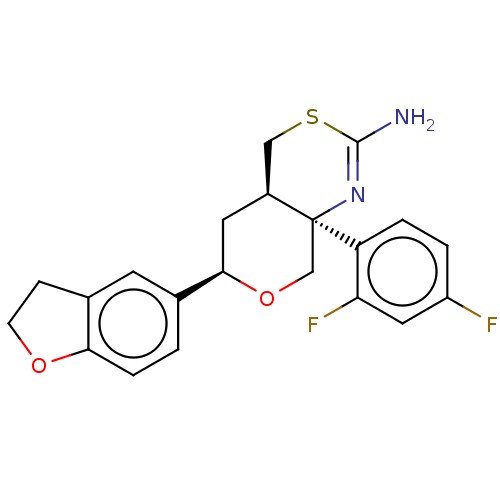
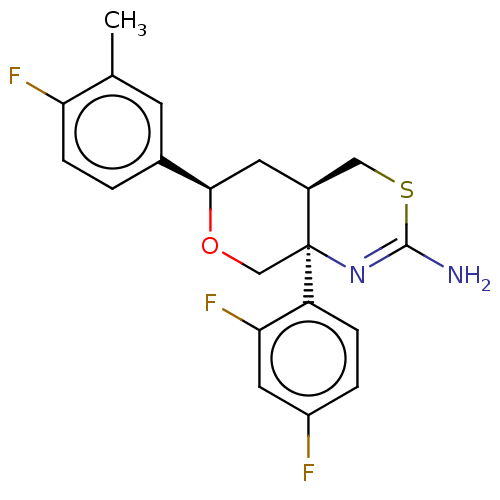
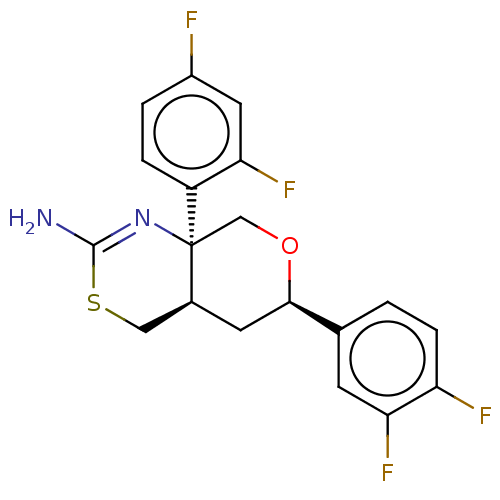
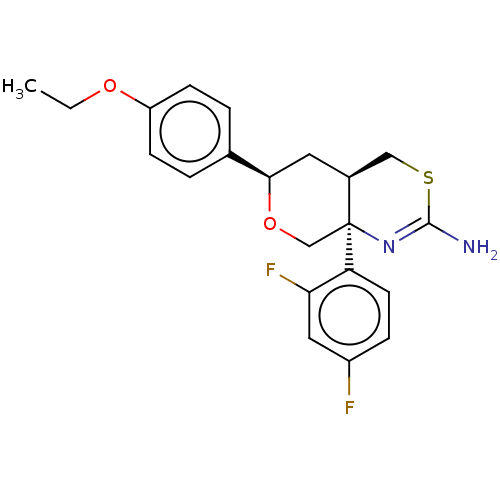
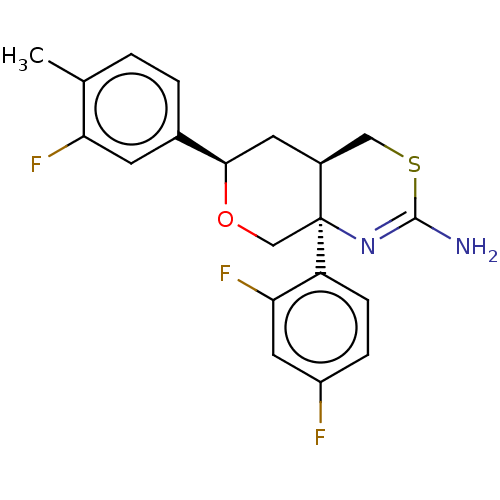
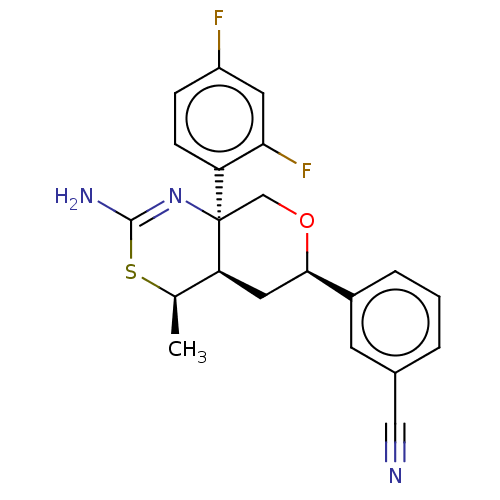
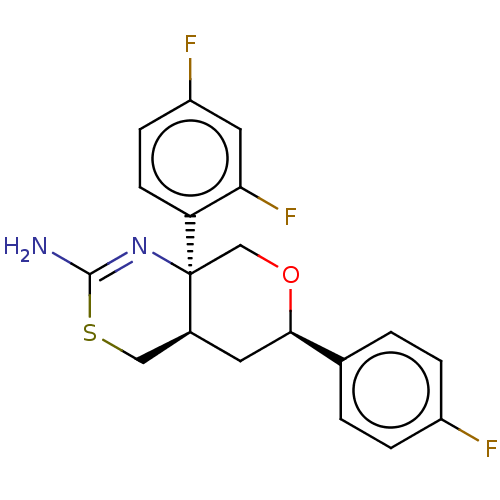
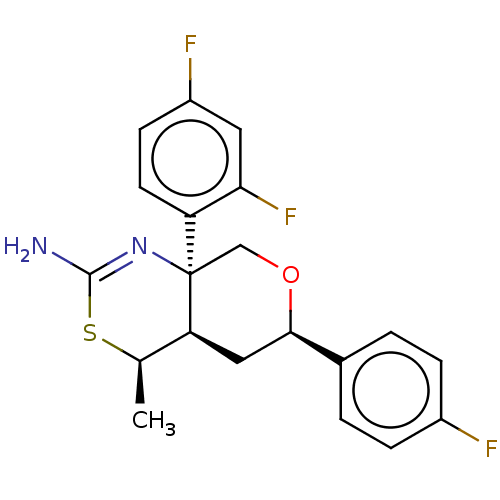
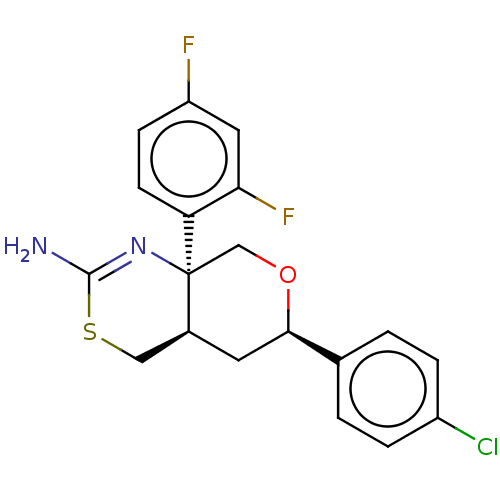
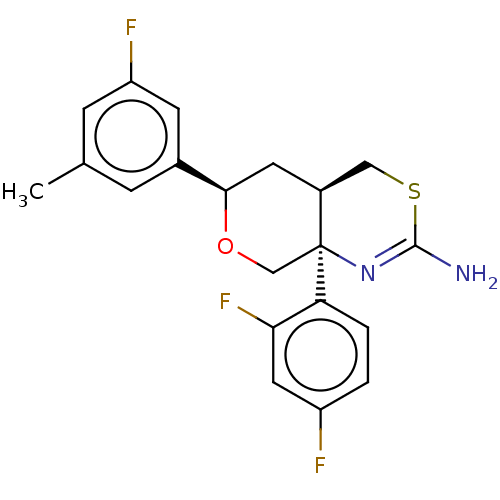
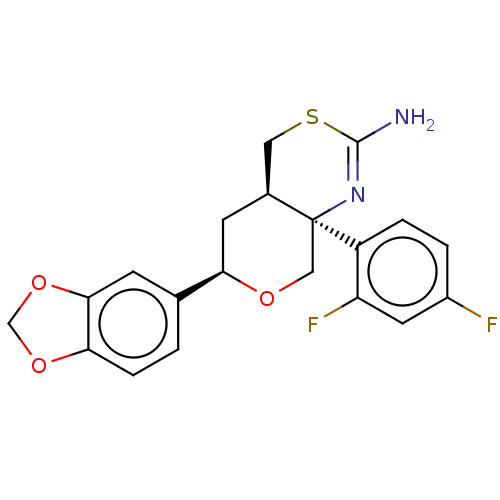
