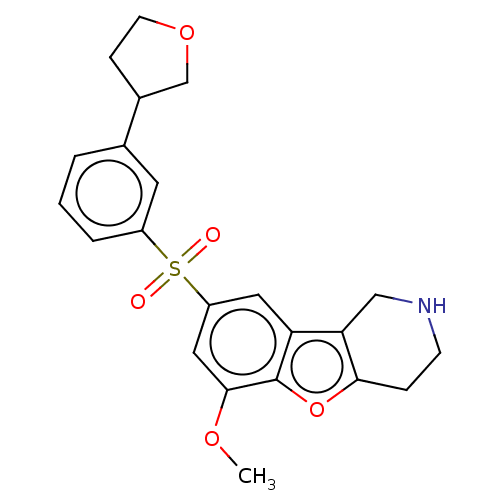
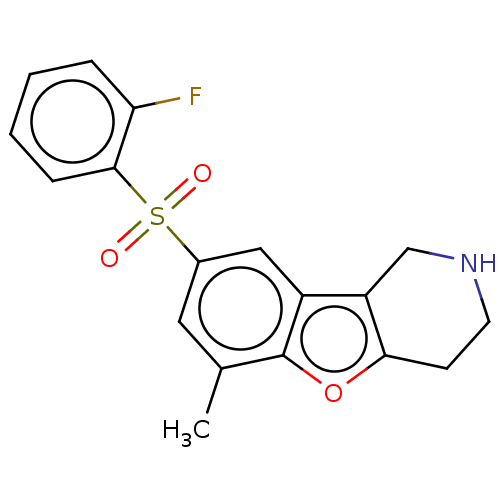
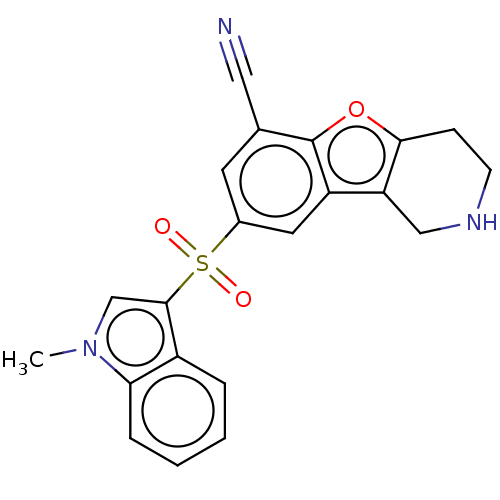
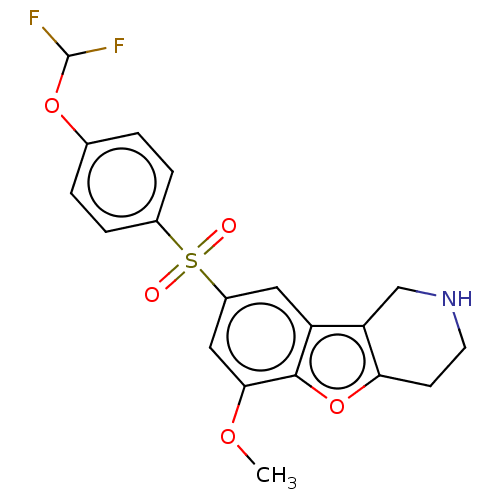
Report error Found 216 Enz. Inhib. hit(s) with all data for entry = 7010
Affinity DataKi: 0.0500nM ΔG°: -58.8kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.0830nM ΔG°: -57.5kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.100nM ΔG°: -57.1kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.110nM ΔG°: -56.8kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.110nM ΔG°: -56.8kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.110nM ΔG°: -56.8kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.120nM ΔG°: -56.6kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.130nM ΔG°: -56.4kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.130nM ΔG°: -56.4kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.130nM ΔG°: -56.4kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.150nM ΔG°: -56.1kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.160nM ΔG°: -55.9kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.160nM ΔG°: -55.9kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.170nM ΔG°: -55.8kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.170nM ΔG°: -55.8kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.190nM ΔG°: -55.5kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.190nM ΔG°: -55.5kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.200nM ΔG°: -55.4kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.200nM ΔG°: -55.4kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.220nM ΔG°: -55.1kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.220nM ΔG°: -55.1kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.230nM ΔG°: -55.0kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.230nM ΔG°: -55.0kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.25nM ΔG°: -54.8kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.260nM ΔG°: -54.7kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.270nM ΔG°: -54.6kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.280nM ΔG°: -54.5kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.280nM ΔG°: -54.5kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.300nM ΔG°: -54.4kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.320nM ΔG°: -54.2kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.320nM ΔG°: -54.2kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.330nM ΔG°: -54.1kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.330nM ΔG°: -54.1kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.350nM ΔG°: -54.0kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.350nM ΔG°: -54.0kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.350nM ΔG°: -54.0kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.360nM ΔG°: -53.9kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.390nM ΔG°: -53.7kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.390nM ΔG°: -53.7kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.400nM ΔG°: -53.6kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.400nM ΔG°: -53.6kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.410nM ΔG°: -53.6kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.420nM ΔG°: -53.5kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.480nM ΔG°: -53.2kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.480nM ΔG°: -53.2kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.490nM ΔG°: -53.1kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.490nM ΔG°: -53.1kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.5nM ΔG°: -53.1kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.550nM ΔG°: -52.9kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair
Affinity DataKi: 0.550nM ΔG°: -52.9kJ/molepH: 7.5 T: 2°CAssay Description:For binding analysis vs. the human receptor, samples were incubated in 50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM EDTA (4% DMSO final) with 10 nM [N-me...More data for this Ligand-Target Pair