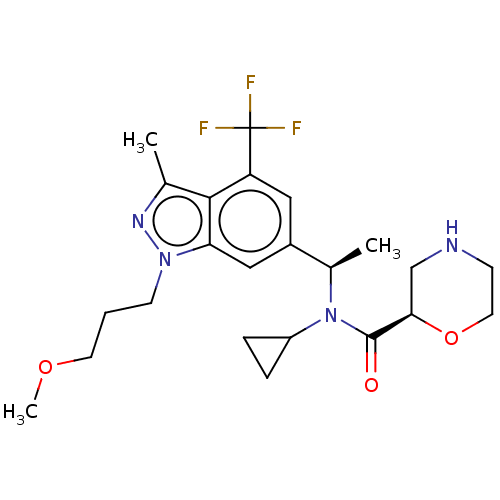
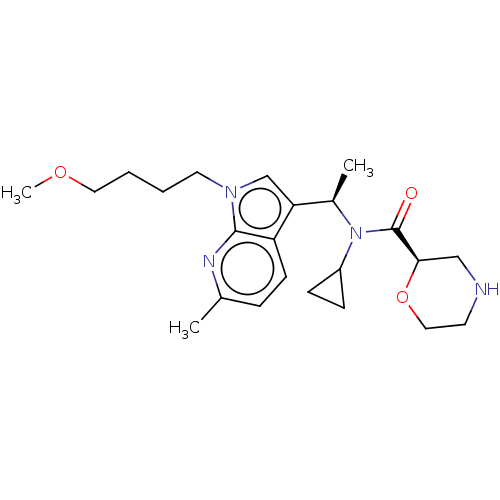
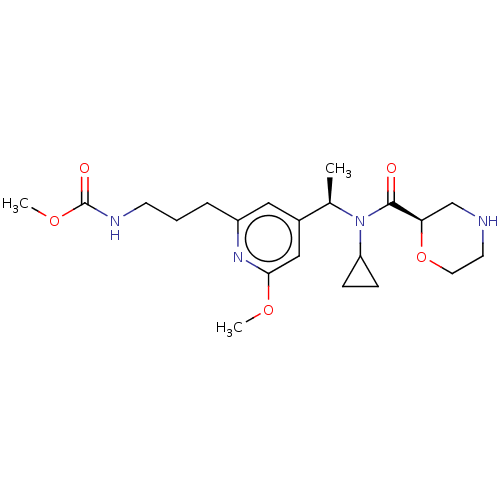
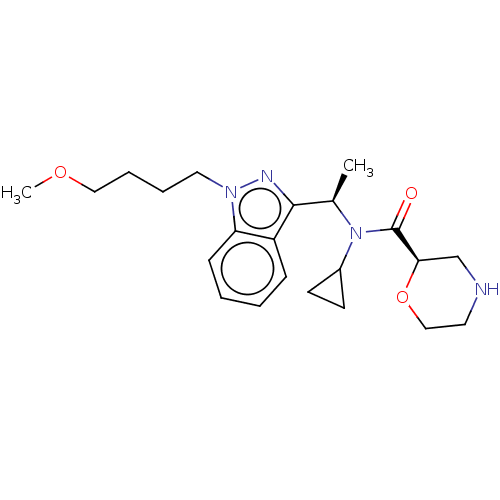
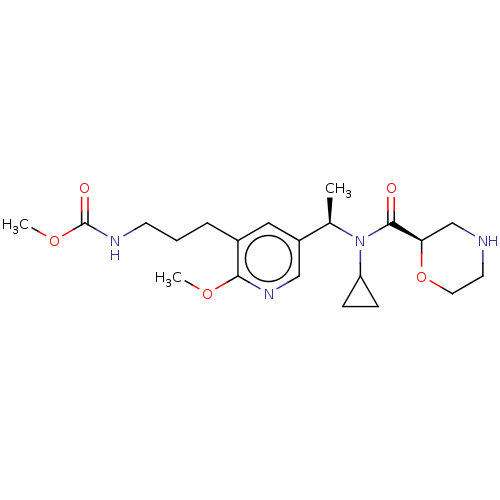
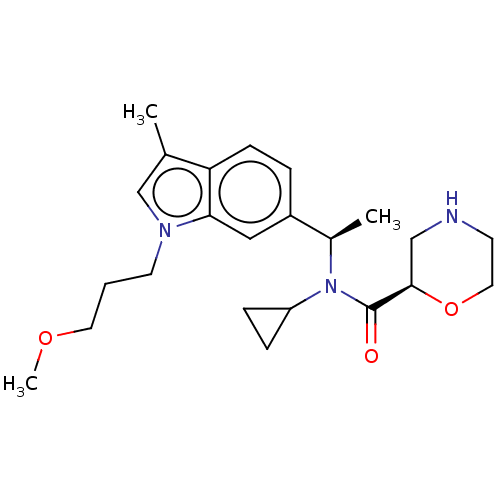
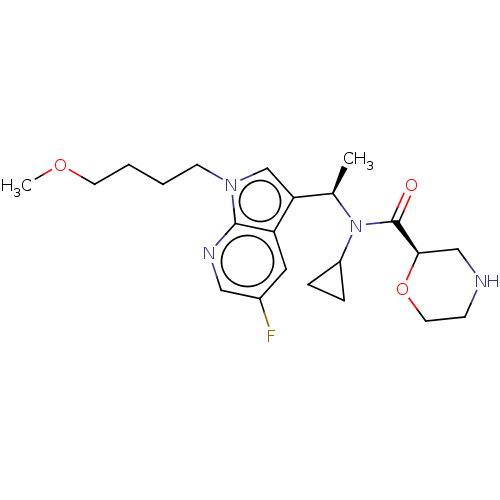
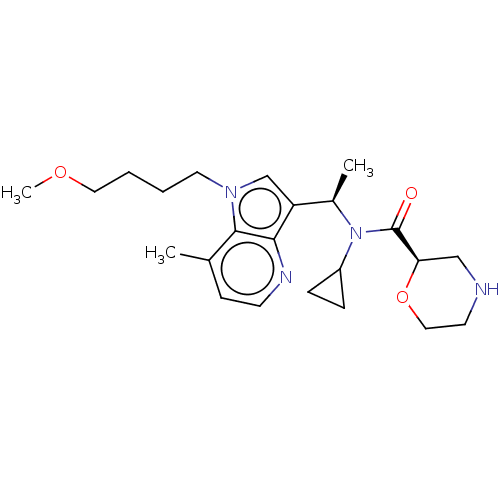
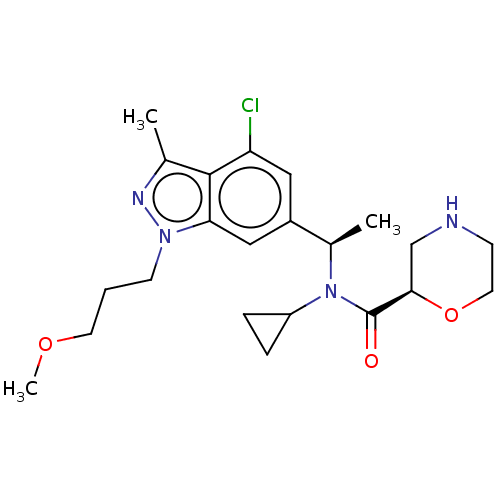
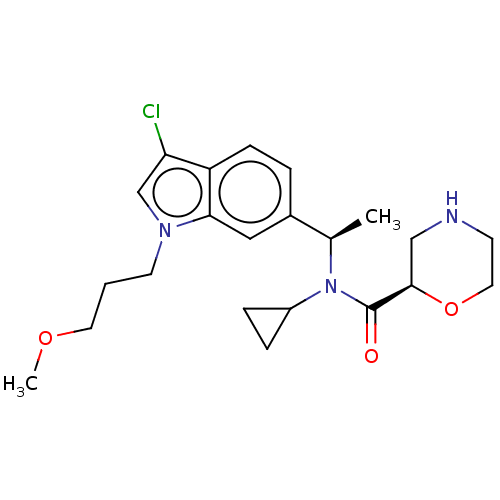
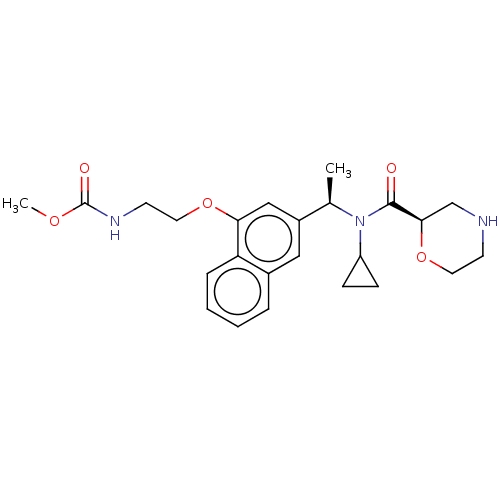
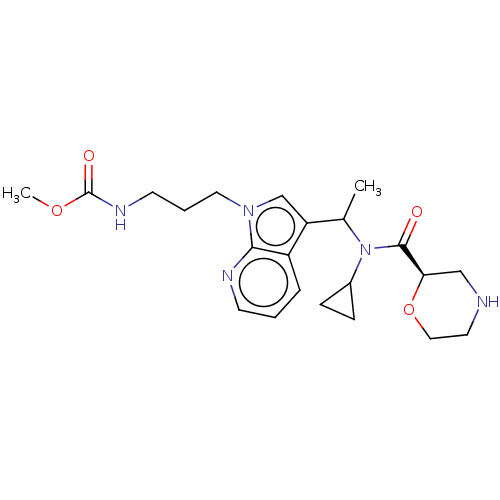
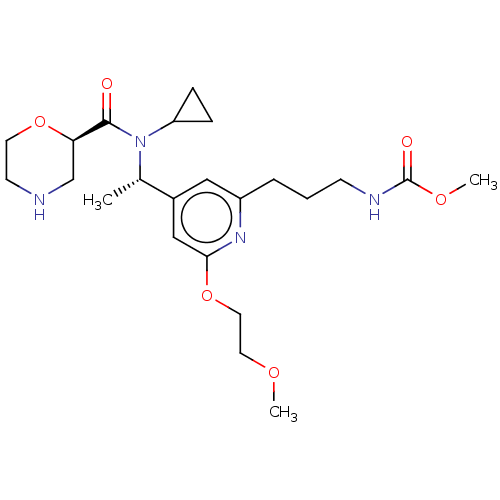
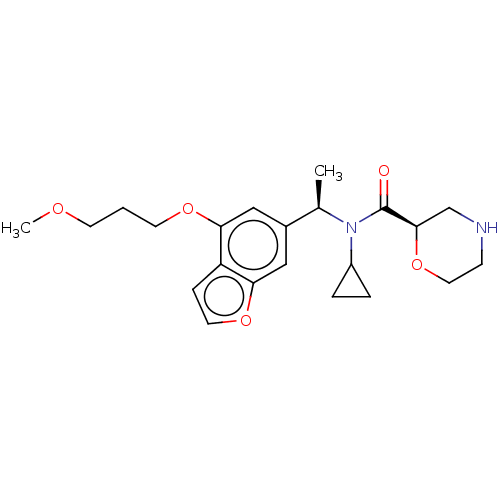
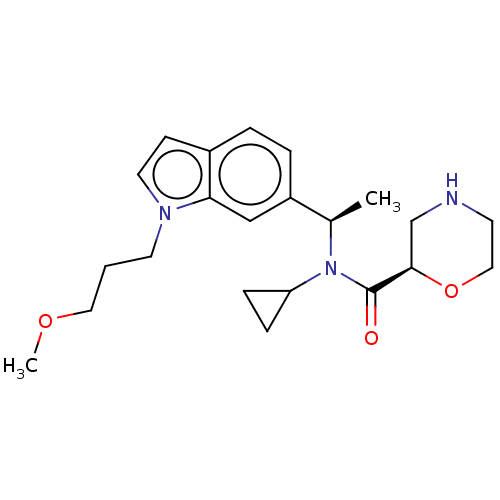
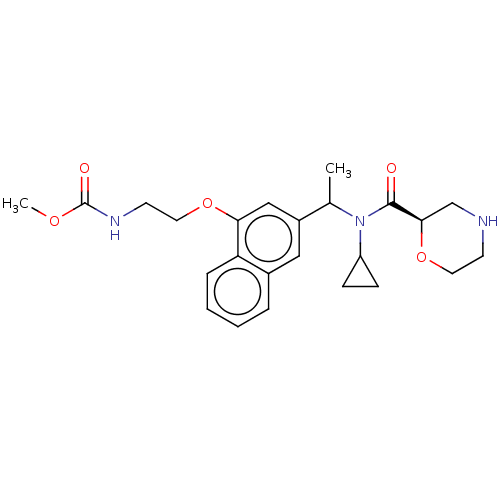
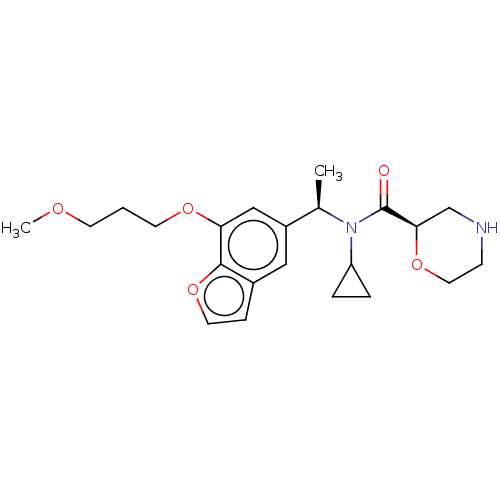
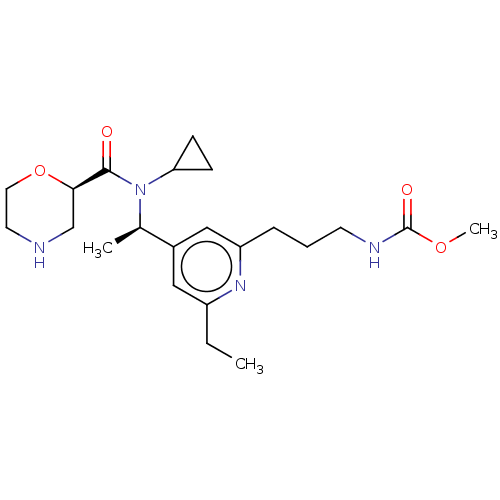
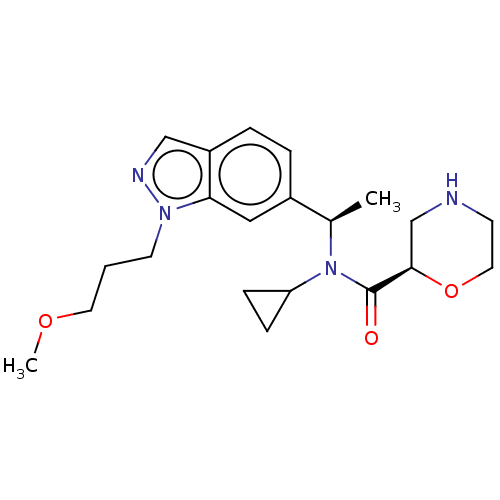
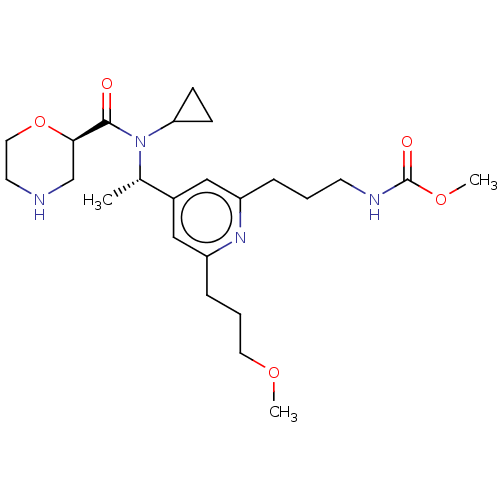
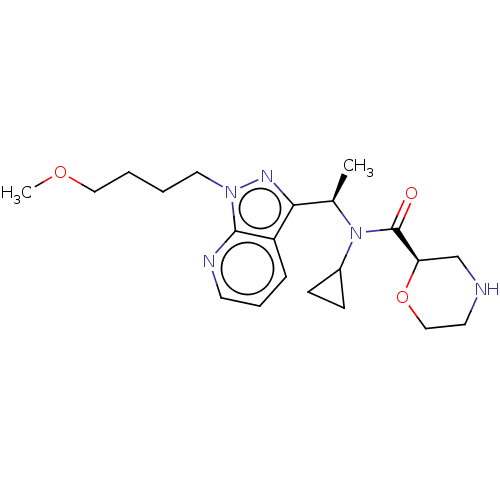
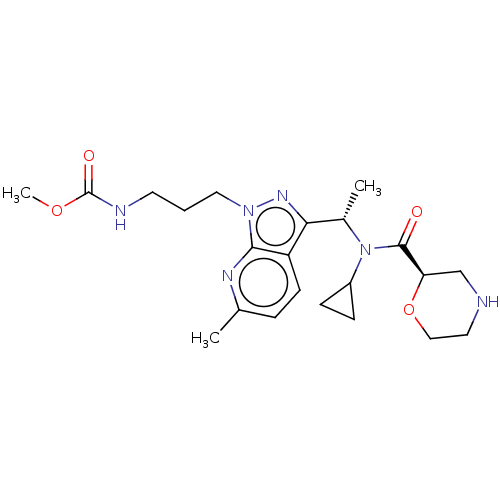
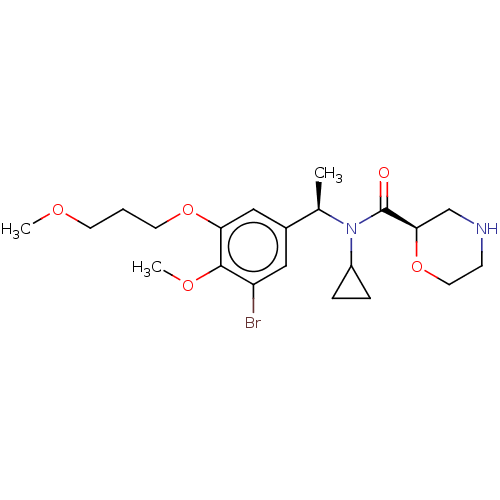
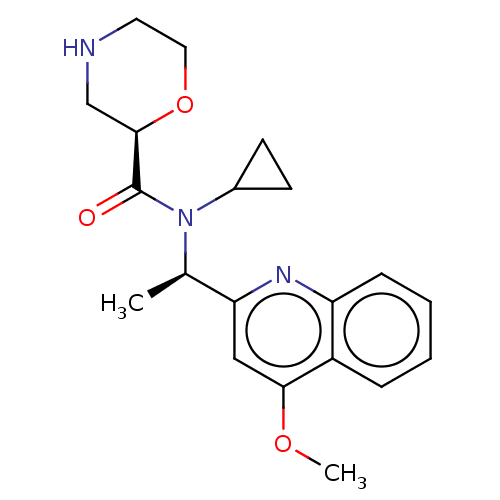
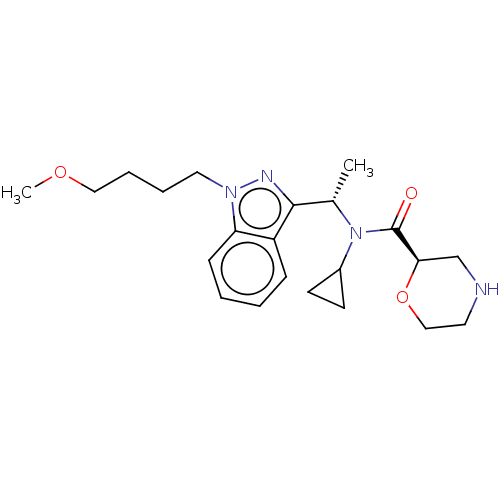
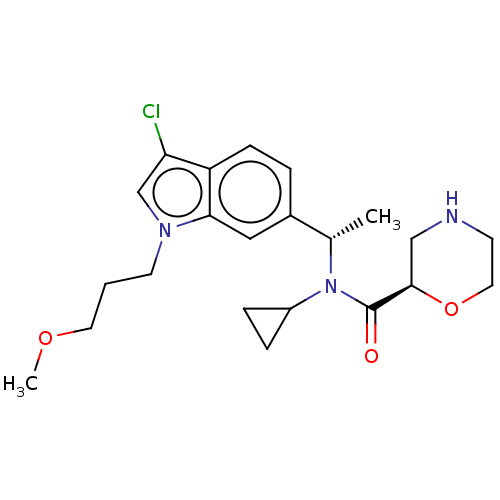
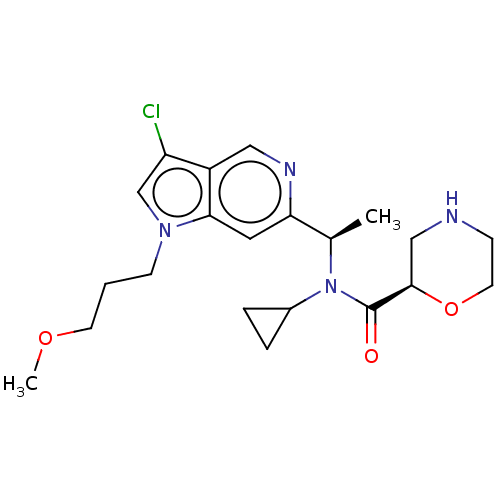
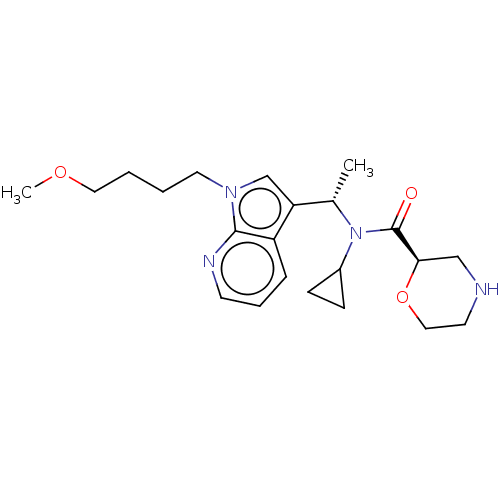
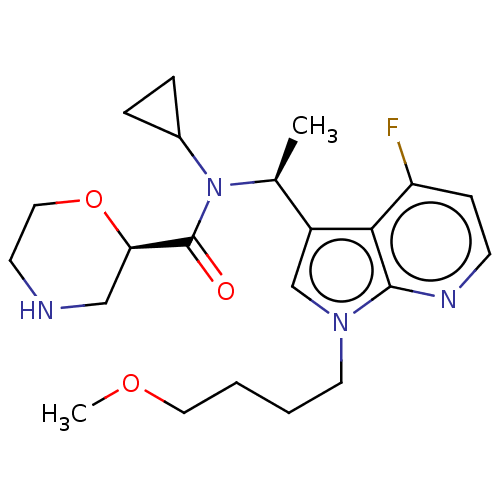
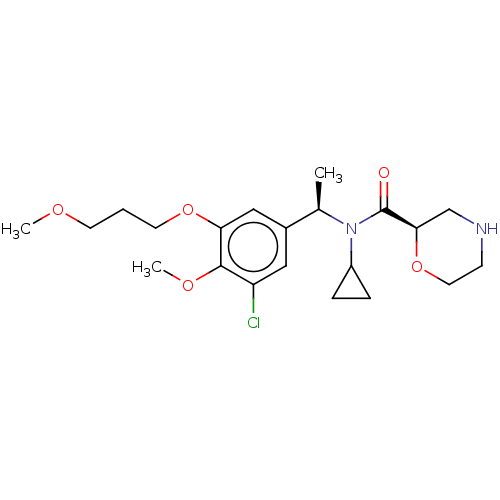
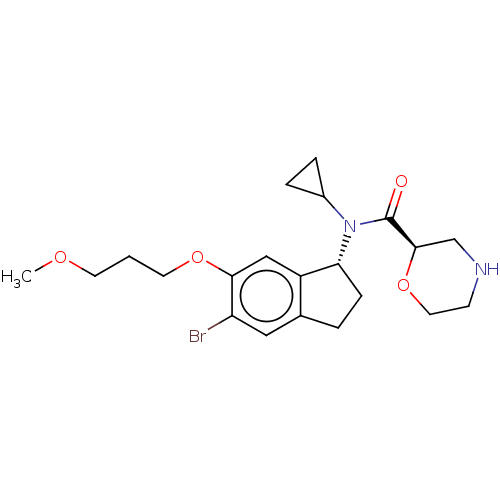
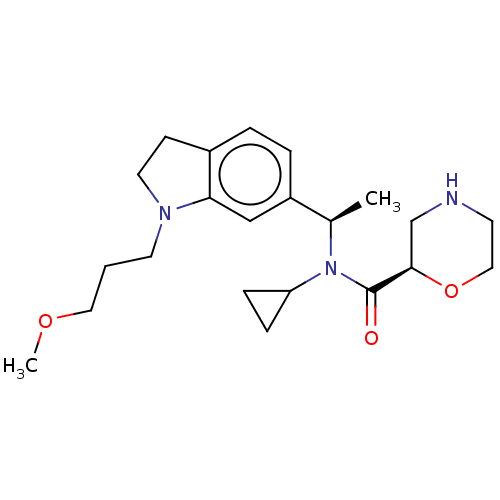
Report error Found 131 Enz. Inhib. hit(s) with all data for entry = 7717
Affinity DataIC50: 0.400nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 0.400nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 2.40nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 2.40nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 3.30nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 3.30nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 3.60nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 3.70nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 3.70nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 3.90nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 4.10nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 4.20nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 4.40nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 4.80nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 5.90nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 6.10nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 6.30nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 6.40nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 6.60nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 6.60nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 6.60nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 6.70nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 6.80nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:A substrate of synthetic peptide (Nma-KHPFHLVIHK(Dnp)-NH2) and test compound were mixed, and fluorescence intensity was assayed using a fluorophotome...More data for this Ligand-Target Pair