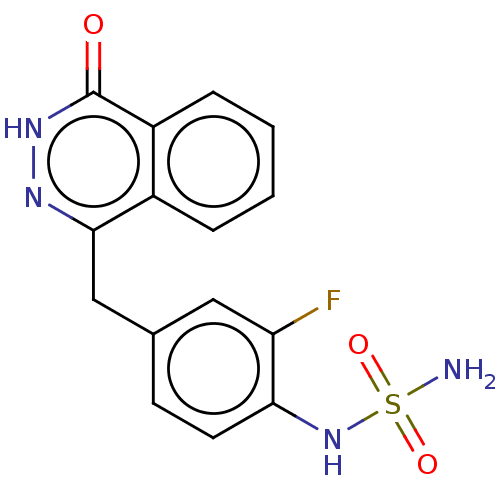
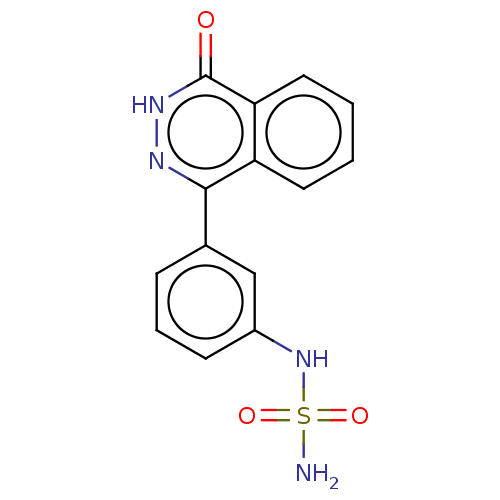
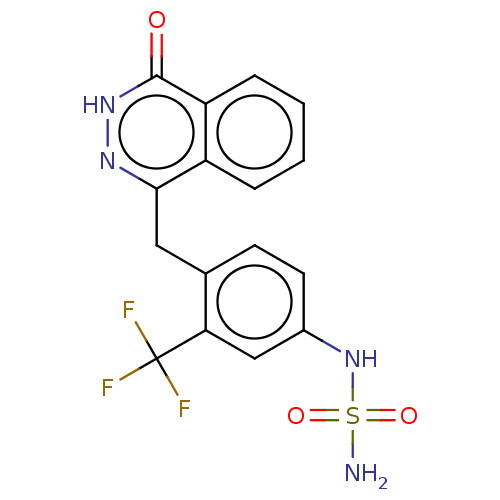
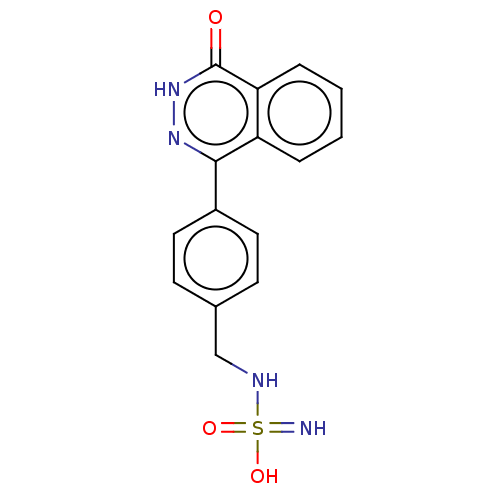
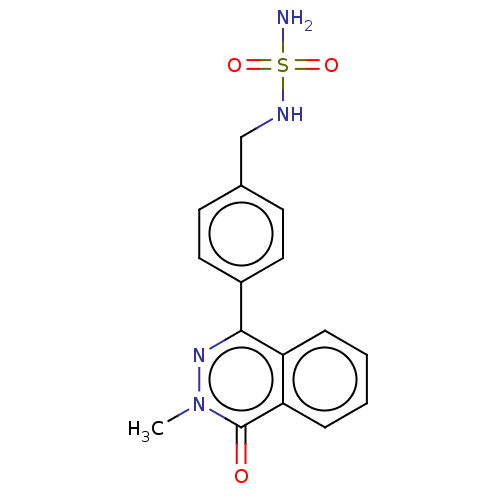
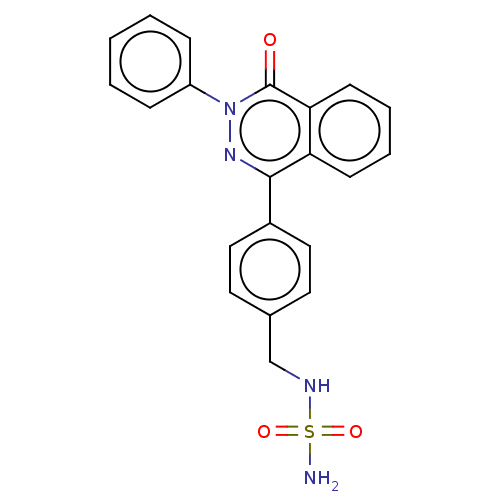
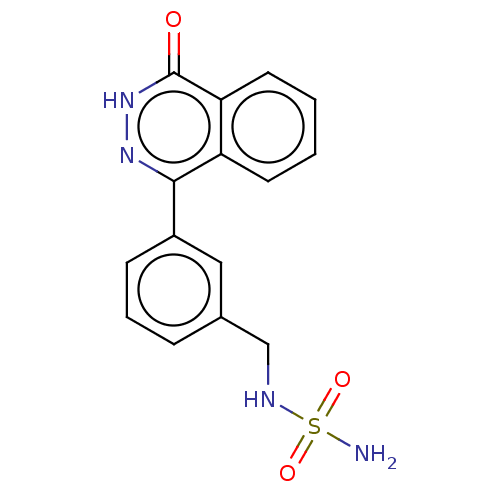
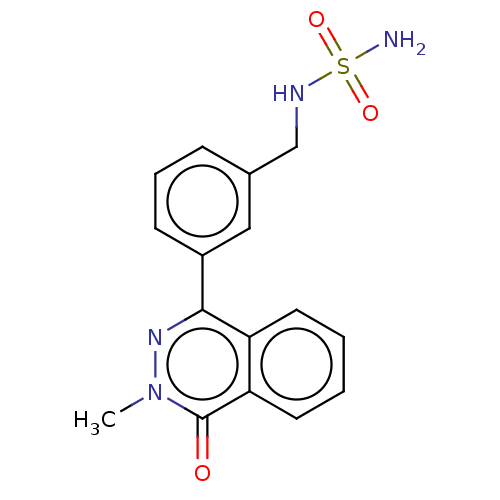
