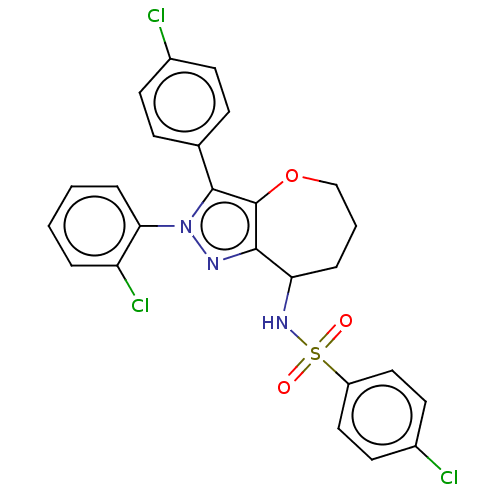
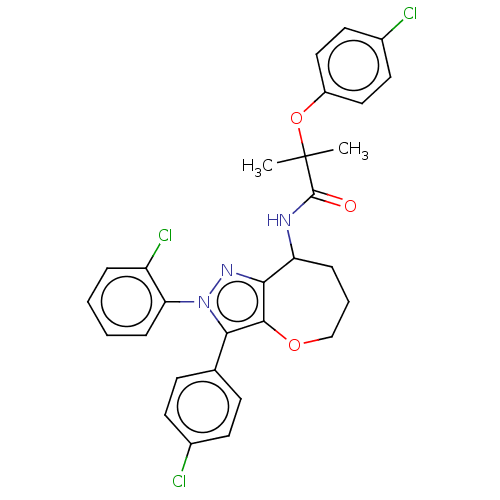
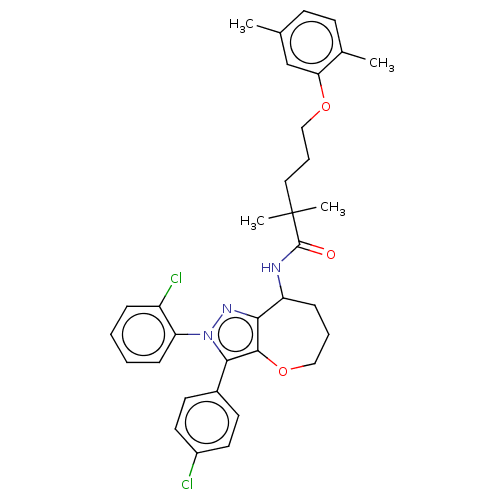
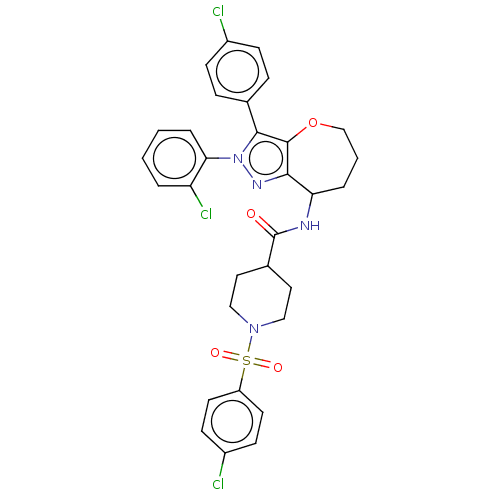
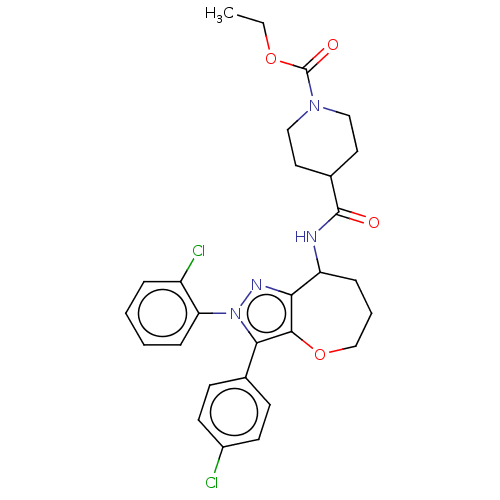
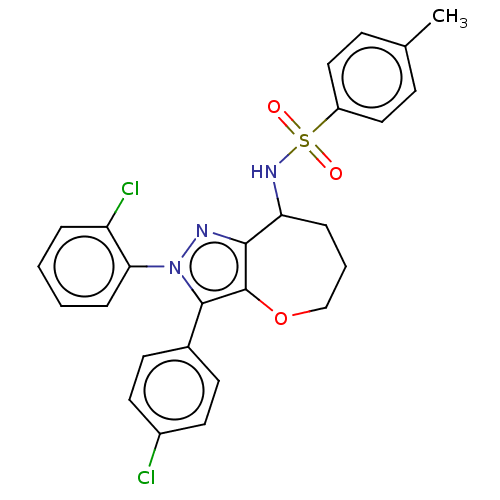
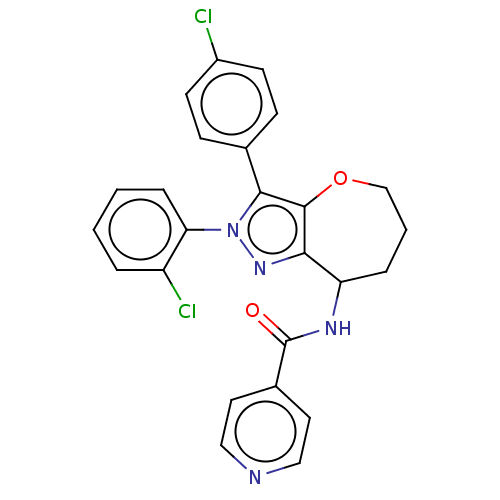
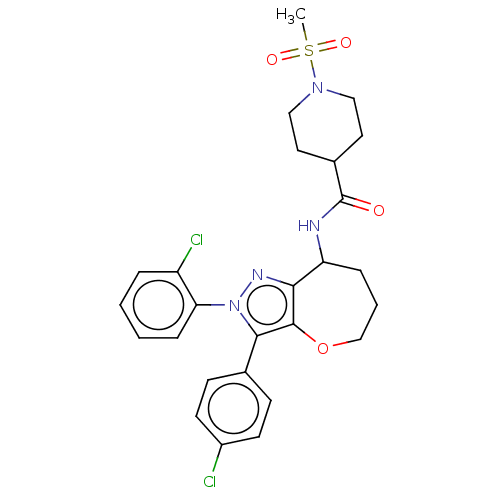
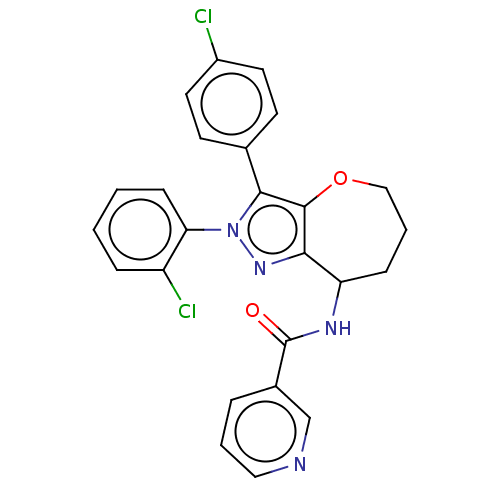
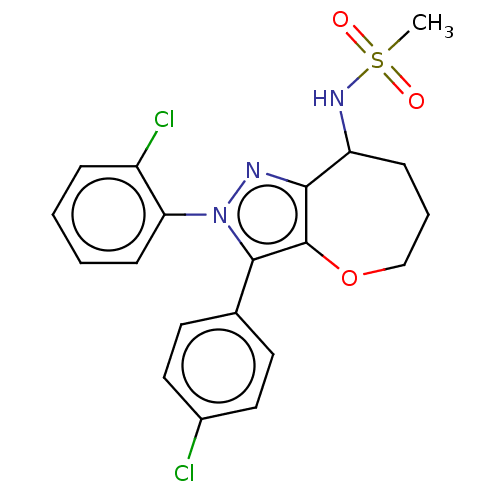
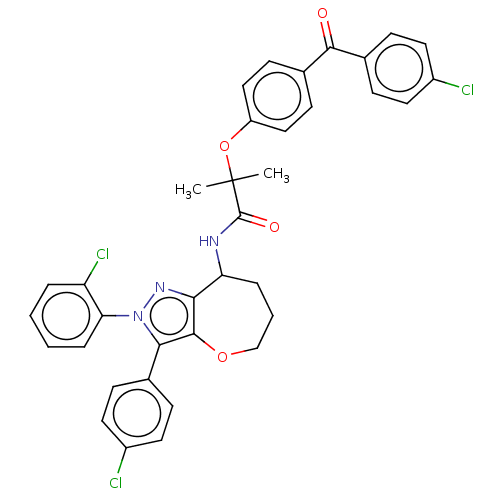
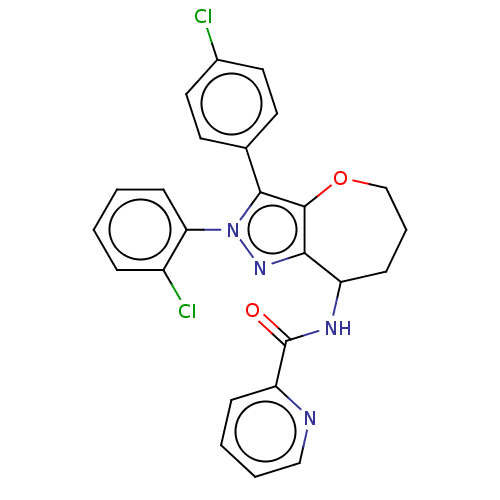
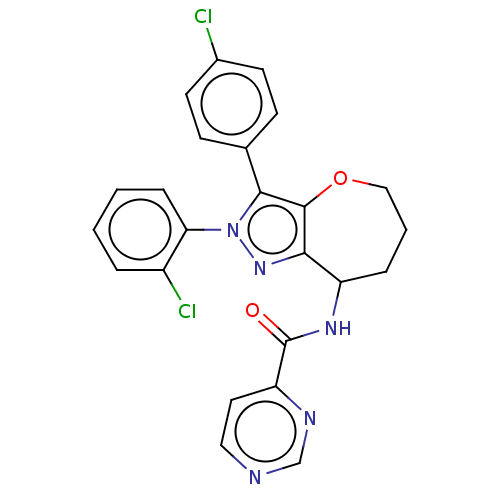
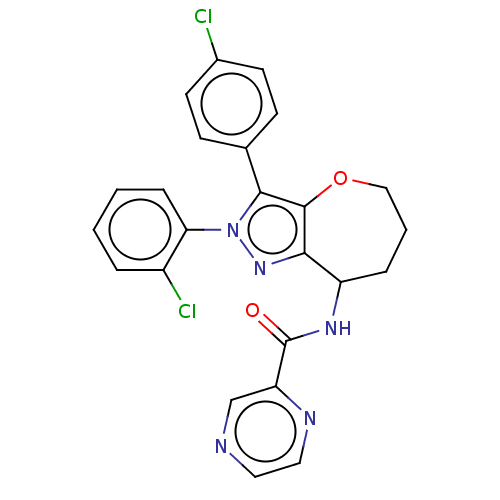
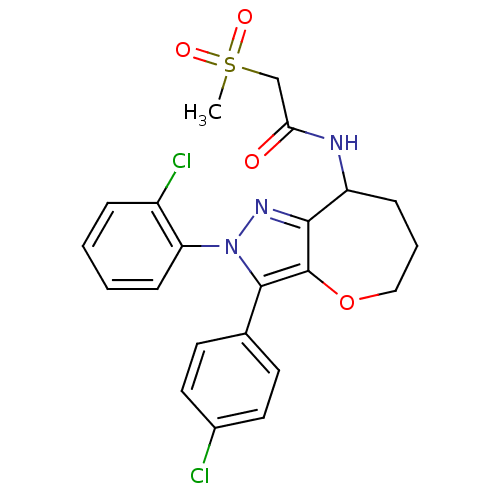
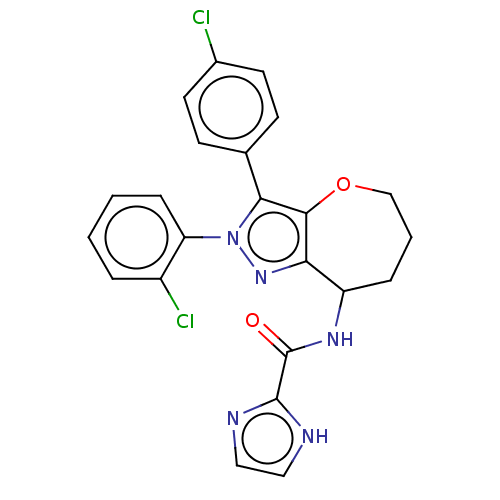
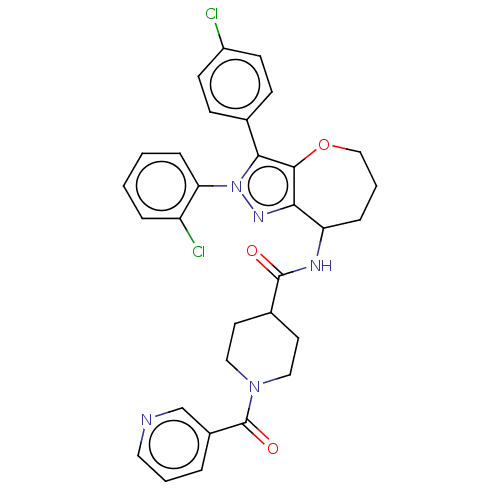
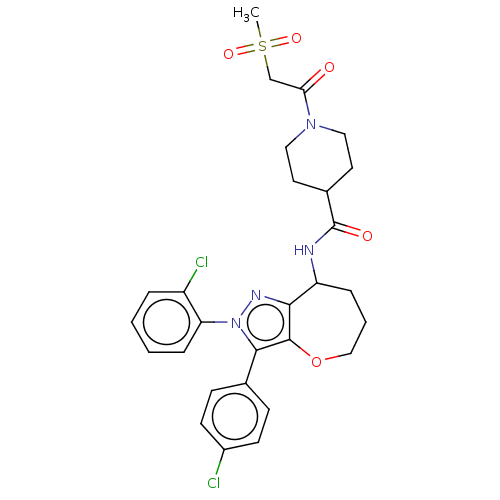
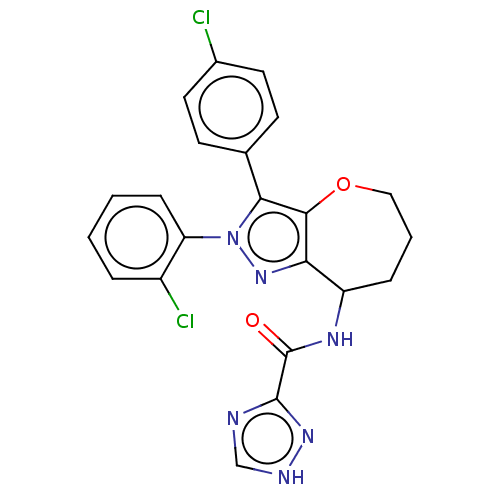
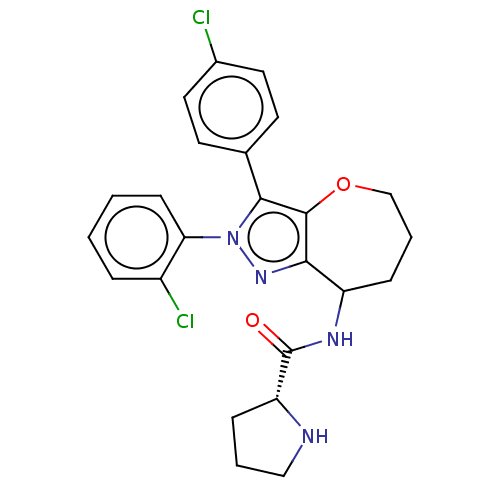
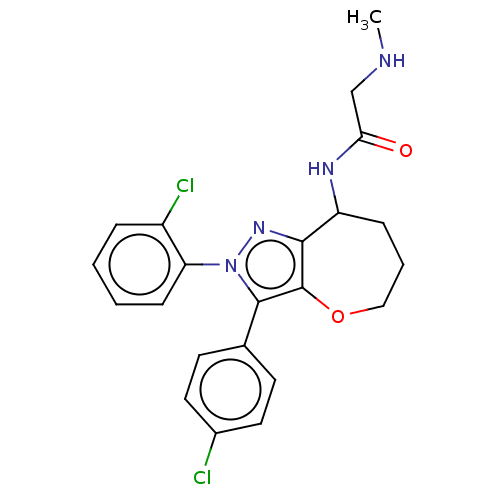
