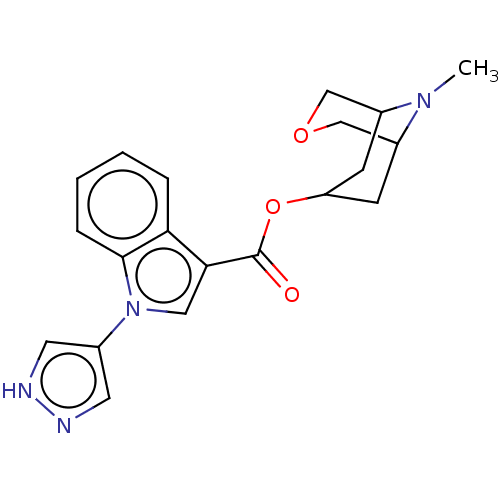
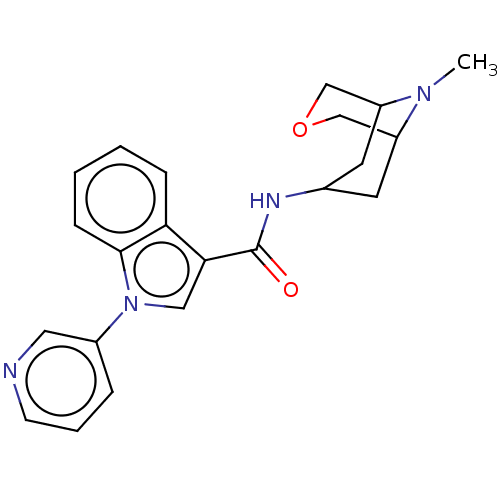
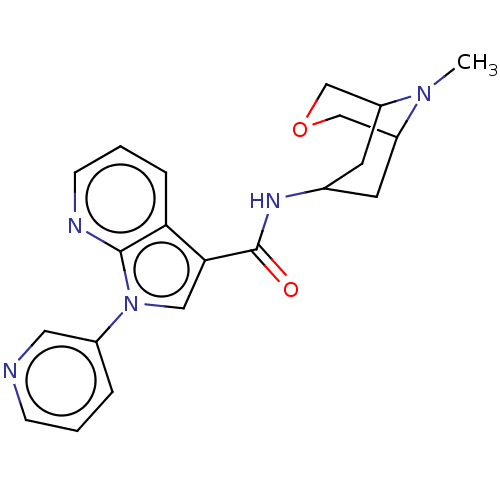
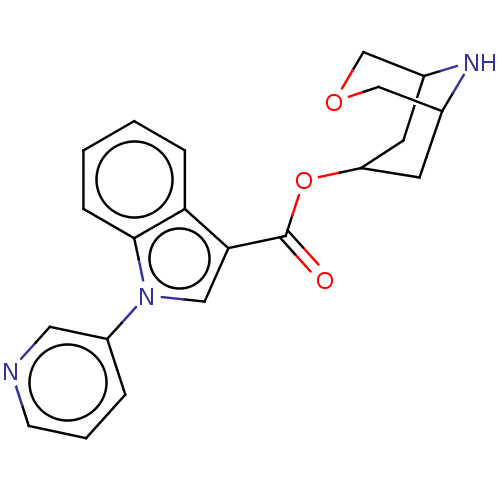
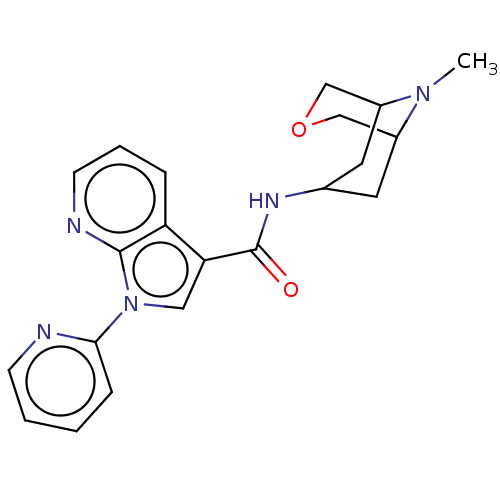
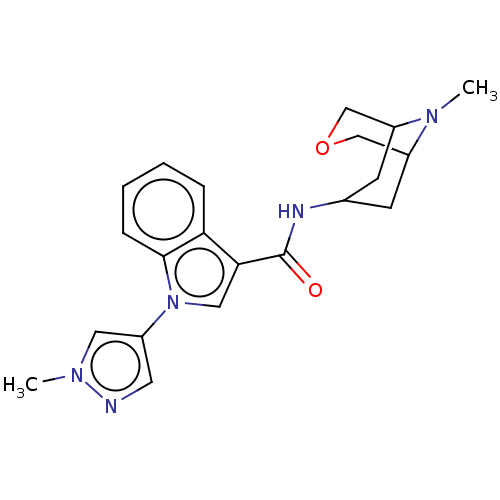
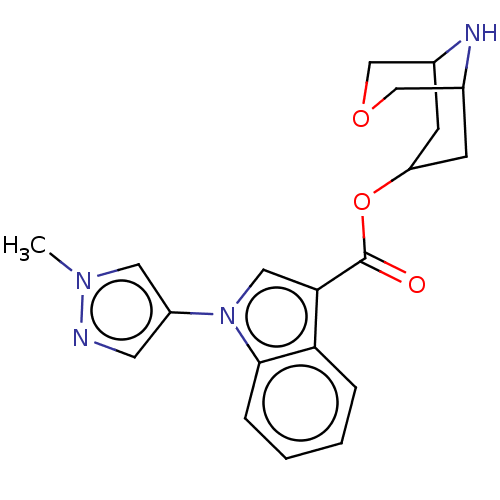
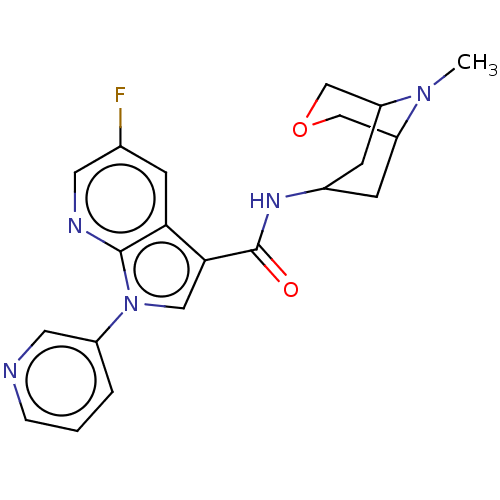
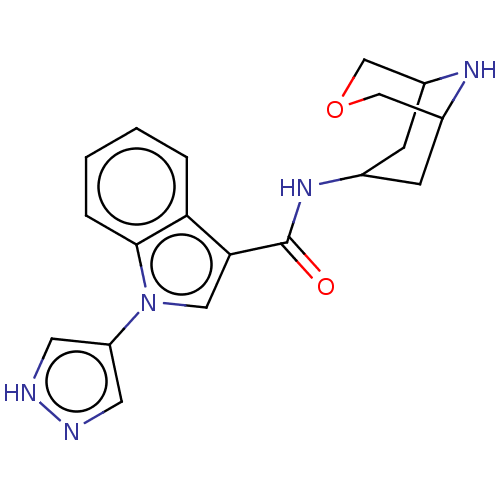
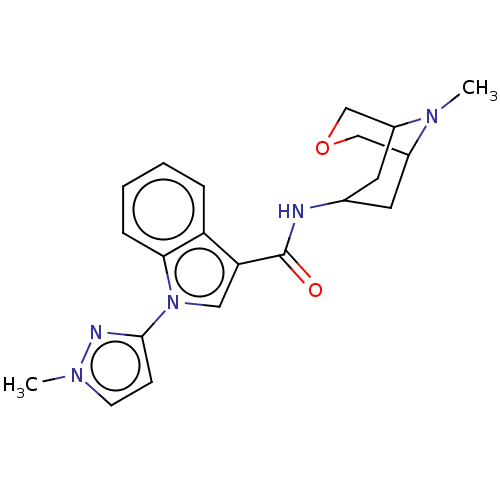
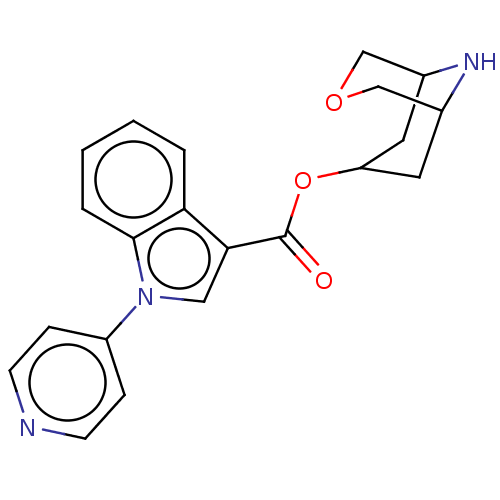
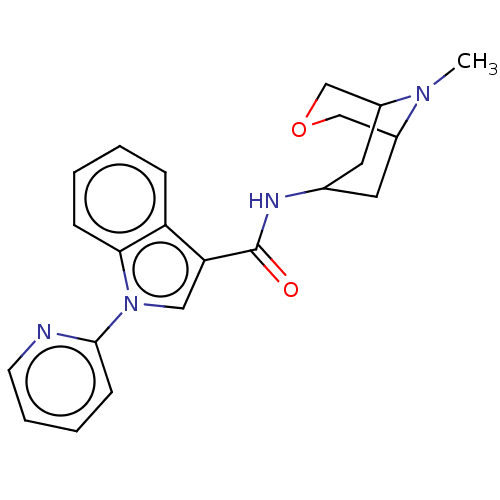
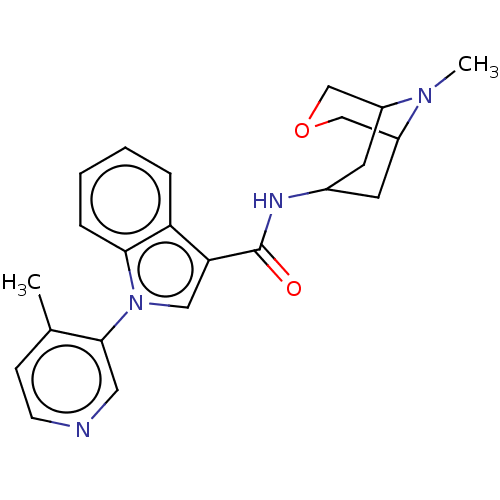
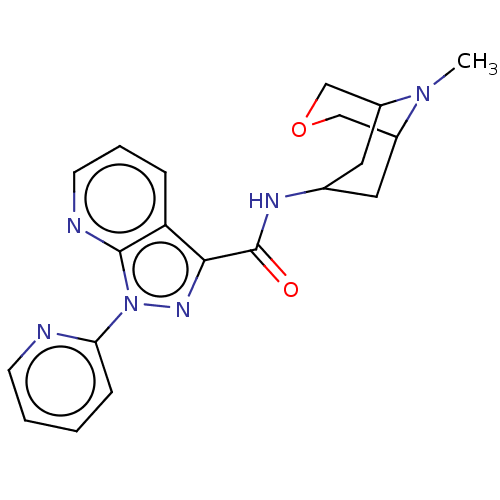
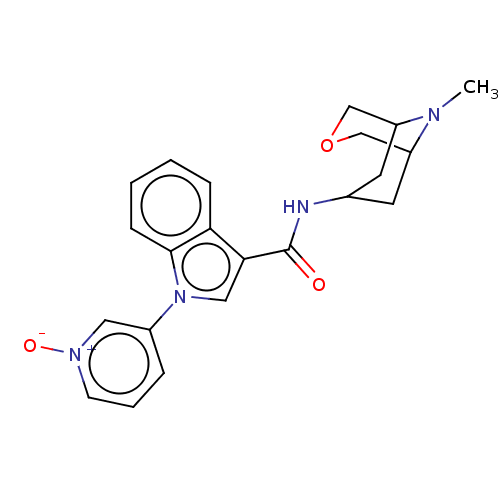