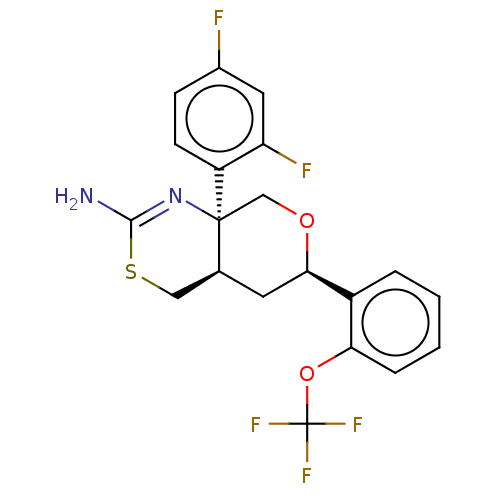
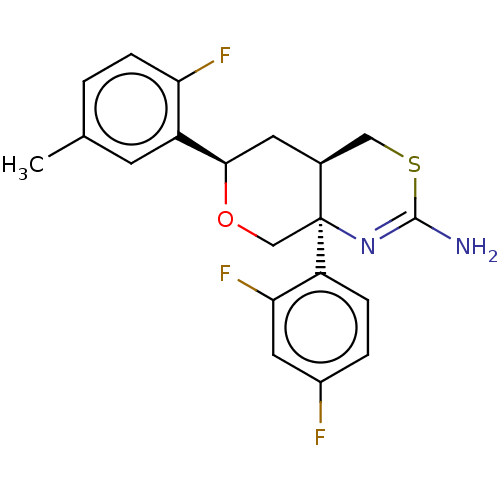
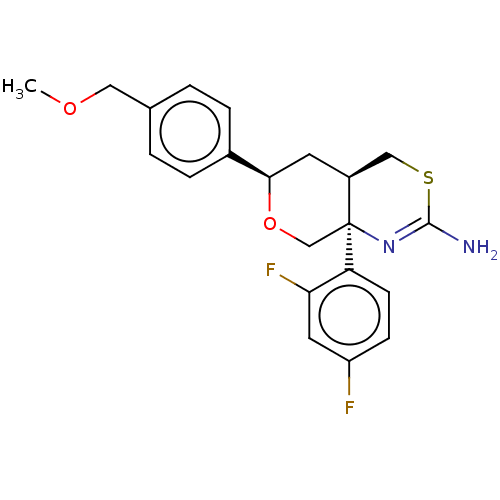
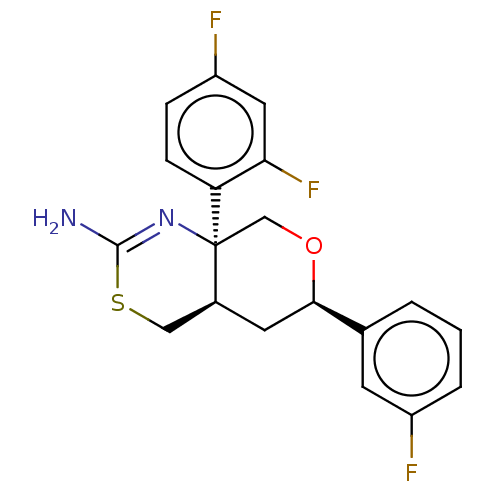
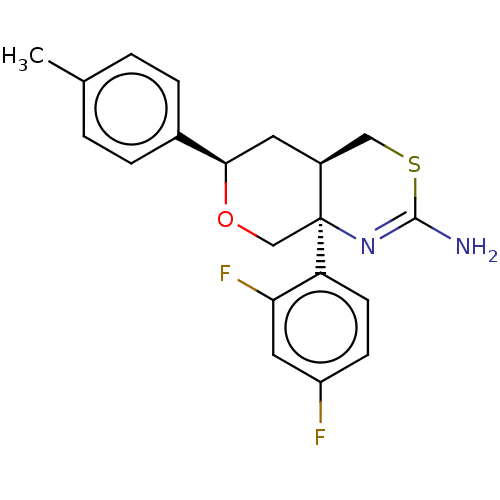
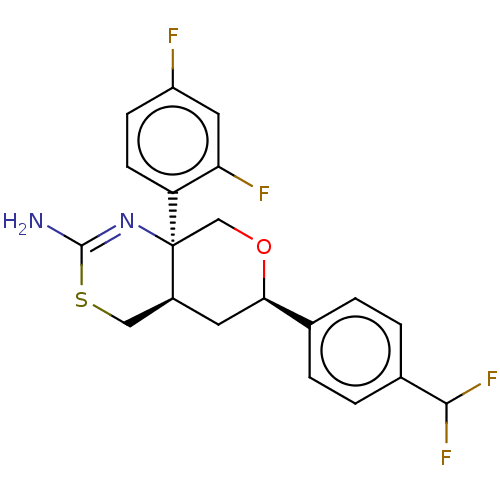
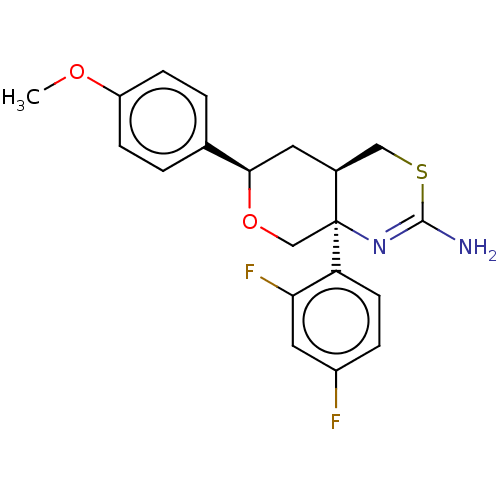
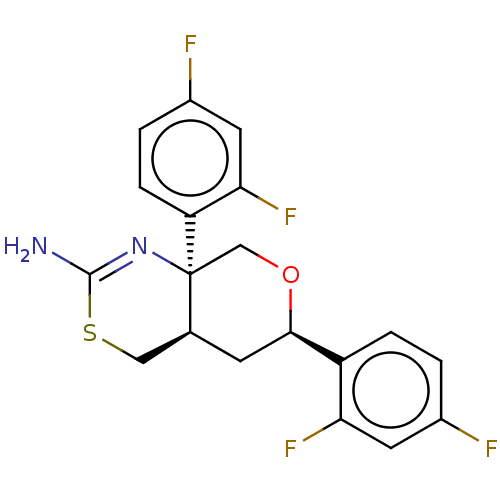
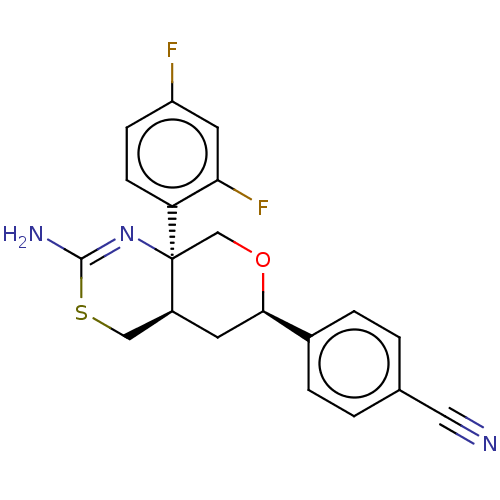
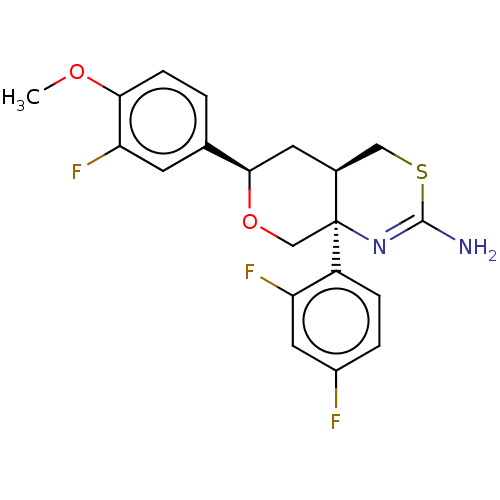
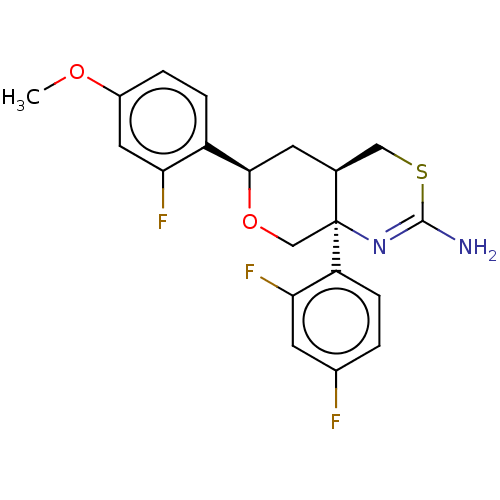
