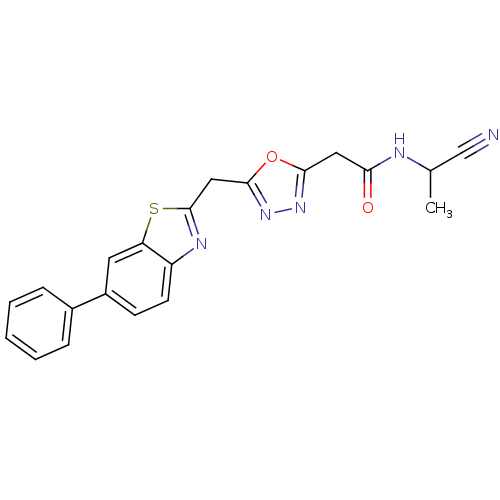
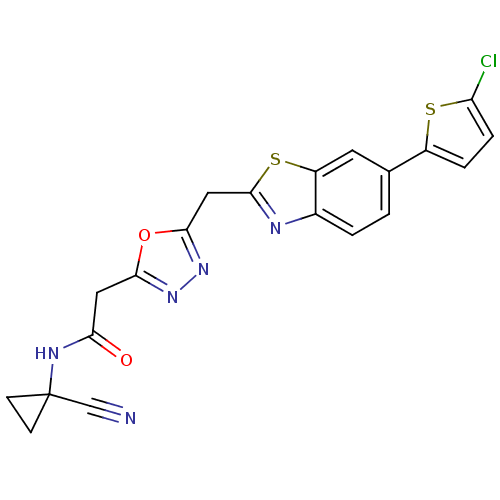
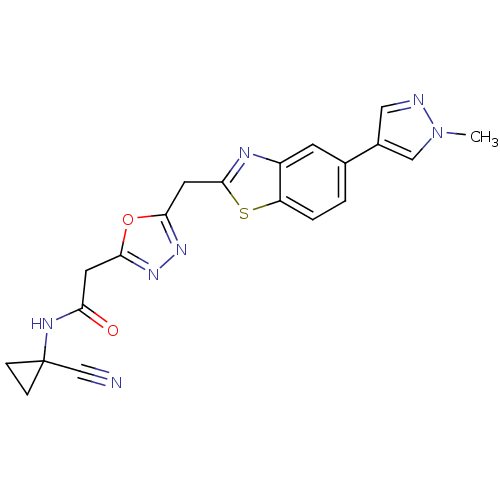
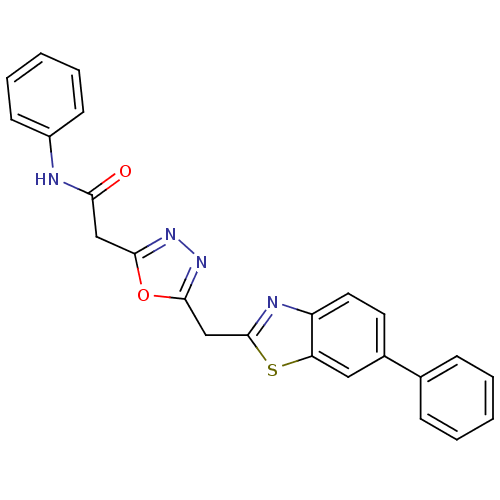
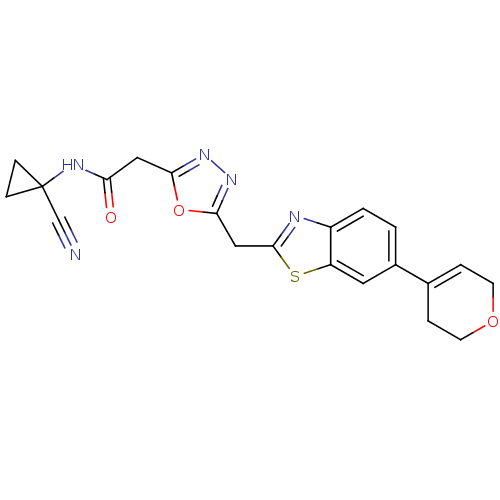
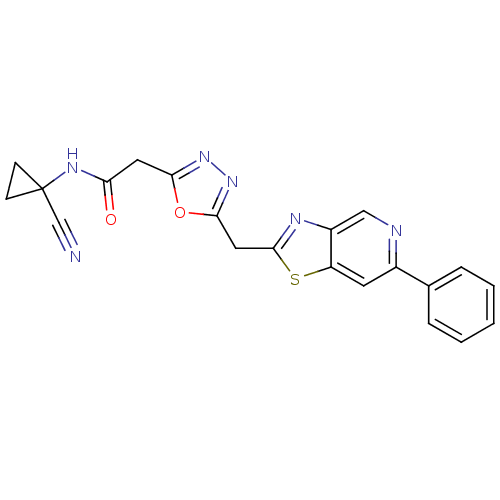
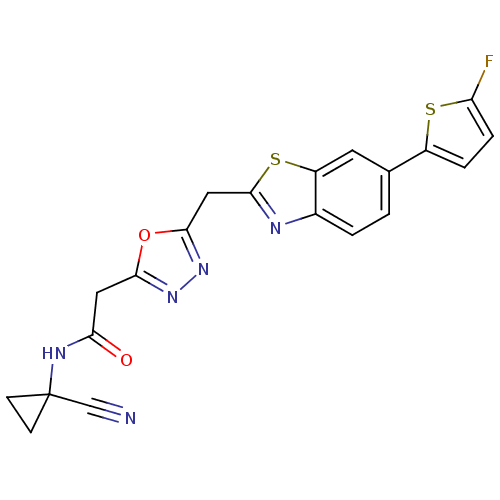
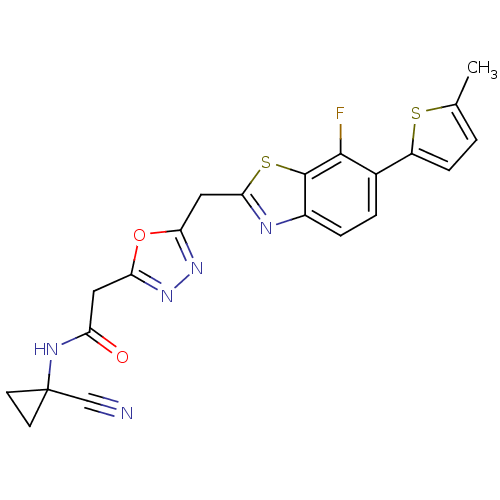
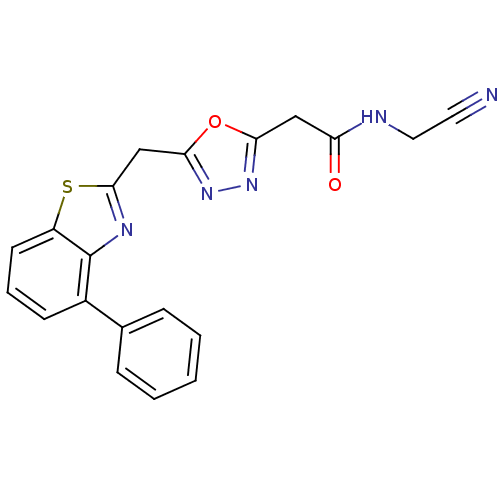
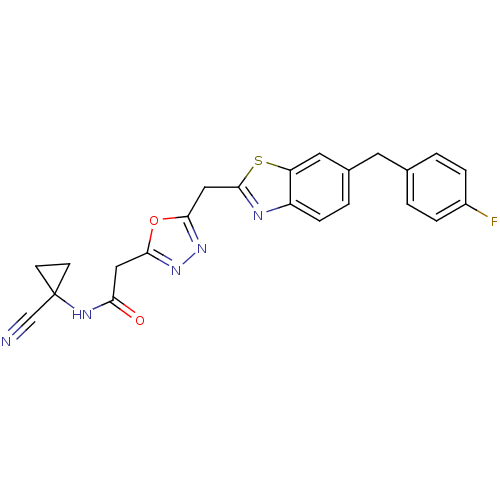
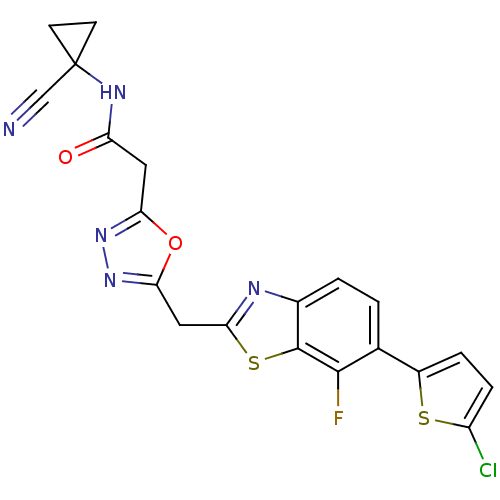
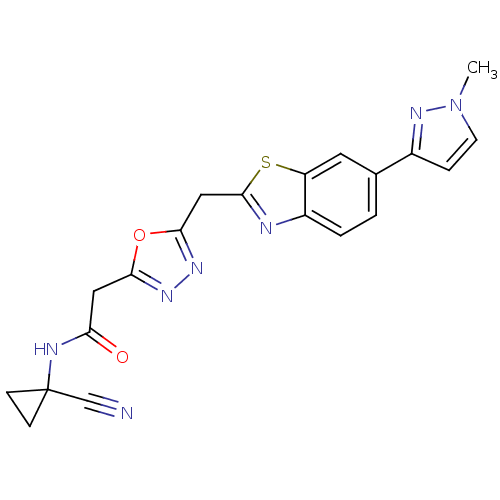
