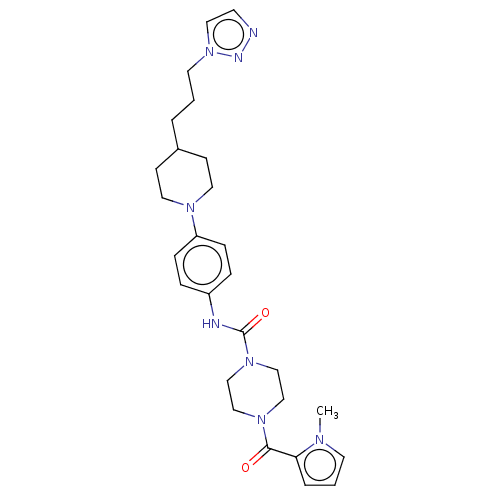
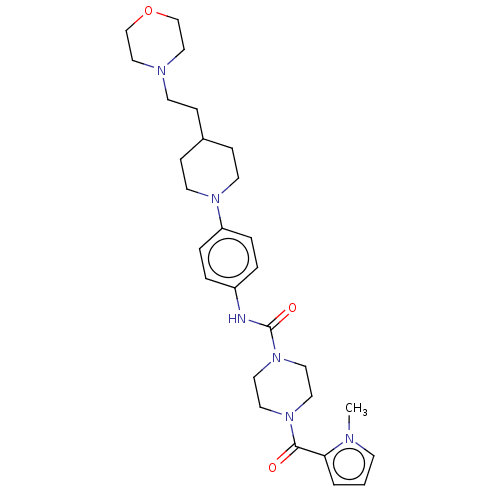
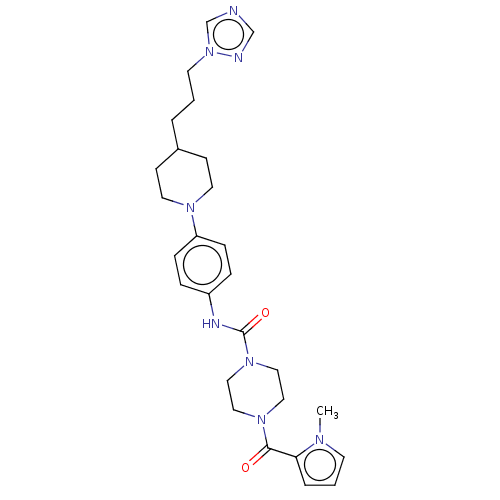
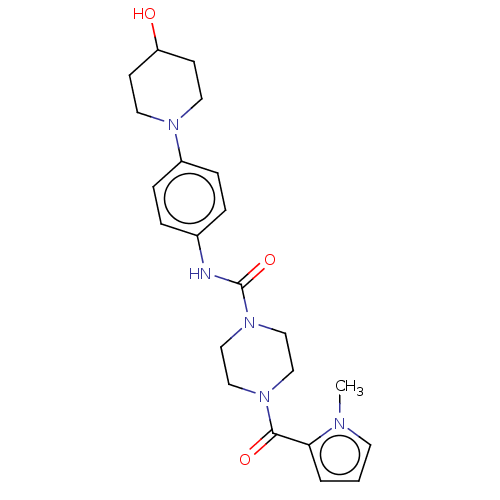
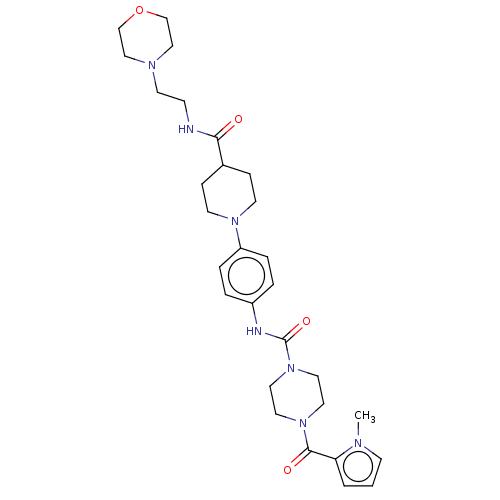
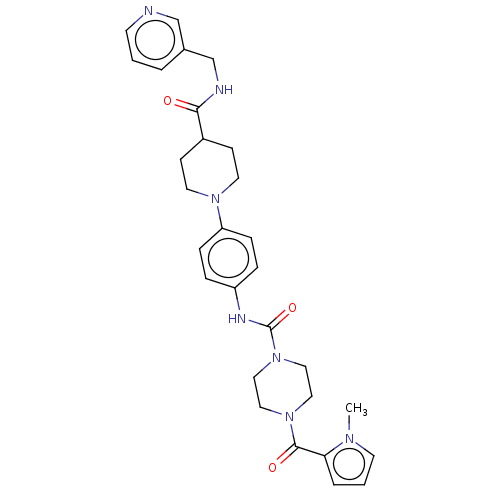
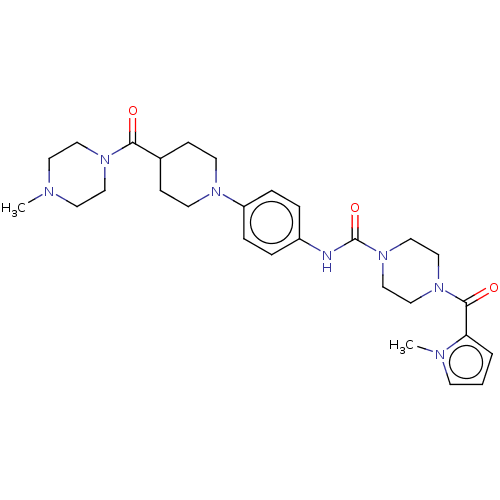
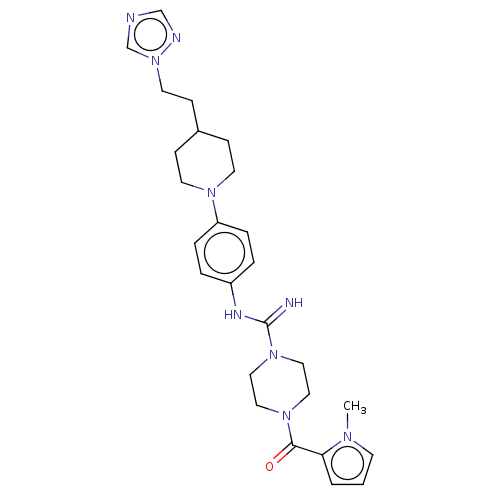
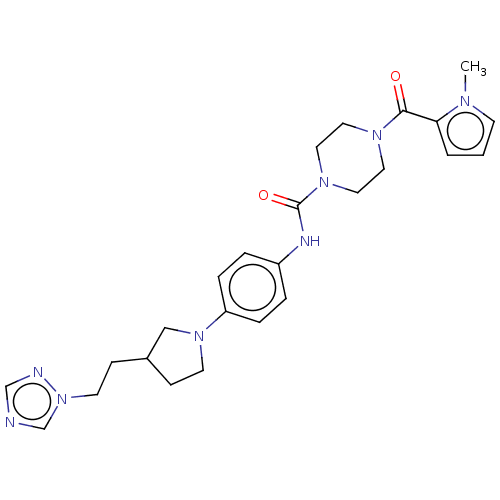
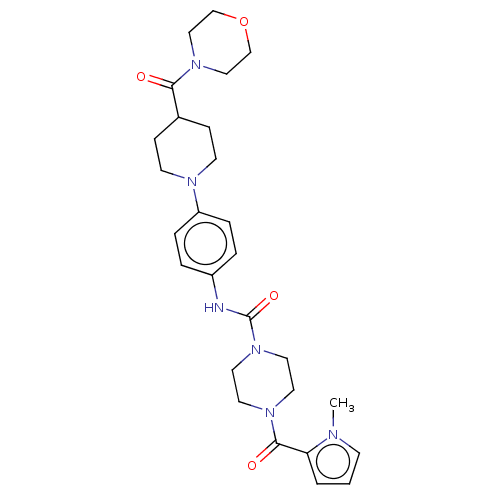
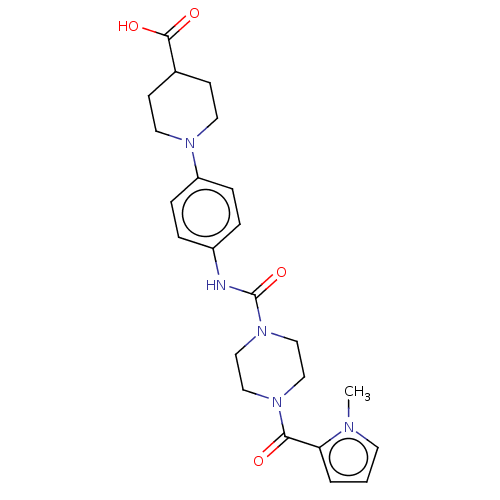
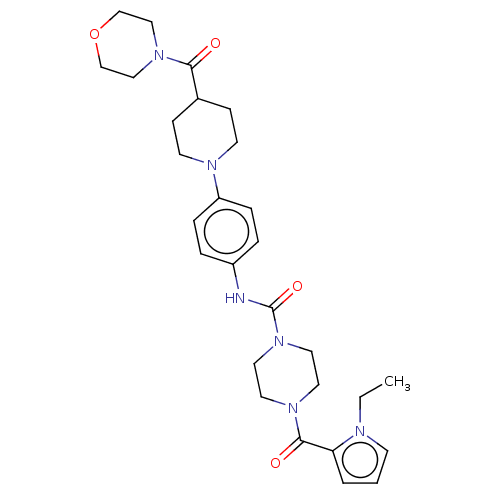
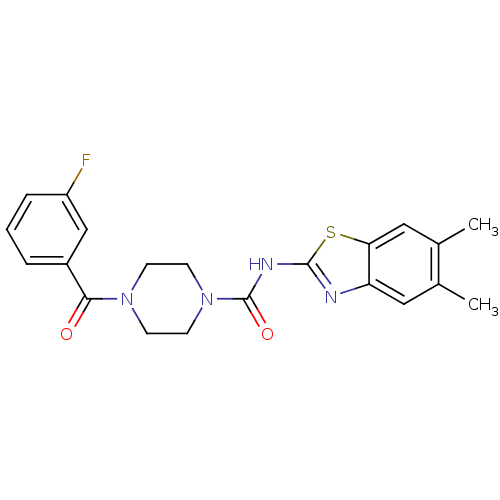