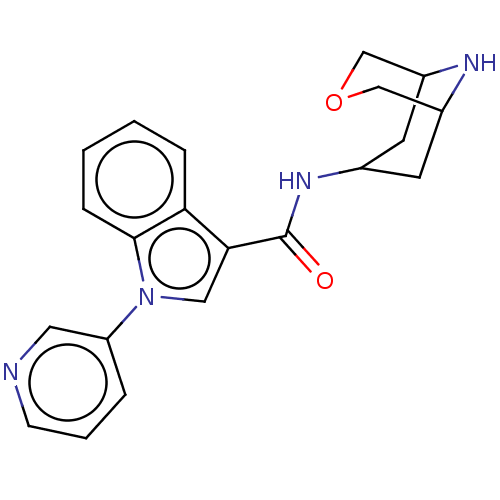
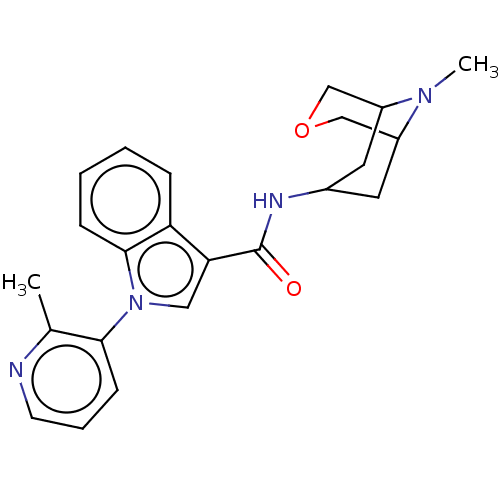
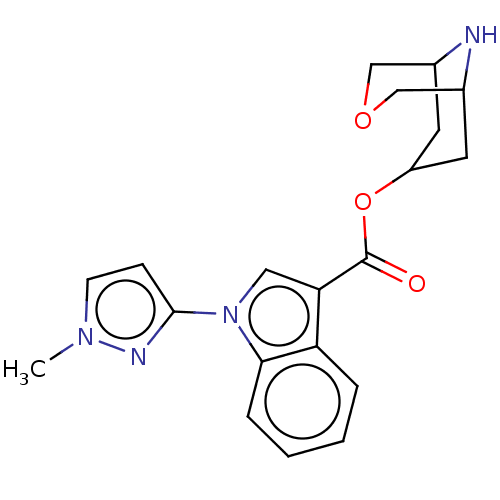
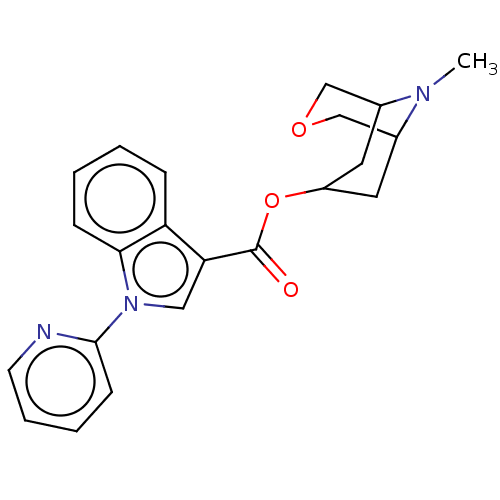
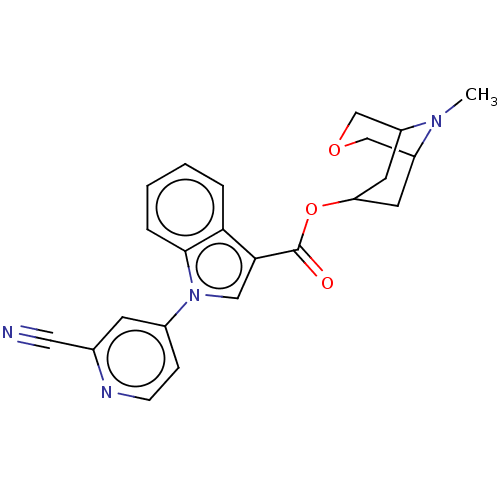
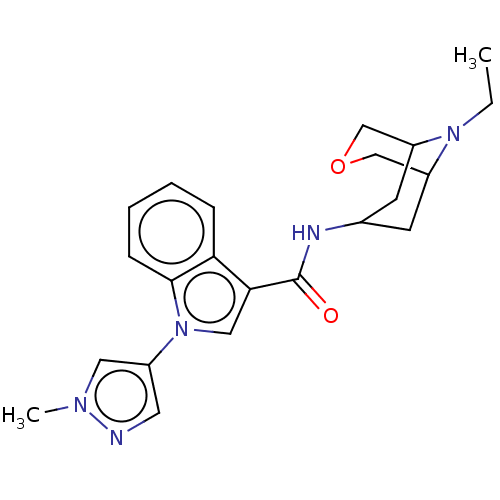
Report error Found 31 Enz. Inhib. hit(s) with all data for entry = 165
Affinity DataIC50: 0.462nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.510nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.620nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.662nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.680nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.690nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.720nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.745nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.75nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.810nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.882nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.950nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.960nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.01nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.34nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.36nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.39nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.44nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.01nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.04nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.86nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 3.68nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMT: 2°CAssay Description:The 5-HT3 antagonist activity of the compounds of the invention was determined by measuring the ability of the compounds to inhibit the calcium flux ...More data for this Ligand-Target Pair