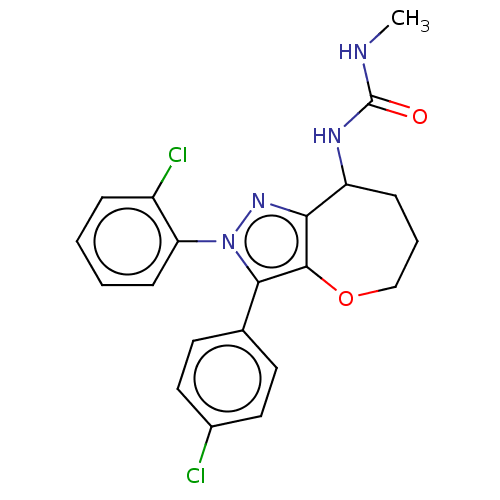
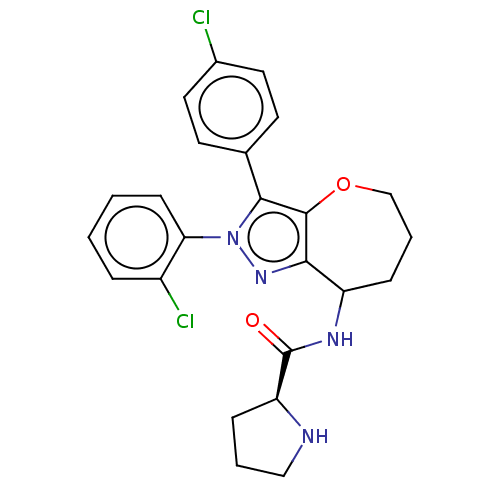
Report error Found 39 Enz. Inhib. hit(s) with all data for entry = 12216
Affinity DataKi: 0.600nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 1.16nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 1.60nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 8.93nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 10nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 16.3nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 18nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 22.4nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 33.8nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 56.7nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 64.5nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 