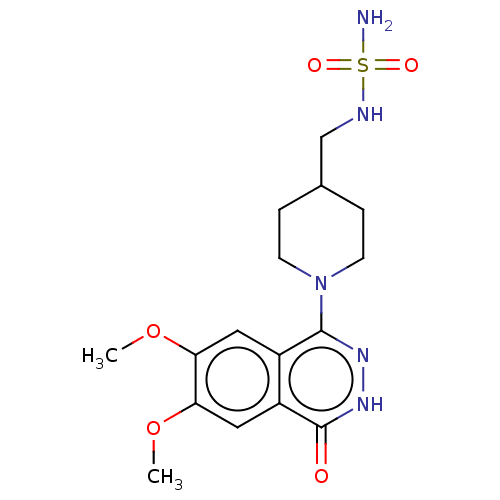
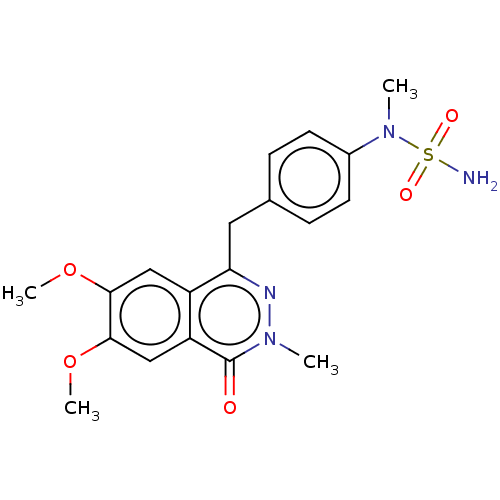
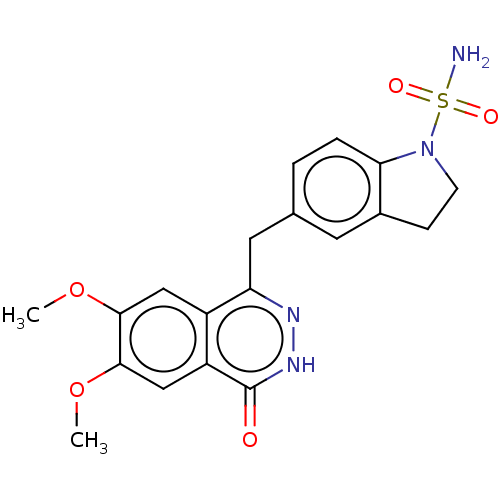
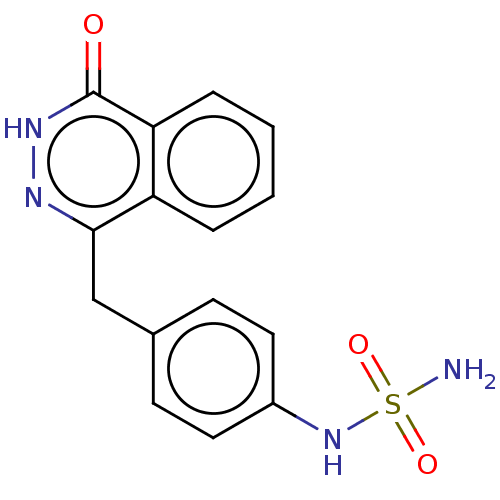
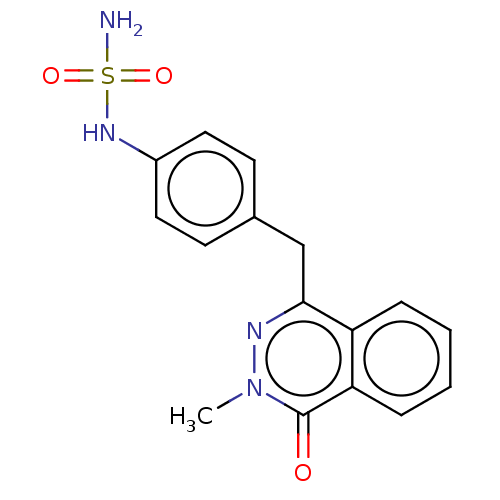
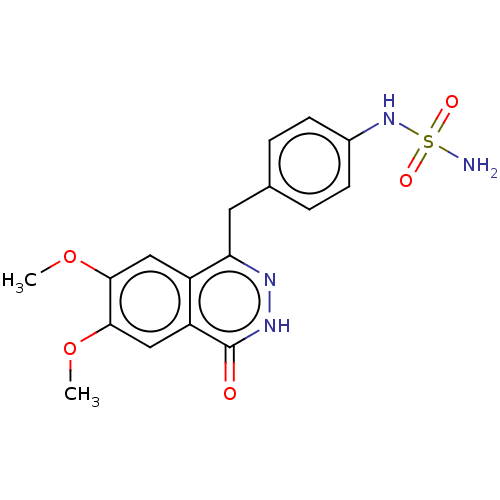
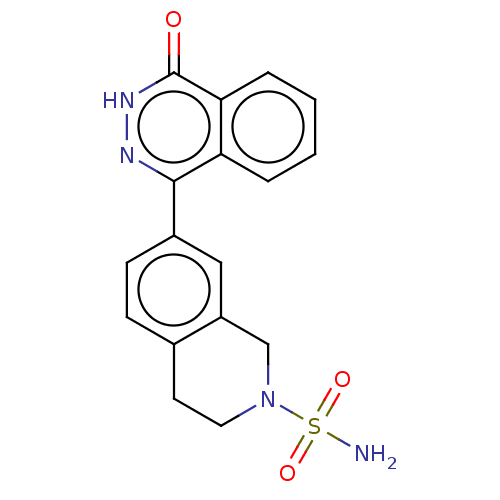
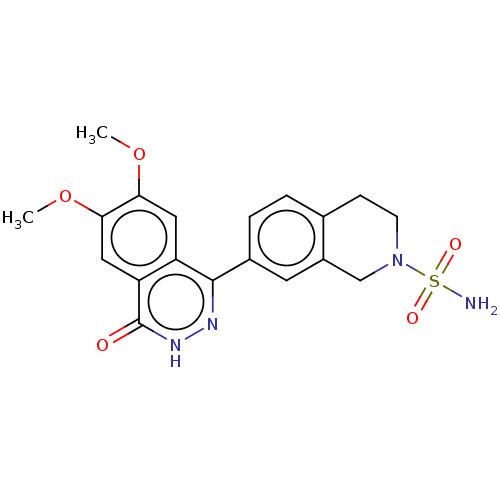
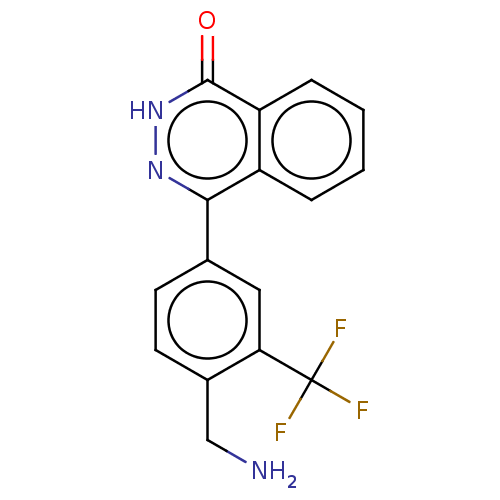
