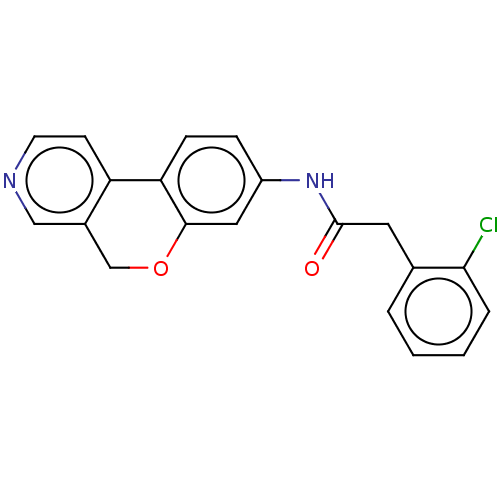
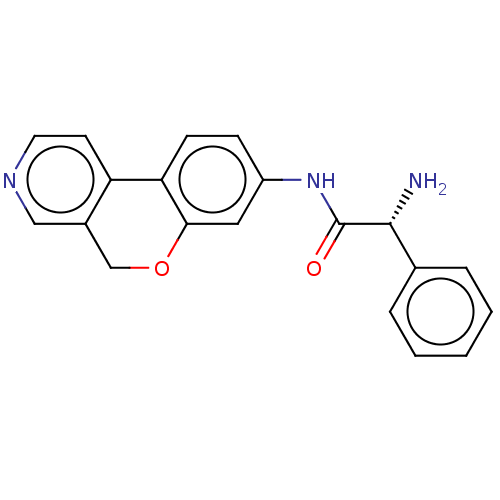
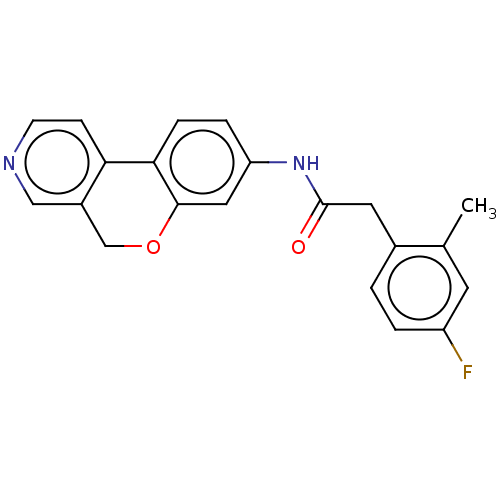
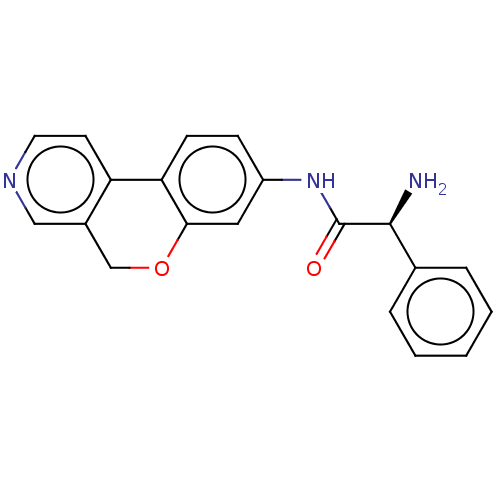
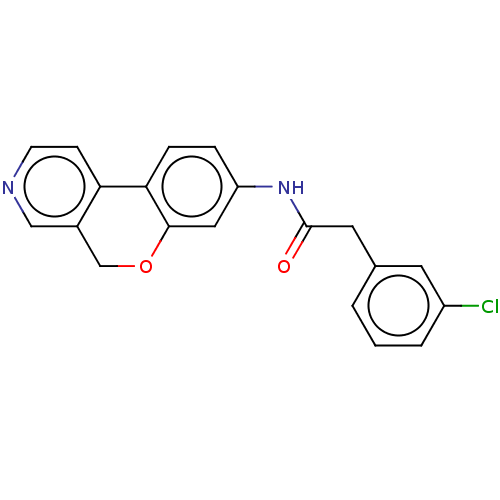
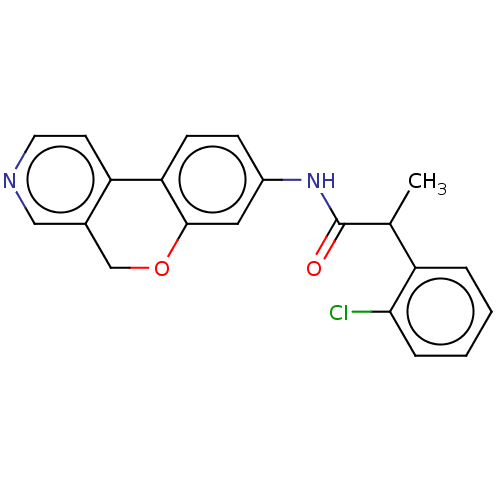
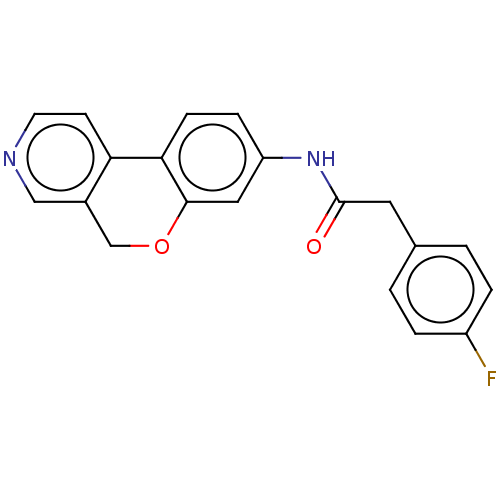
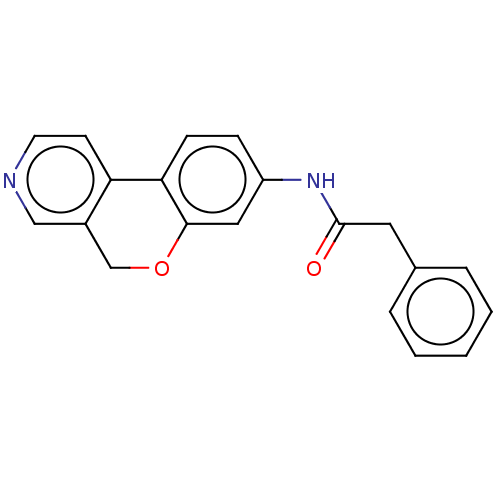
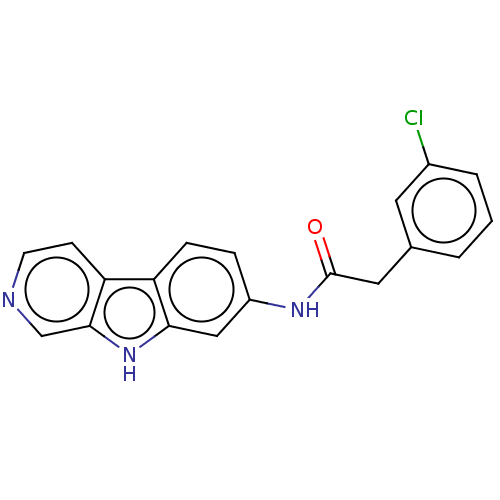
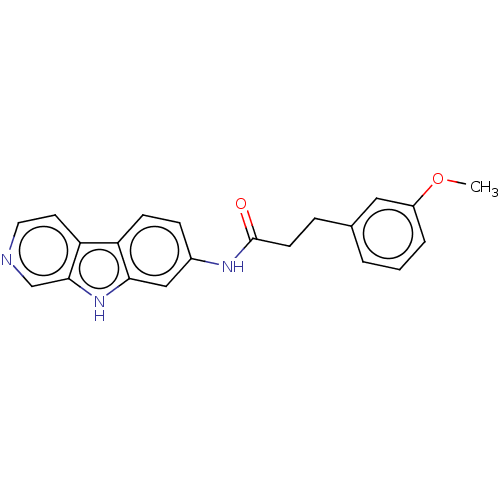
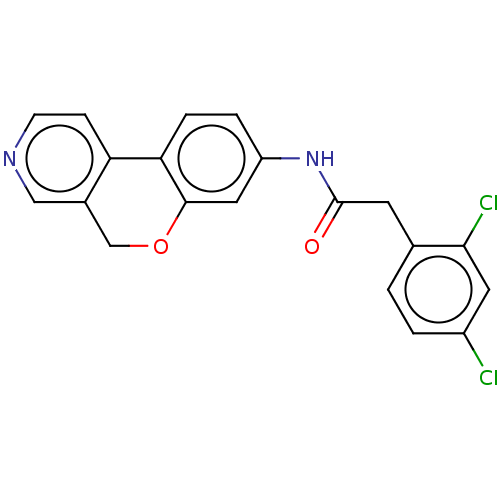
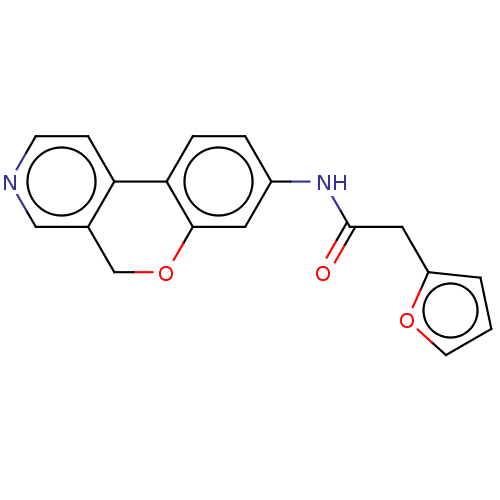
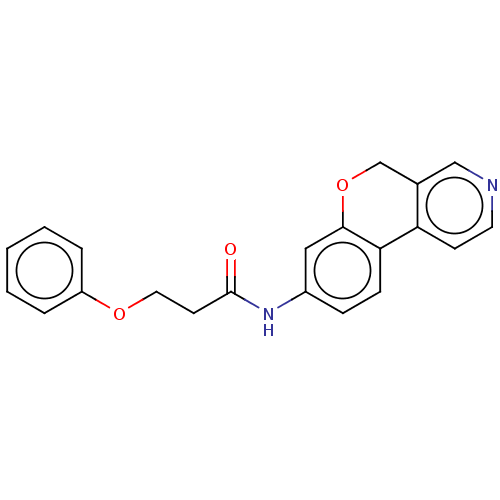
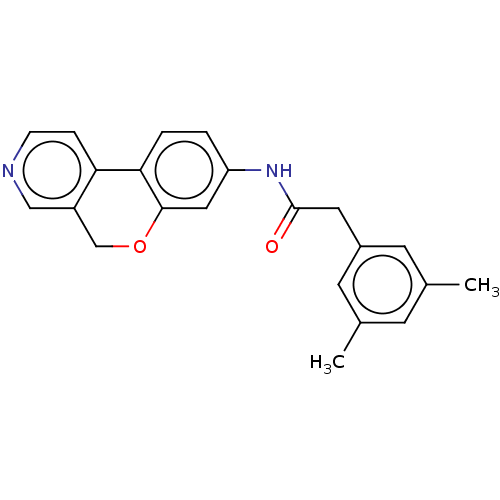
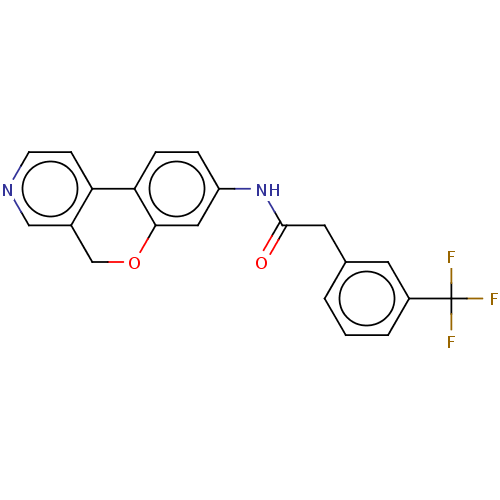
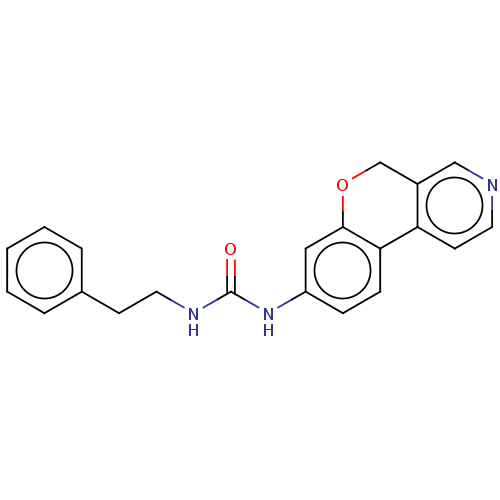
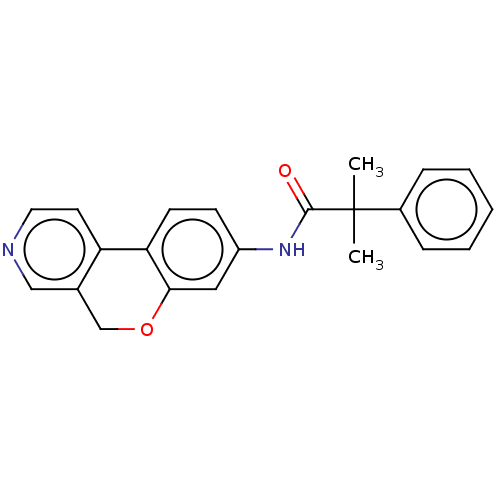