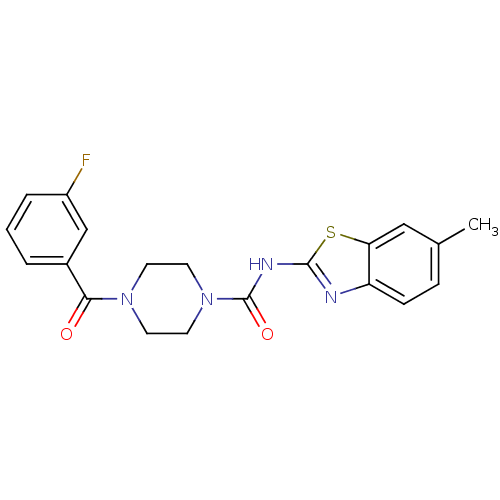
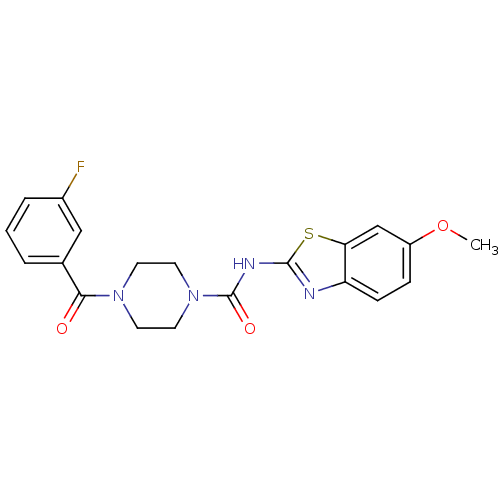
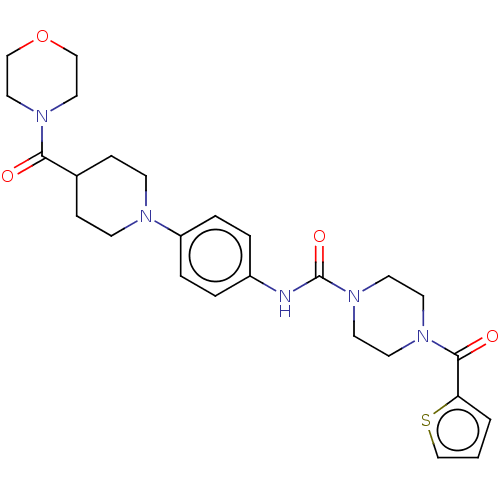
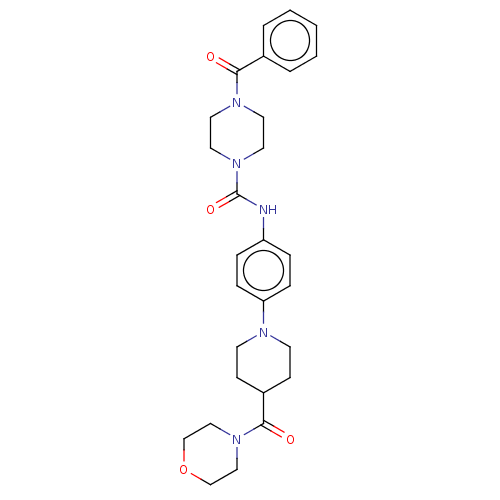
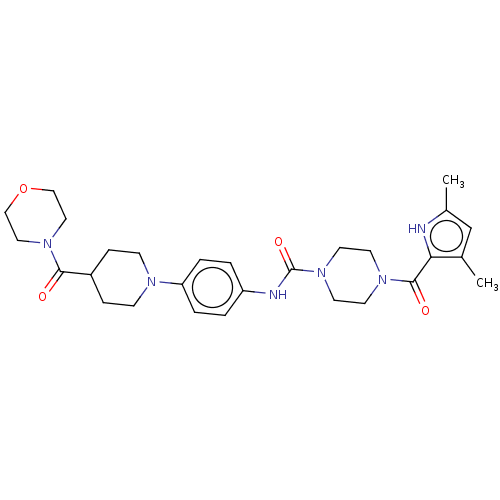
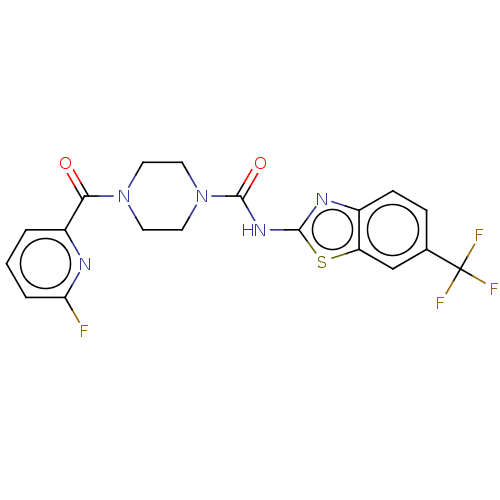
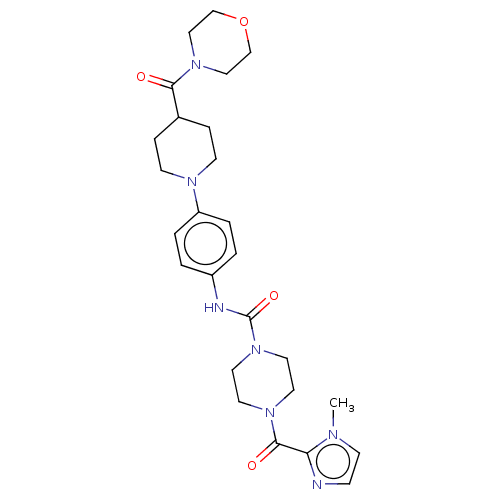
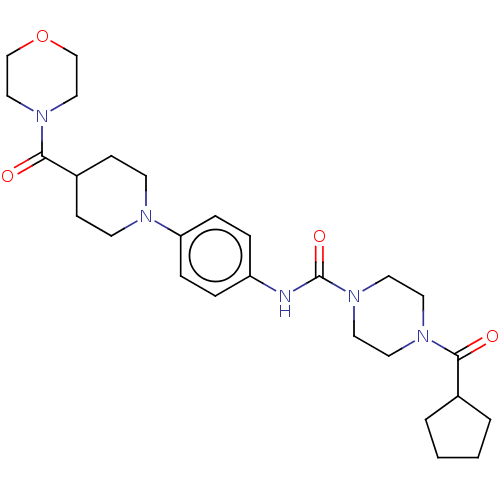
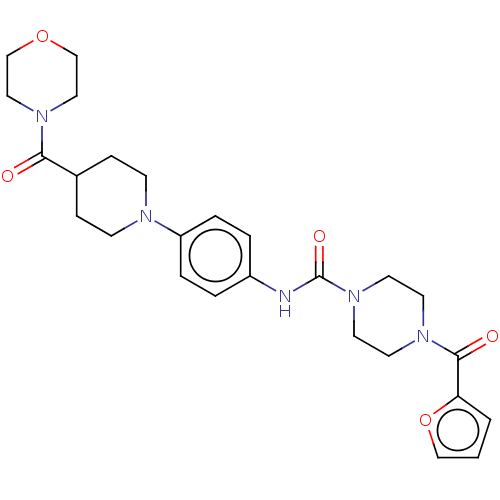
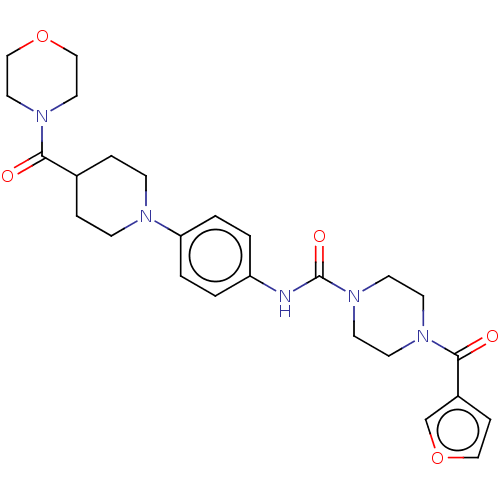
Report error Found 35 Enz. Inhib. hit(s) with all data for entry = 6530
Affinity DataIC50: 32nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 37nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 40nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 43nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 46nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 54nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 58nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 59nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 60nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 60nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 67nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 71nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 76nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 85nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 86nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 91nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 106nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 204nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 222nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 260nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 318nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 340nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 437nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 732nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMpH: 8.0 T: 2°CAssay Description:The test was carried out according to the method of Urade, Y. et al. (J. Biol. Chem., 262, 3820-3825, (1987)). More specifically, the reaction mixtur...More data for this Ligand-Target Pair