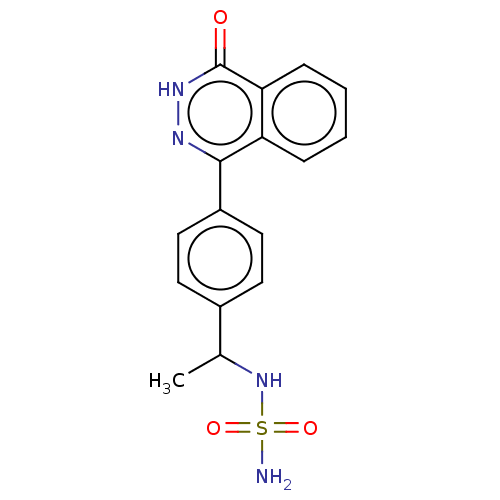
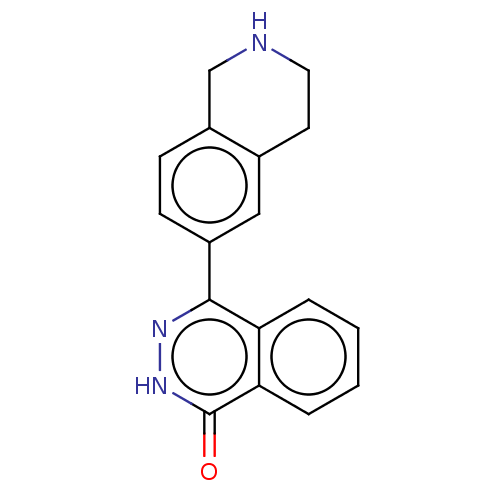
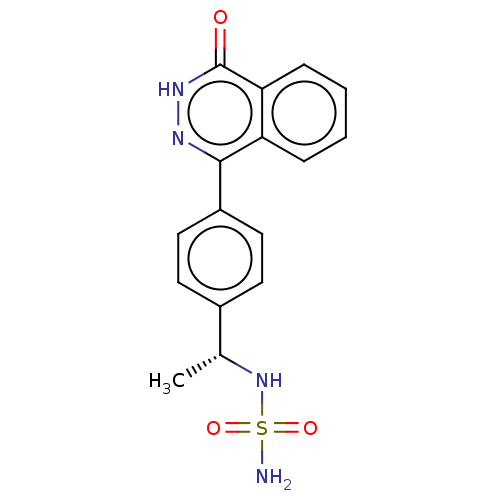
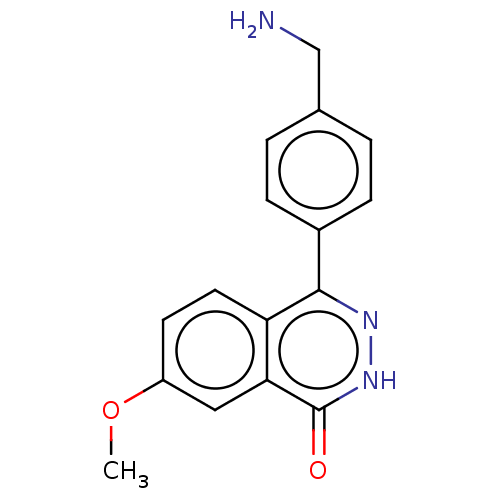
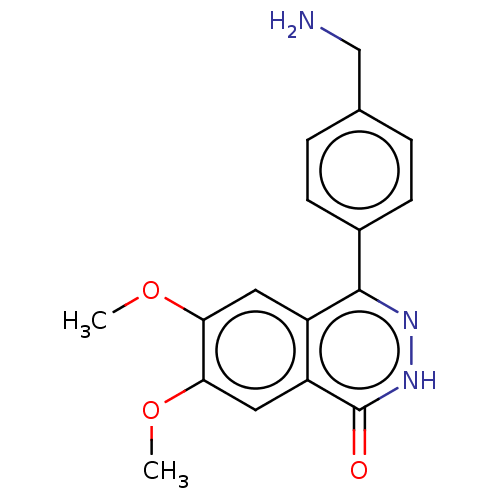
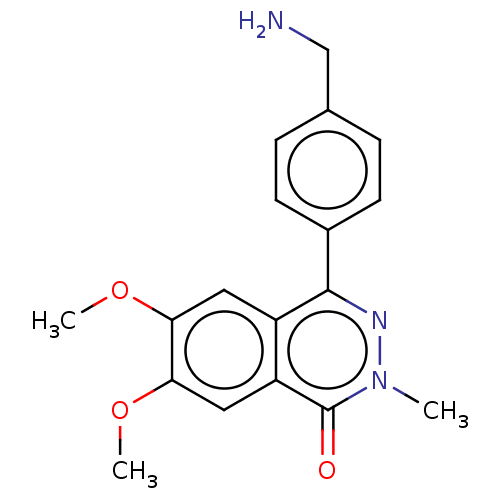
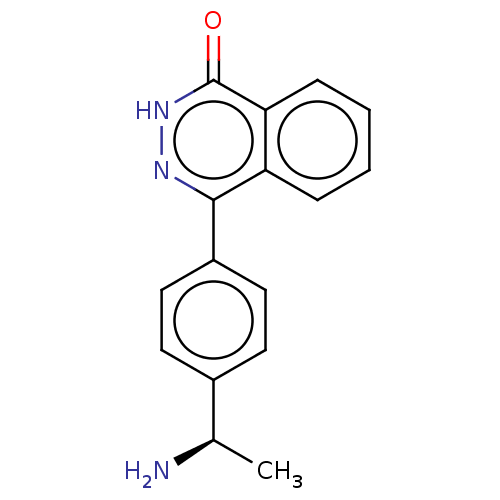
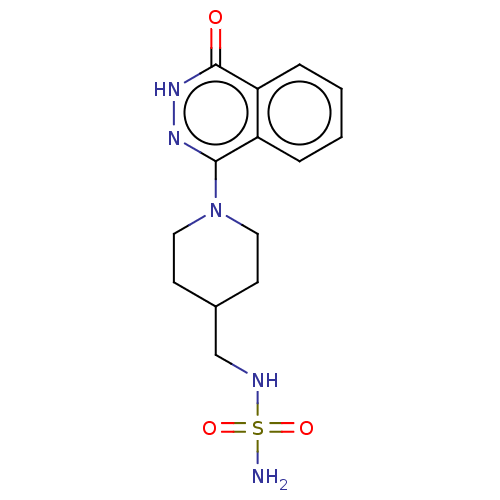