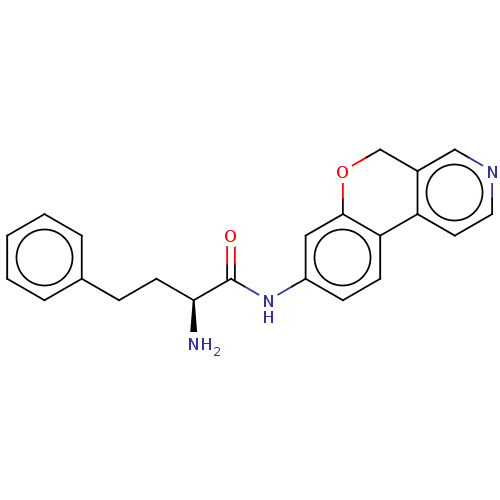
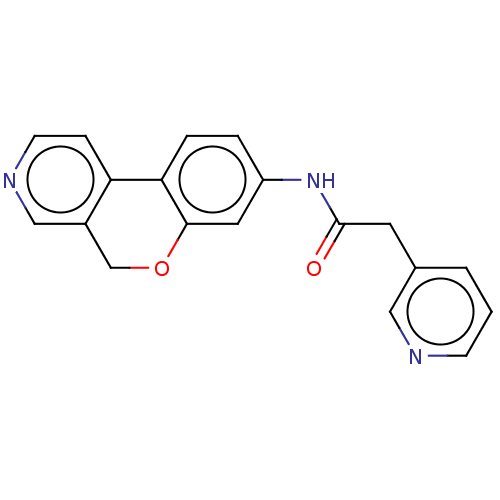
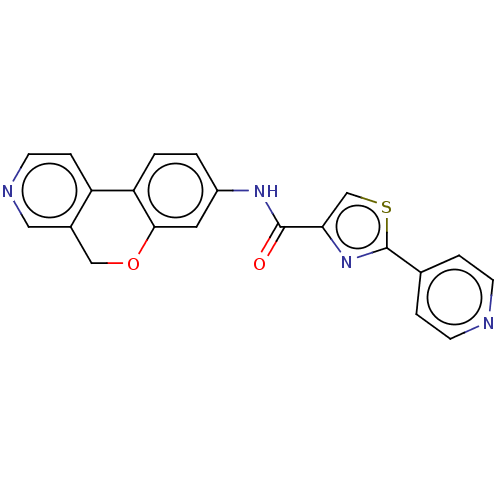
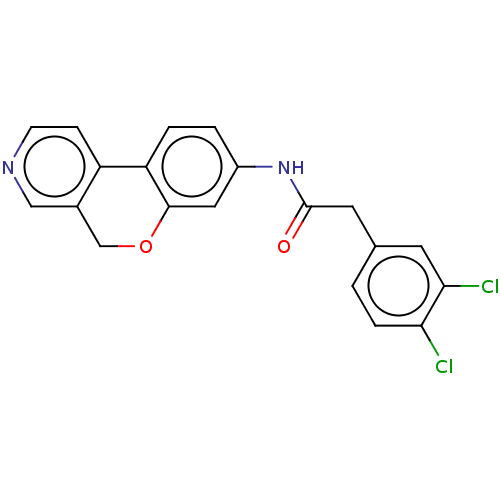
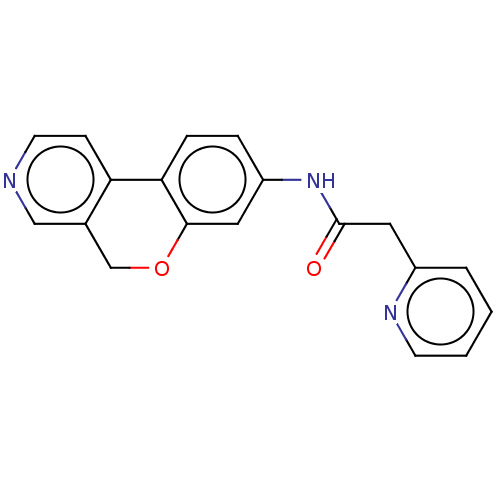
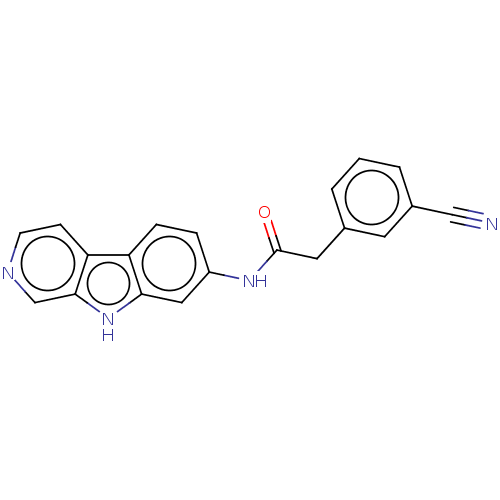
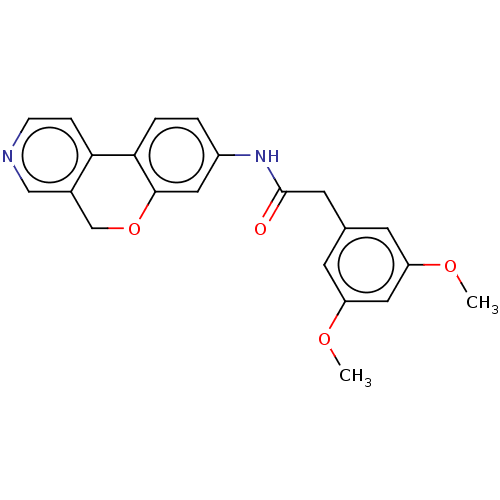
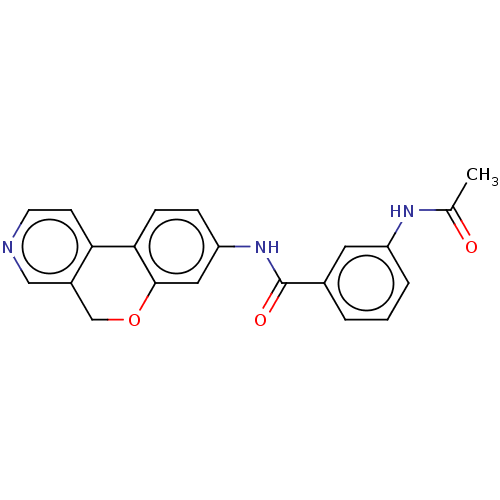
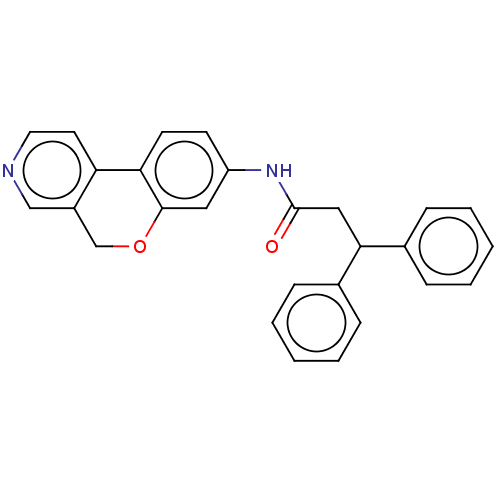
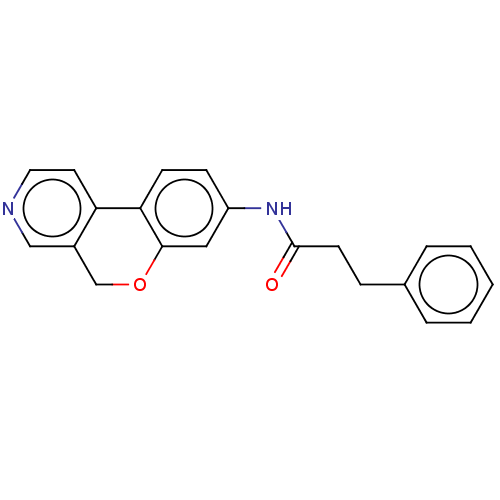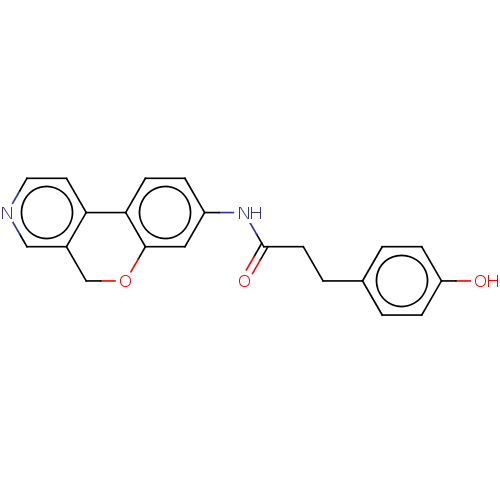Report error Found 96 Enz. Inhib. hit(s) with all data for entry = 1534
Affinity DataIC50: 5.97nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 6.27nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 9.43nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 9.5nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 11.1nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 16.7nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 19.9nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 21nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 21.8nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 24.8nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 33.5nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 37.8nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 38.7nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 40.4nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 43.6nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 46.3nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 50.9nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 51.2nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 54.4nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 55.6nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 56.2nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 58.7nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 60.8nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 74.5nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 82.2nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 111nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 128nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 133nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 134nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 159nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 161nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 189nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 204nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 207nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 233nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 248nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 284nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 287nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 295nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 316nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 323nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 362nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 367nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 368nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 383nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 387nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 393nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 434nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair
Affinity DataIC50: 471nMAssay Description:The effectiveness of compounds of the present invention as ROCK inhibitors can be determined in a 30 μL assay containing 20 mM HEPES, pH 7.5, 20...More data for this Ligand-Target Pair