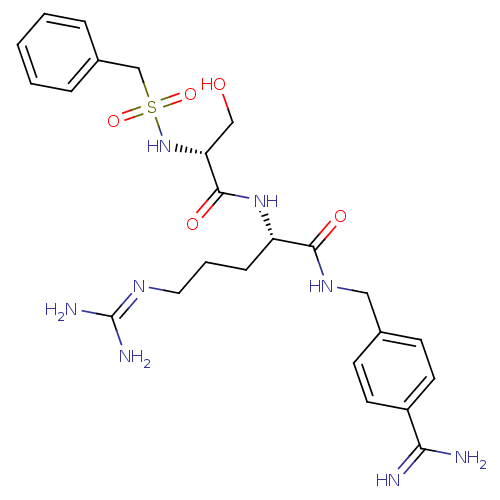
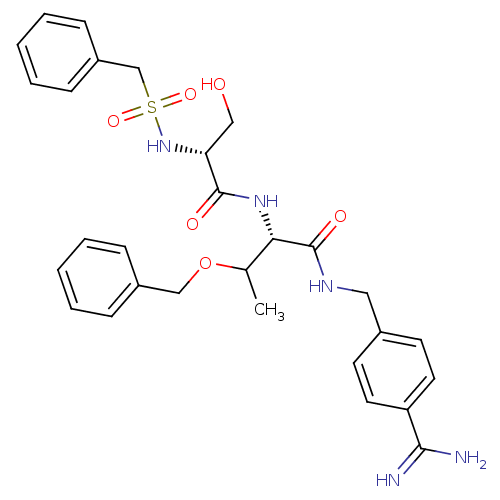
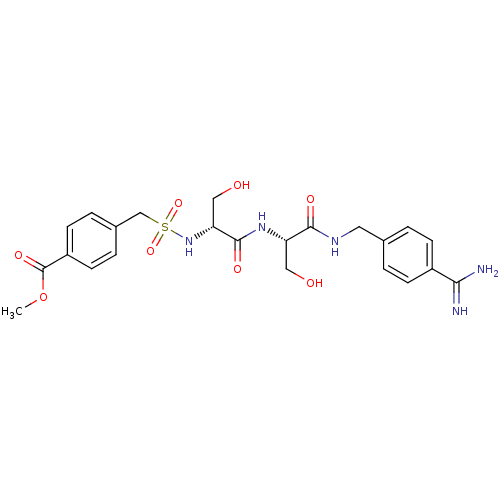
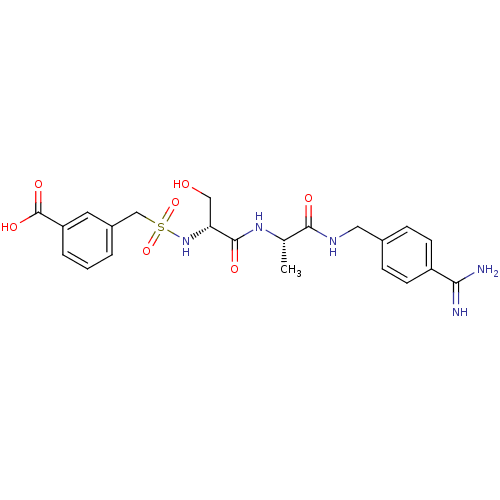
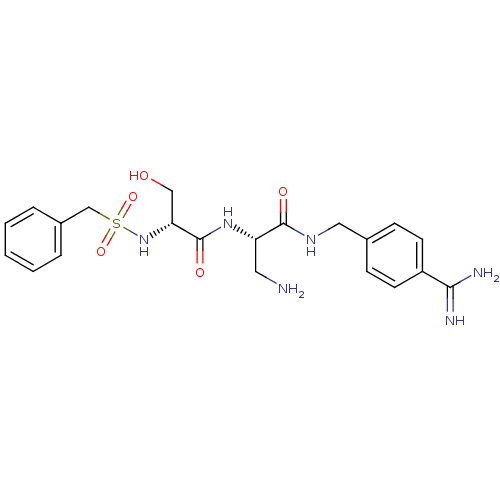
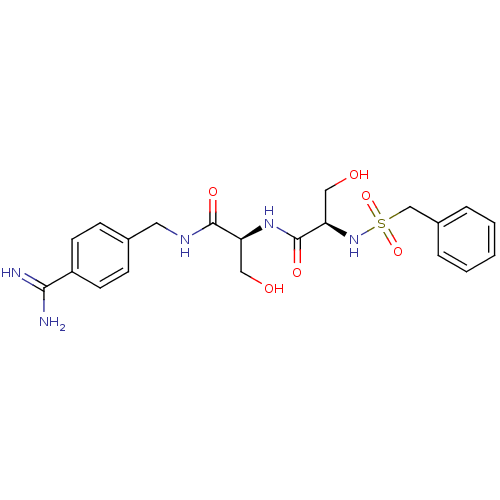
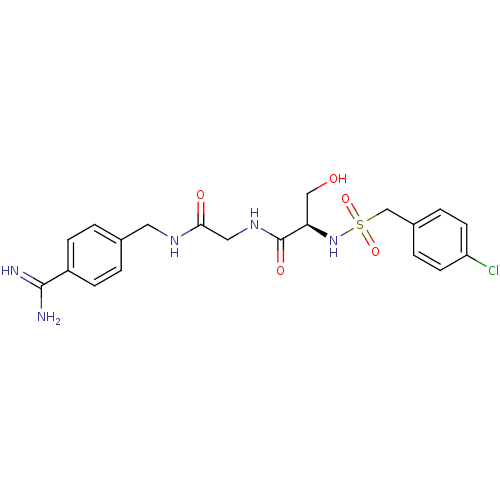
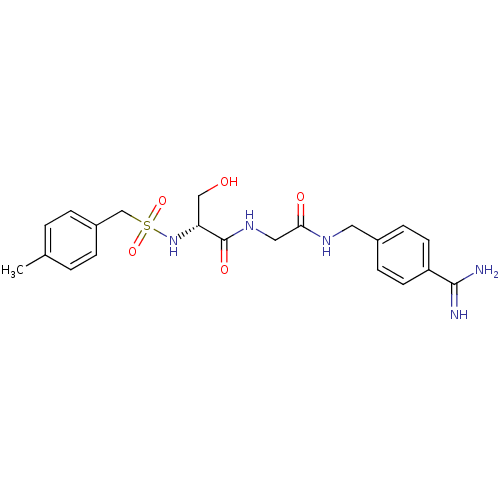
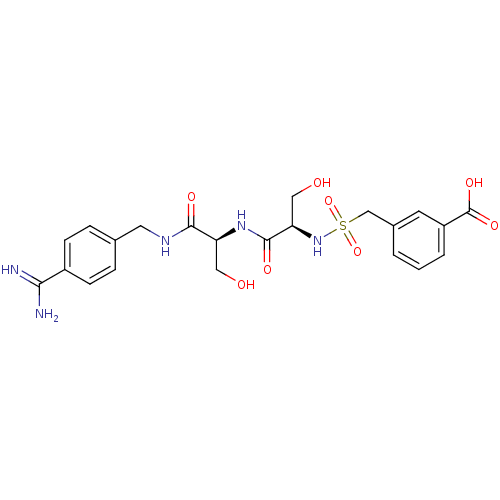
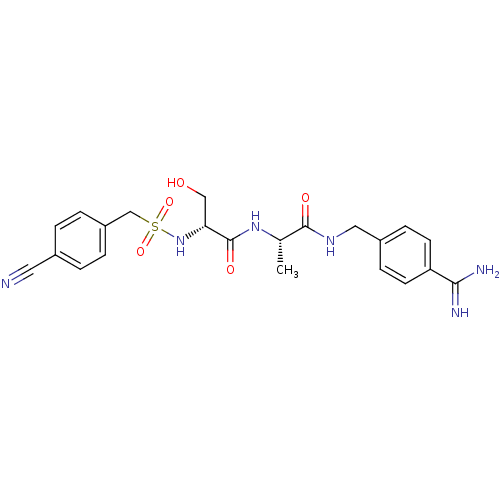
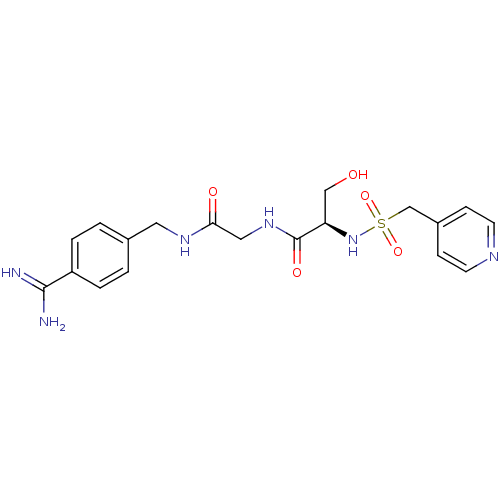
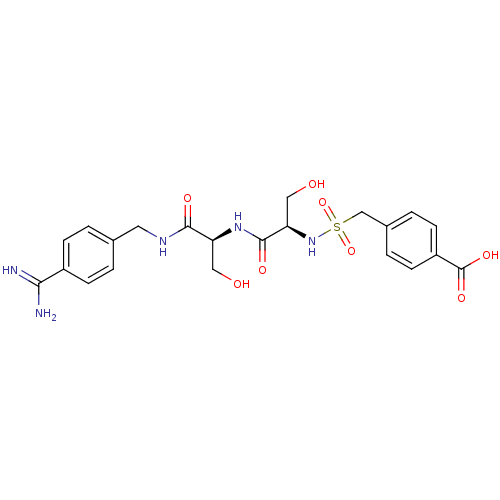
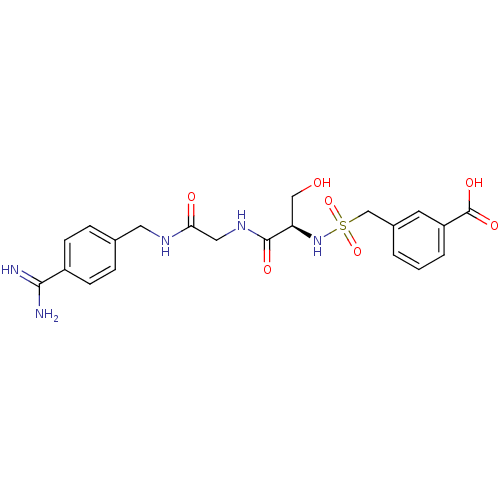
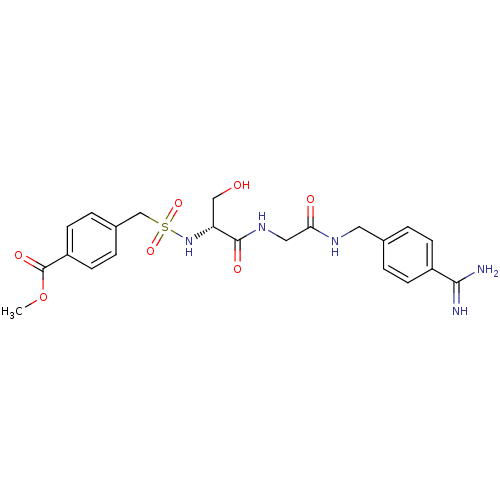
Report error Found 120 Enz. Inhib. hit(s) with all data for entry = 5887
Affinity DataKi: 4.70nM ΔG°: -47.4kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 6.80nM ΔG°: -46.5kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 6.80nM ΔG°: -46.5kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 7nM ΔG°: -46.4kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 8.90nM ΔG°: -45.8kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 11nM ΔG°: -45.3kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 13nM ΔG°: -44.9kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 20nM ΔG°: -43.8kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 21nM ΔG°: -43.7kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 21nM ΔG°: -43.7kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 22nM ΔG°: -43.6kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 24nM ΔG°: -43.3kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 25nM ΔG°: -43.2kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 25nM ΔG°: -43.2kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 28nM ΔG°: -43.0kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 30nM ΔG°: -42.8kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 30nM ΔG°: -42.8kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 31nM ΔG°: -42.7kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 32nM ΔG°: -42.6kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 36nM ΔG°: -42.3kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 36nM ΔG°: -42.3kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 38nM ΔG°: -42.2kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 42nM ΔG°: -42.0kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 58nM ΔG°: -41.2kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 58nM ΔG°: -41.2kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 61nM ΔG°: -41.0kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 75nM ΔG°: -40.5kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 83nM ΔG°: -40.3kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 89nM ΔG°: -40.1kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 100nM ΔG°: -39.8kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 100nM ΔG°: -39.8kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 100nM ΔG°: -39.8kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against ThrombinMore data for this Ligand-Target Pair
Affinity DataKi: 110nM ΔG°: -39.6kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 110nM ΔG°: -39.6kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 110nM ΔG°: -39.6kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 110nM ΔG°: -39.6kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 120nM ΔG°: -39.4kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 120nM ΔG°: -39.4kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 130nM ΔG°: -39.2kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 150nM ΔG°: -38.8kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 150nM ΔG°: -38.8kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 160nM ΔG°: -38.7kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 180nM ΔG°: -38.4kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 180nM ΔG°: -38.4kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 180nM ΔG°: -38.4kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 200nM ΔG°: -38.1kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against PlasminMore data for this Ligand-Target Pair
Affinity DataKi: 210nM ΔG°: -38.0kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against TrypsinMore data for this Ligand-Target Pair
Affinity DataKi: 230nM ΔG°: -37.8kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 230nM ΔG°: -37.8kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against Plasminogen activator urokinaseMore data for this Ligand-Target Pair
Affinity DataKi: 270nM ΔG°: -37.4kJ/moleT: 2°CAssay Description:Inhibition constant of the compound against PlasminMore data for this Ligand-Target Pair