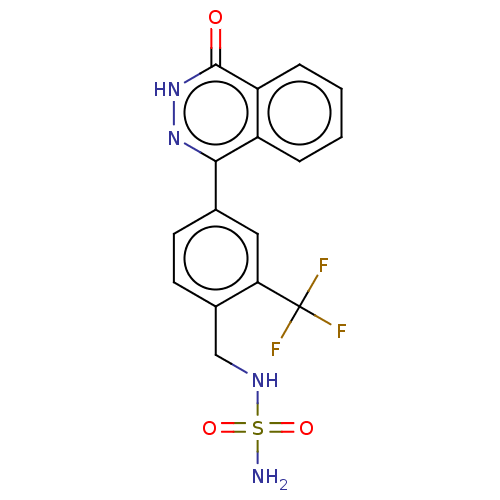
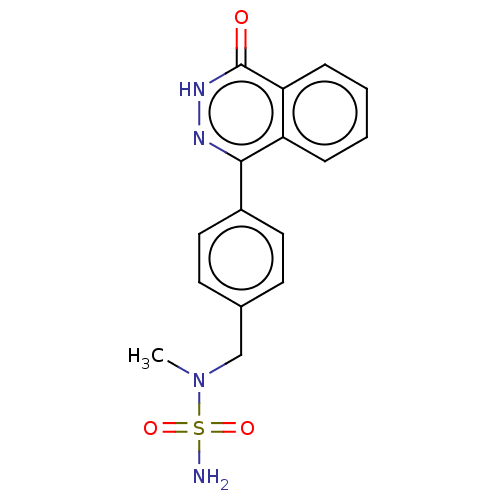
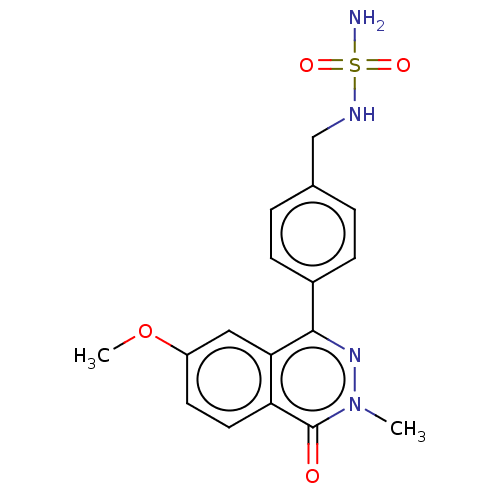
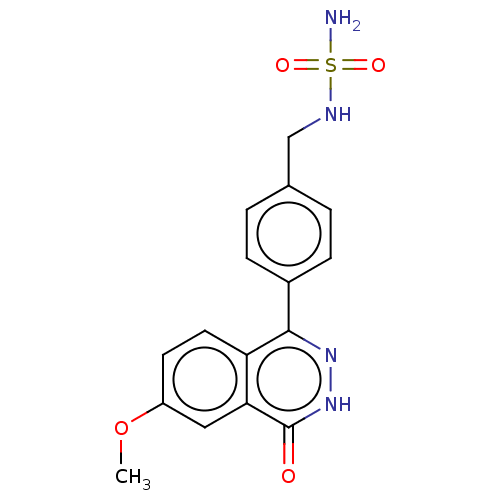
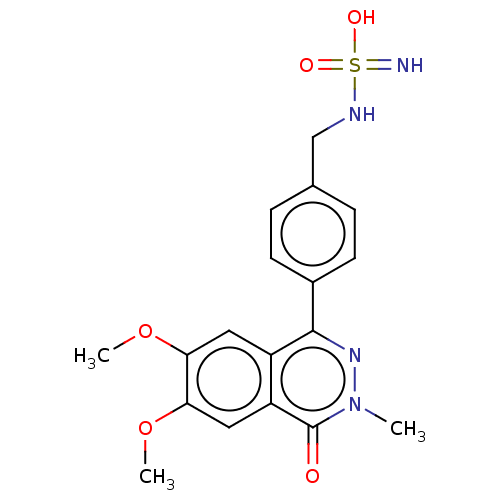
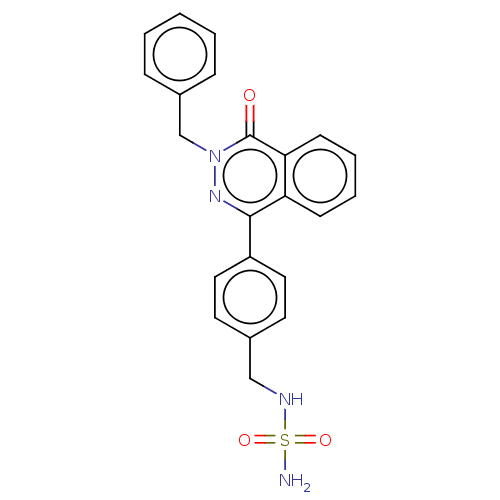
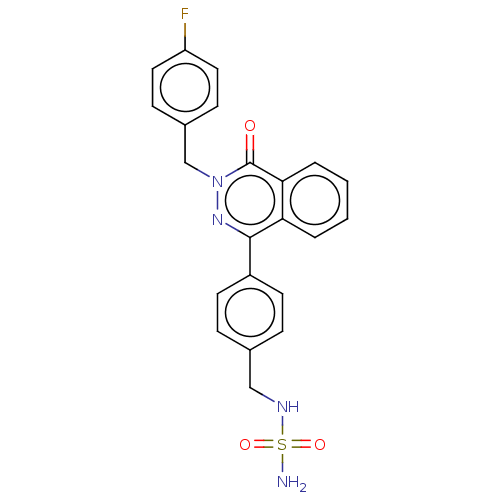
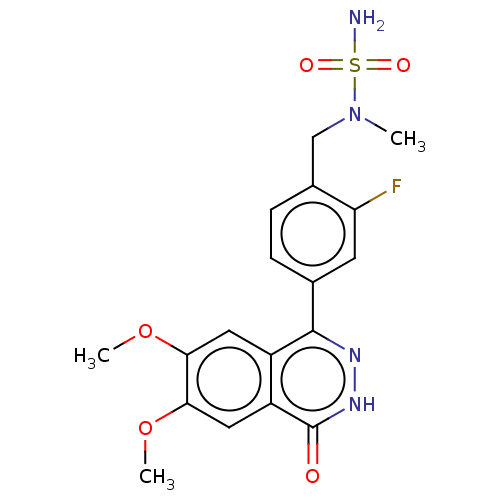
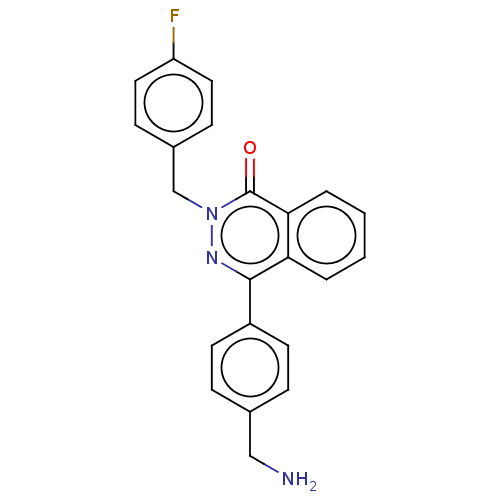
