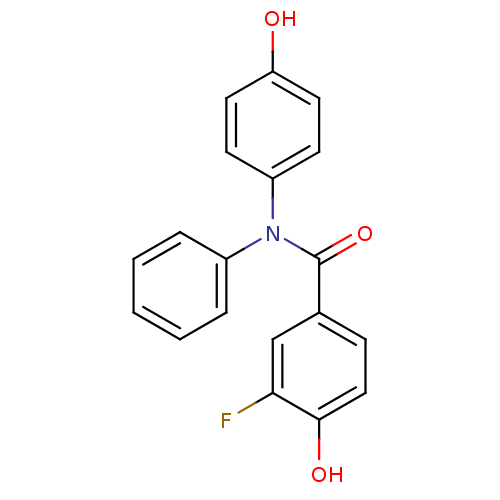
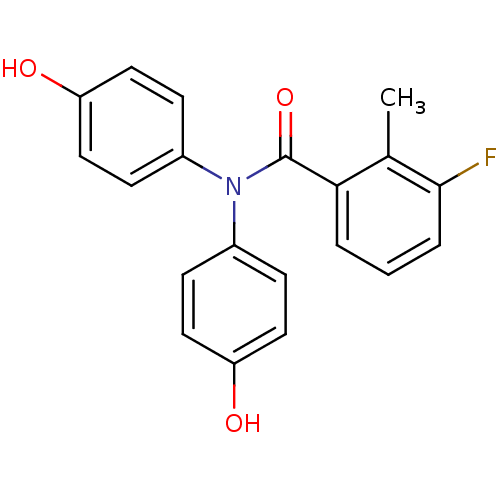
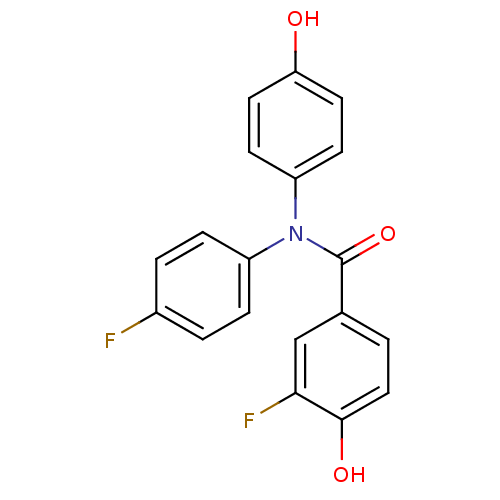
Report error Found 20 Enz. Inhib. hit(s) with all data for entry = 5965
Affinity DataKi: 3.75nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 3.81nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 6.06nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 6.44nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 7.13nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 9nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 14.8nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 15.1nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 15.2nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 19nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 21.7nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 25.0nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 35.9nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 57nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 72nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 76.9nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 81.6nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair
Affinity DataKi: 646nMAssay Description:The ER binding affinity of the compounds was determined using an in vitro competitive radioligand binding assay was [2,4,6,7-3H(N)]-Estradiol ([3H]E2...More data for this Ligand-Target Pair