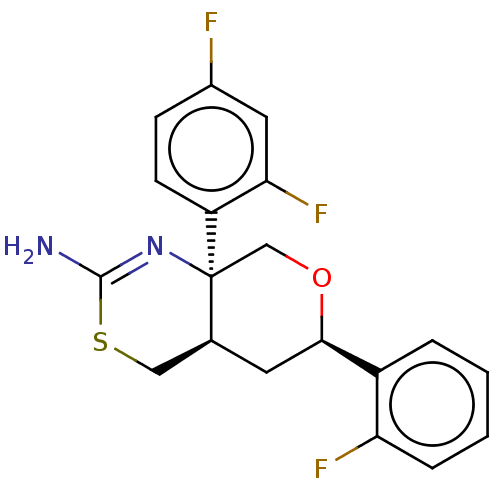
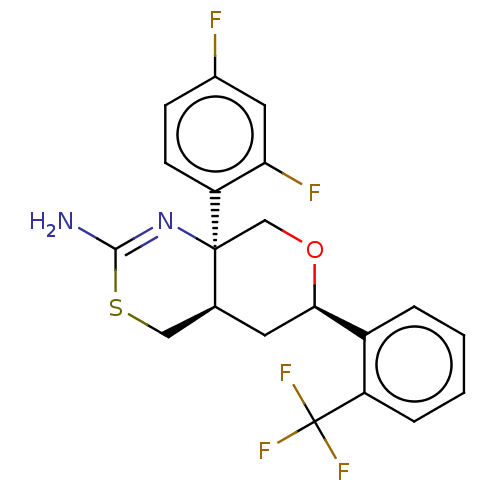
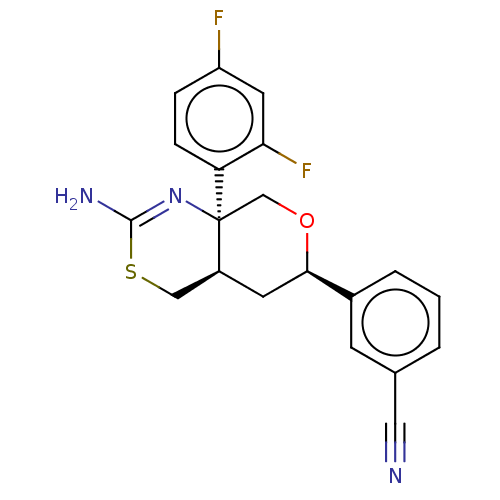
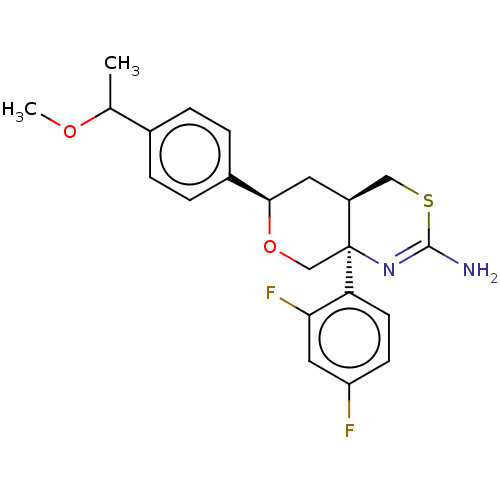
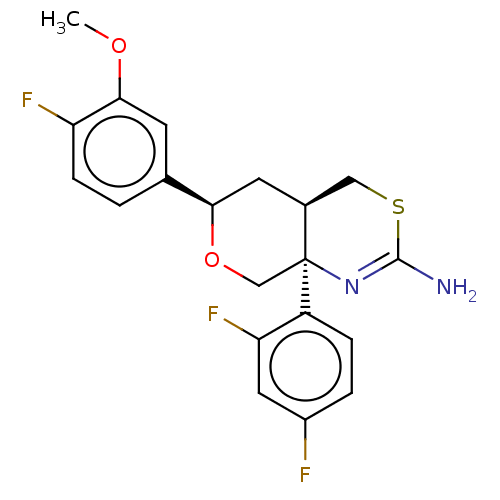
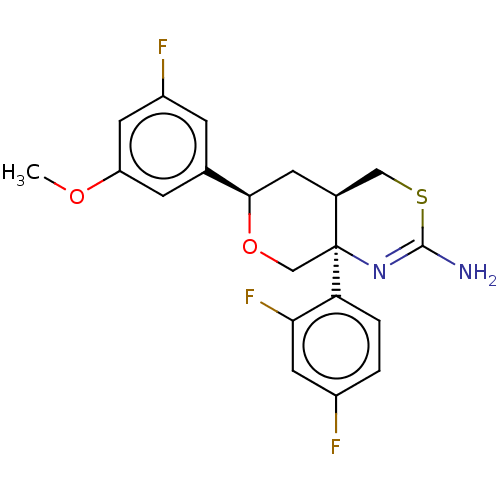
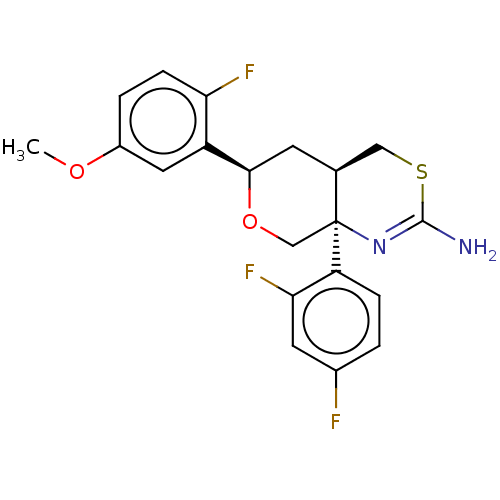
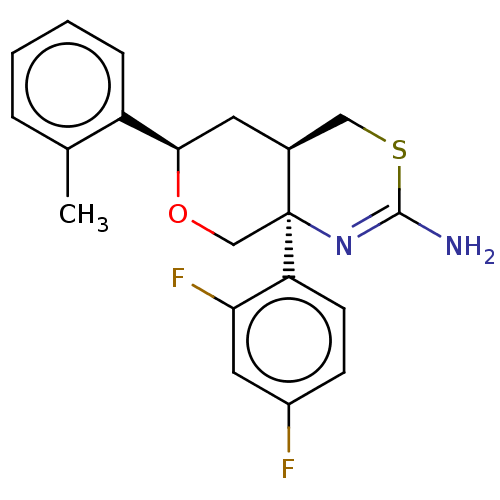
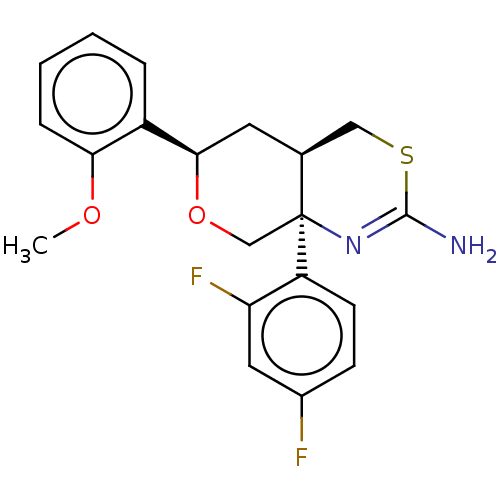
Report error Found 33 Enz. Inhib. hit(s) with all data for entry = 7553
Affinity DataIC50: 6nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 19nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 21nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 21nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 22nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 22nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 24nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 28nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 29nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 39nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 42nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 44nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 83nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 86nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 97nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 99nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 161nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 210nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 214nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 335nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 397nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 970nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 2.21E+3nMpH: 4.5 T: 2°CAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair