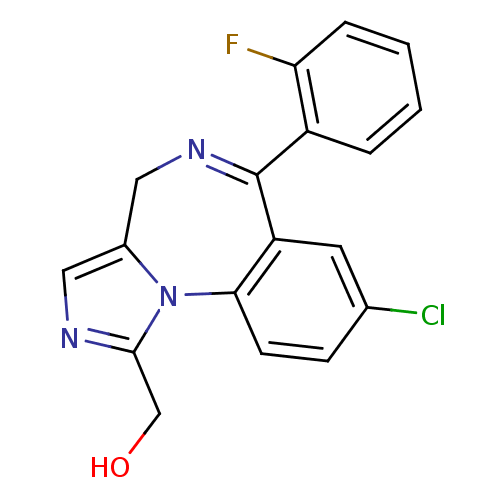
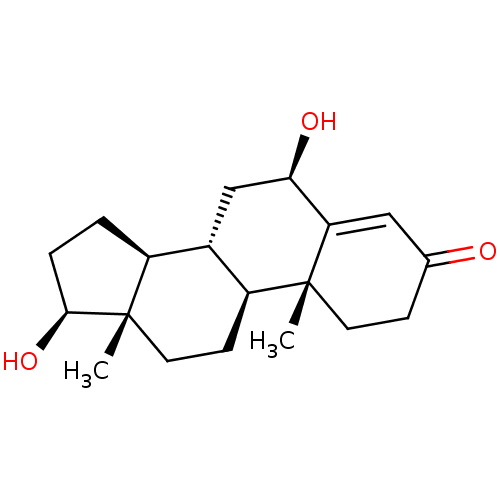
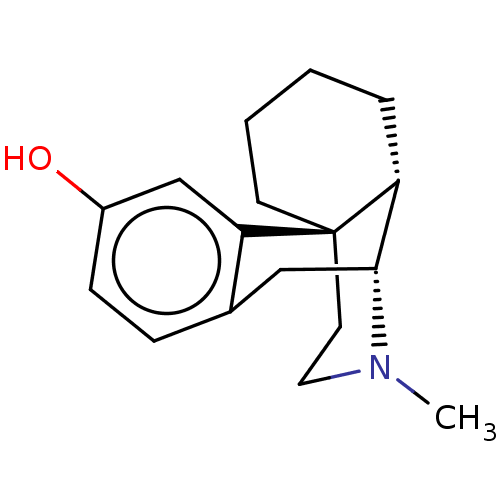
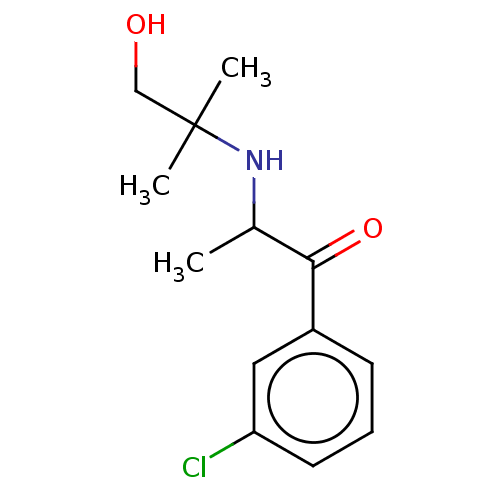
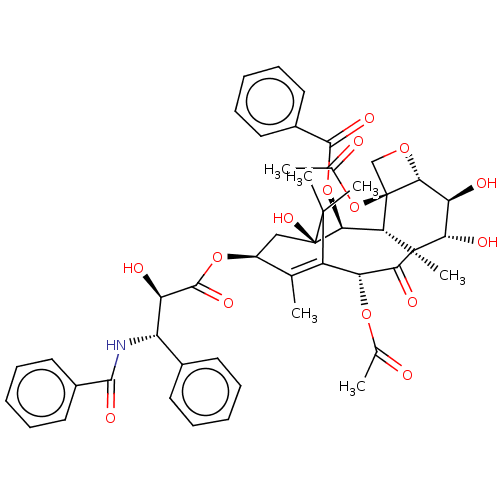
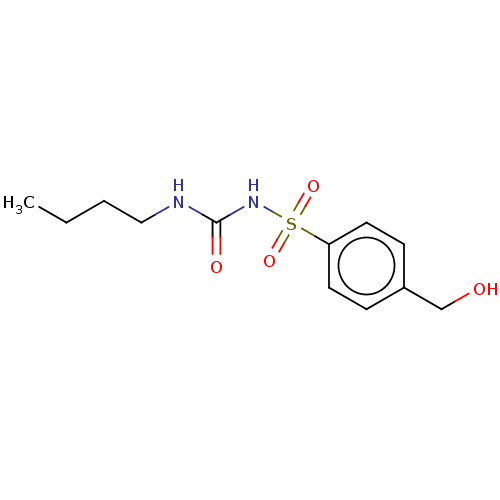
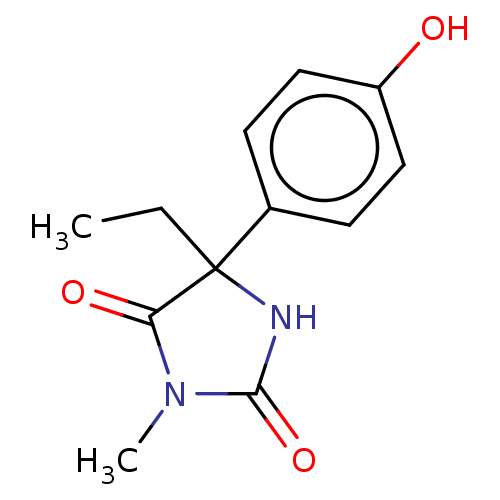
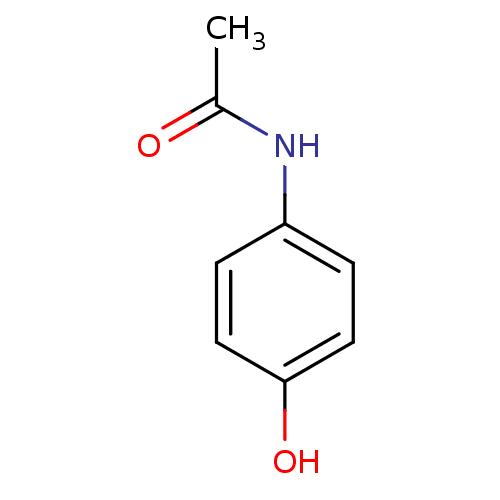
Report error Found 9 Enz. Inhib. hit(s) with all data for entry = 7942
Affinity DataIC50: 2.70E+3nMAssay Description:Six test compound concentrations (0.1, 0.25, 1, 2.5, 10, 25 μM in DMSO; final DMSO concentration=0.26%) are incubated with human liver microsome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.05E+4nMAssay Description:Six test compound concentrations (0.1, 0.25, 1, 2.5, 10, 25 μM in DMSO; final DMSO concentration=0.275%) are incubated with human liver microsom...More data for this Ligand-Target Pair
Affinity DataIC50: 1.77E+4nMAssay Description:Six test compound concentrations (0.1, 0.25, 1, 2.5, 10, 25 μM in DMSO; final DMSO concentration=0.25%) are incubated with human liver microsome...More data for this Ligand-Target Pair
Affinity DataIC50: 1.92E+4nMAssay Description:Six test compound concentrations (0.1, 0.25, 1, 2.5, 10, 25 μM in DMSO; final DMSO concentration=0.3%) are incubated with human liver microsomes...More data for this Ligand-Target Pair
Affinity DataIC50: 2.16E+4nMAssay Description:Six test compound concentrations (0.1, 0.25, 1, 2.5, 10, 25 μM in DMSO; final DMSO concentration=0.3%) are incubated with human liver microsomes...More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Six test compound concentrations (0.1, 0.25, 1, 2.5, 10, 25 μM in DMSO; final DMSO concentration=0.25%) are incubated with human liver microsome...More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Six test compound concentrations (0.1, 0.25, 1, 2.5, 10, 25 μM in DMSO; final DMSO concentration=0.25%) are incubated with human liver microsome...More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Six test compound concentrations (0.1, 0.25, 1, 2.5, 10, 25 μM in DMSO; final DMSO concentration=0.3%) are incubated with human liver microsomes...More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Six test compound concentrations (0.1, 0.25, 1, 2.5, 10, 25 μM in DMSO; final DMSO concentration=0.3%) are incubated with human liver microsomes...More data for this Ligand-Target Pair