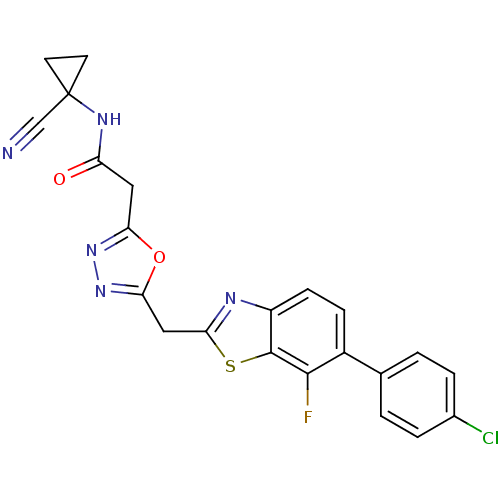
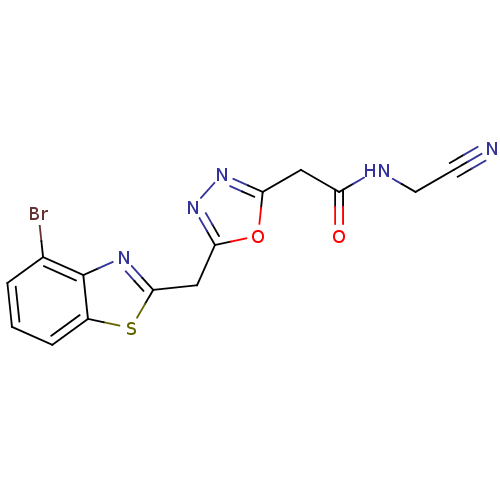
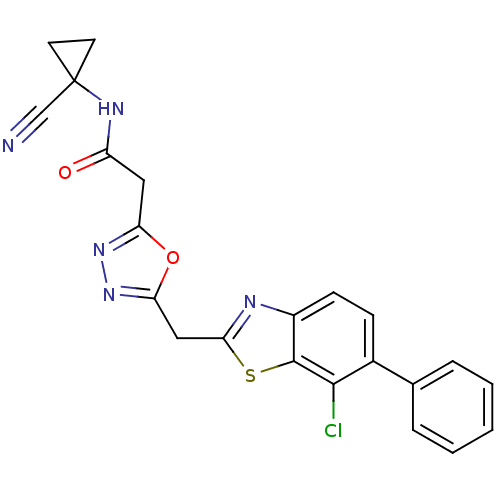
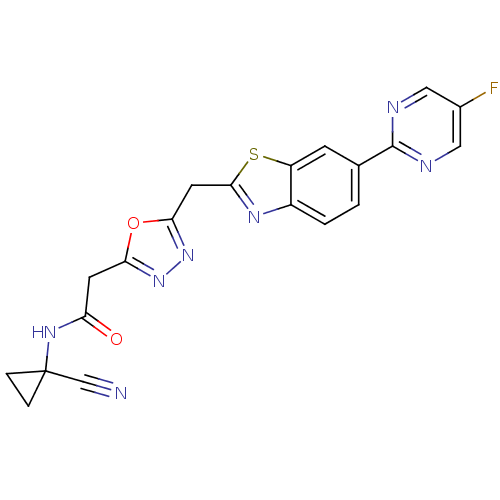
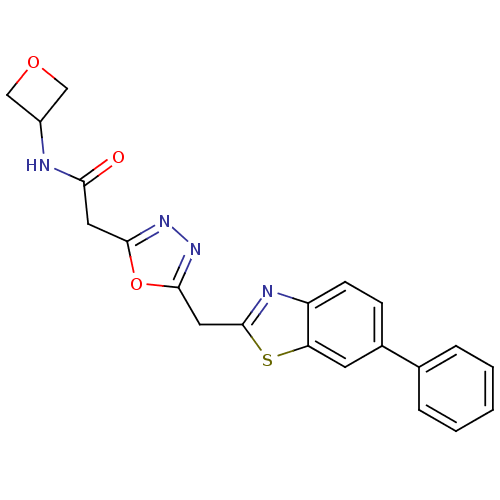
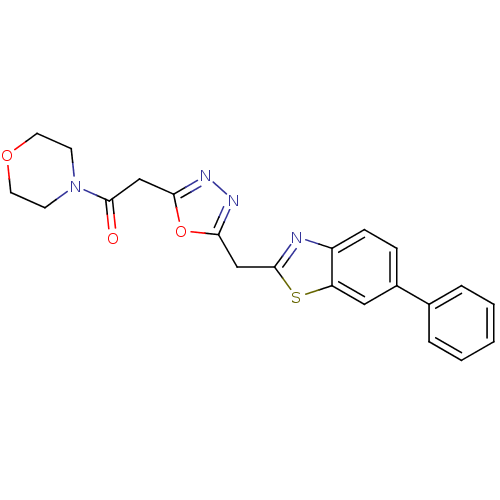
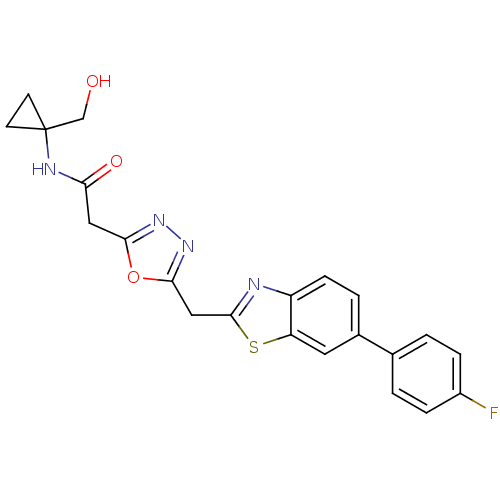
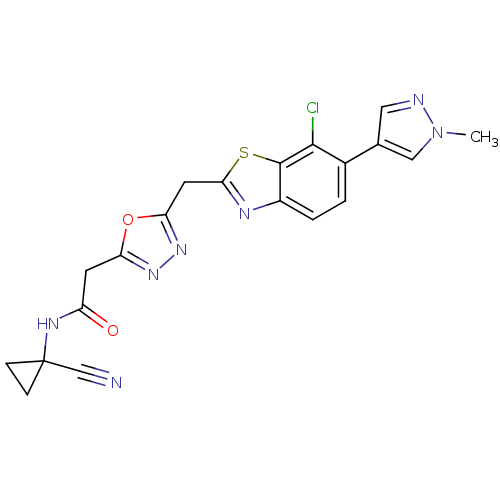
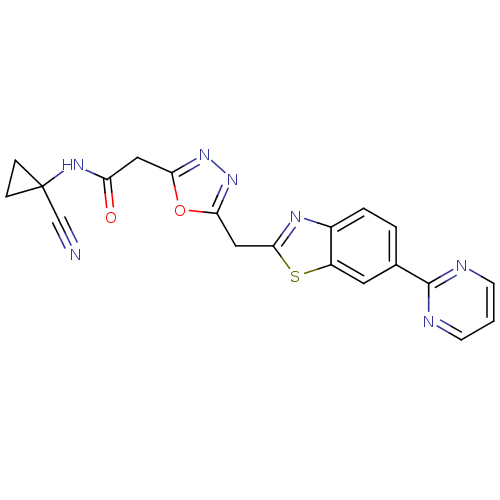
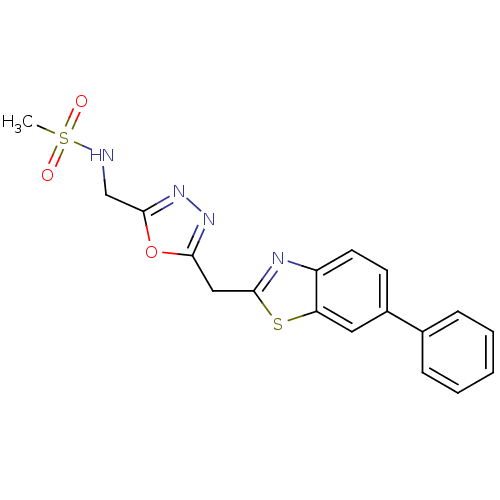
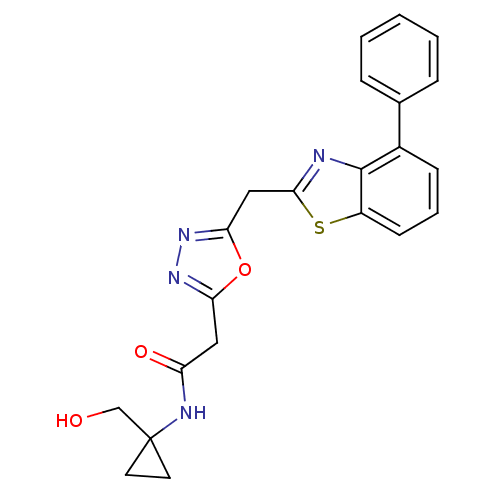
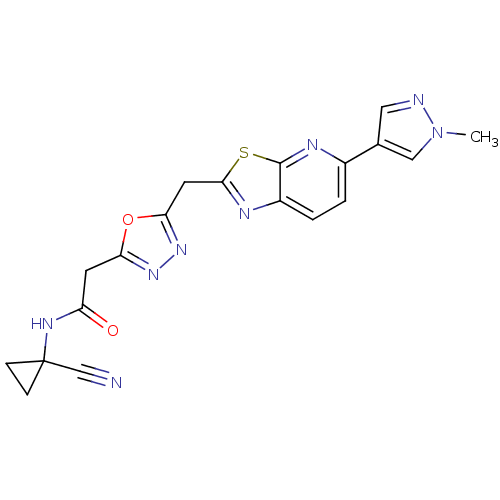
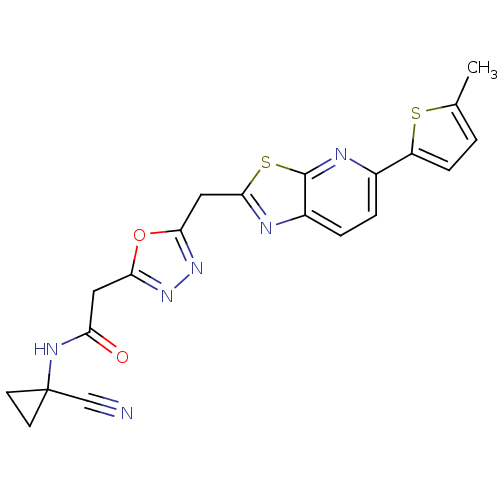