65.6nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 77.8nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 111nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 127nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataKi: 165nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 290nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 456nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 805nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 877nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 922nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 963nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataKi: 988nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataKi: 1.13E+3nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 1.30E+3nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+3nMAssay Description:Mouse brain membranes were used as the source material for CB1 receptors. The displacement of specifically bound tritiated CP-55.940 from these membr...More data for this Ligand-Target Pair
Affinity DataKi: 1.53E+3nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataKi: 1.65E+3nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataKi: 1.93E+3nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataIC50: 2.58E+3nMAssay Description:Cardiotoxicity potential of BNS808 and BNS822 on the hERG potassium channels was evaluated using the automated patch clamp method (SyncroPatch 384PE)...More data for this Ligand-Target Pair
Affinity DataIC50: 5.39E+3nMAssay Description:Cardiotoxicity potential of BNS808 and BNS822 on the hERG potassium channels was evaluated using the automated patch clamp method (SyncroPatch 384PE)...More data for this Ligand-Target Pair
Affinity DataKi: 5.70E+3nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataKi: 6.31E+3nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataKi: 8.34E+3nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataKi: >9.11E+3nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataKi: >9.11E+3nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataKi: >9.11E+3nMAssay Description:Human kidney membranes were used as the source material for CB2 receptors. The displacement of specifically bound tritiated CP-55,940 from these memb...More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+4nMAssay Description:Cardiotoxicity potential of BNS808 and BNS822 on the hERG potassium channels was evaluated using the automated patch clamp method (SyncroPatch 384PE)...More data for this Ligand-Target Pair