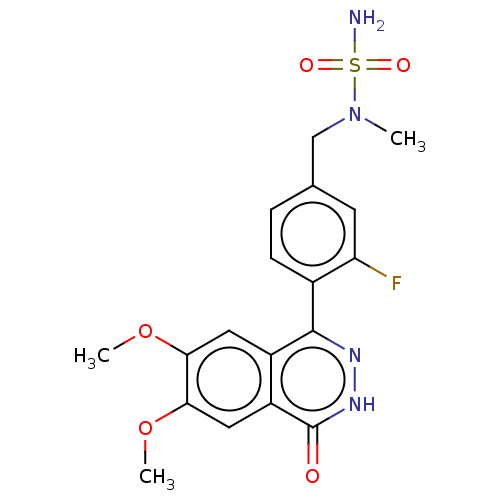
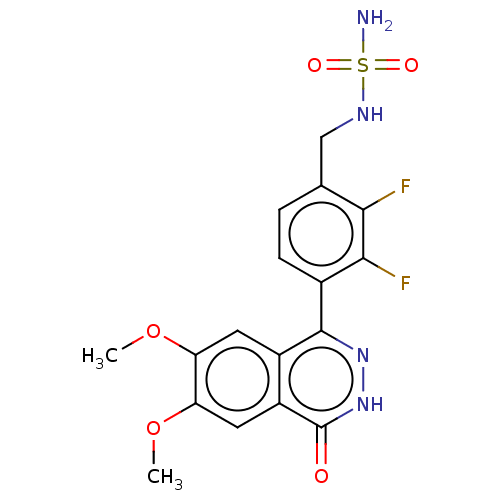
Report error Found 127 Enz. Inhib. hit(s) with all data for entry = 6468
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 1.00E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 5.50E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 5.50E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 5.50E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 5.50E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 5.50E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 5.50E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 5.50E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 5.50E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 1(Human)
Txinno Bioscience
US Patent
Txinno Bioscience
US Patent
Affinity DataIC50: 5.50E+3nMAssay Description:ENPP1 hydrolyzes nucleotides or nucleotide derivatives to produce nucleoside -5′-monophosphate and pyrophosphate. in addition, ENPP1 hydrolyzes...More data for this Ligand-Target Pair