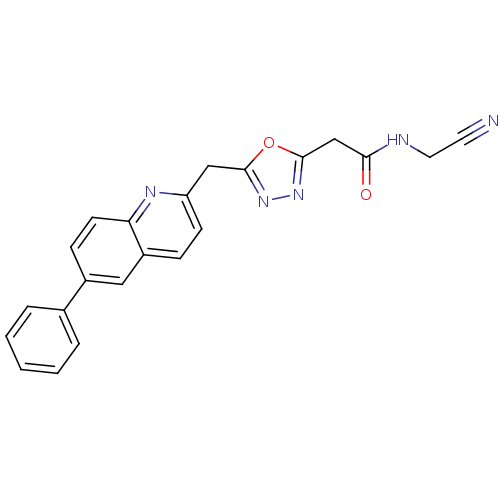
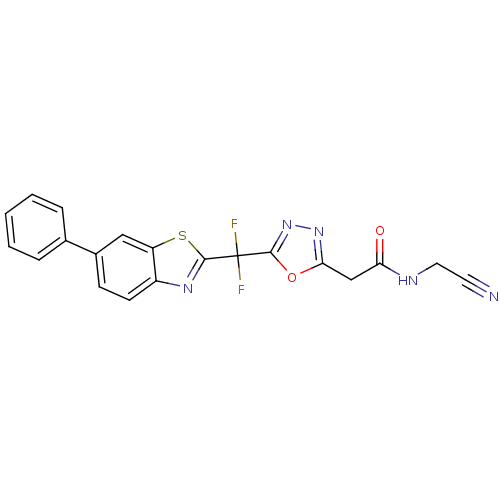
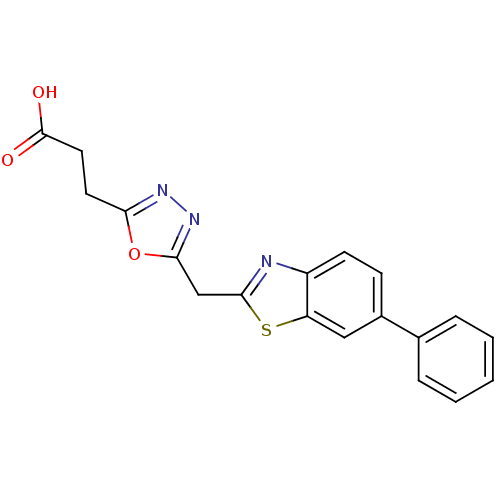
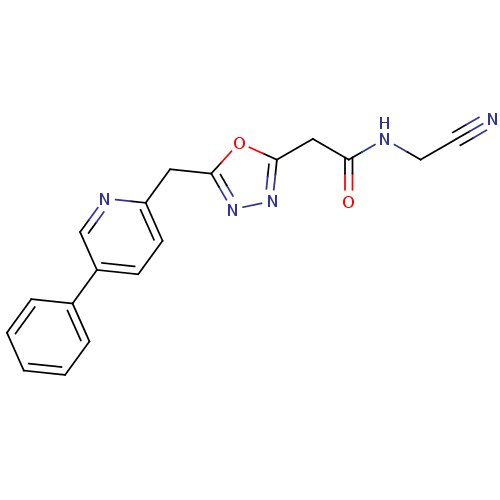
Report error Found 39 Enz. Inhib. hit(s) with all data for entry = 6348
Affinity DataIC50: 2nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 8nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 8nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 22nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 27nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 28nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 36nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 41nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 42nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 44nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 46nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 52nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 52nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 65nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 69nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 73nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 78nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 83nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 85nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 110nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 160nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 190nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 250nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 260nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 390nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 400nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+3nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30E+4nMT: 2°CAssay Description:After the present compound dissolved in DMSO was added to become 0.5% DMSO to the reaction buffer consisting of 20 mM tris hydrochloric acid (pH7.4),...More data for this Ligand-Target Pair