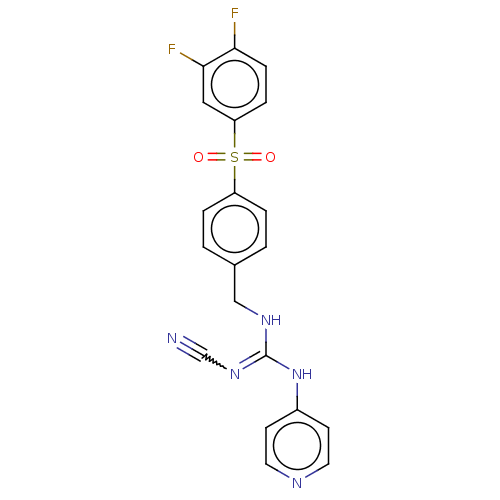
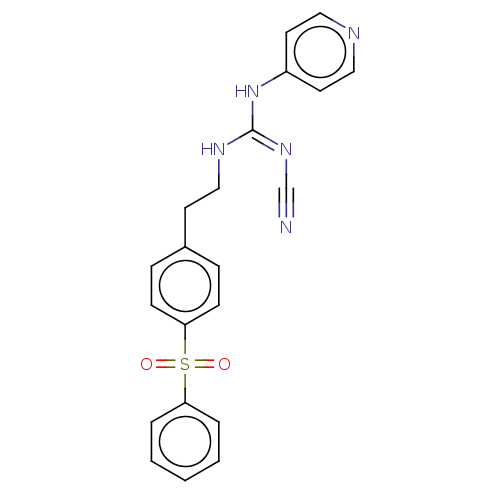
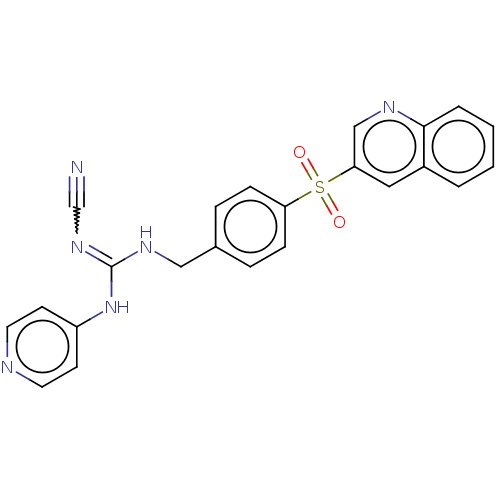
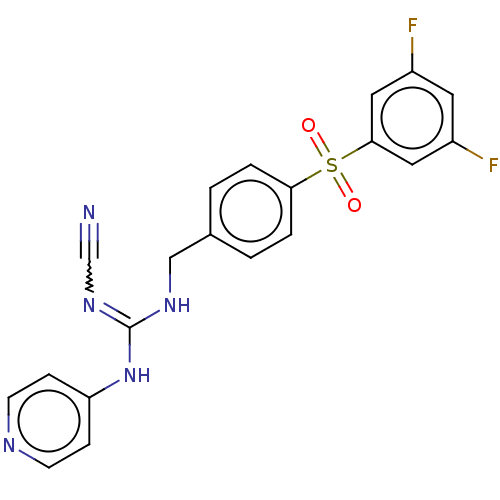
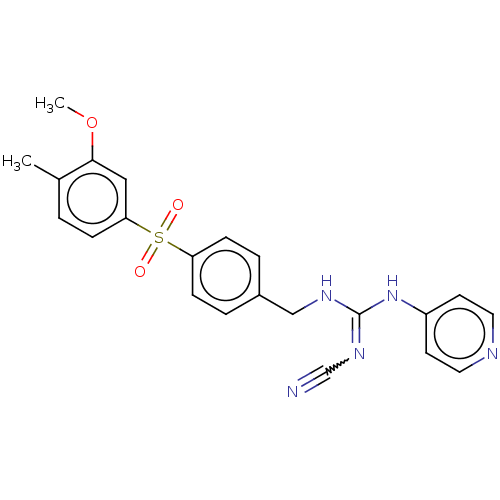
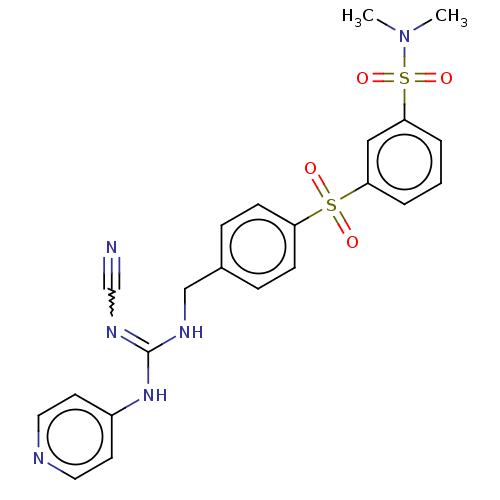
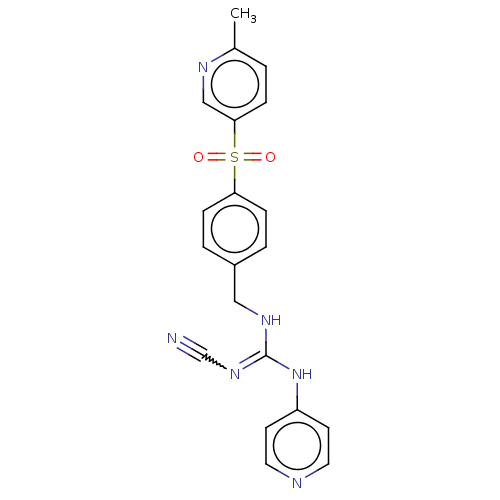
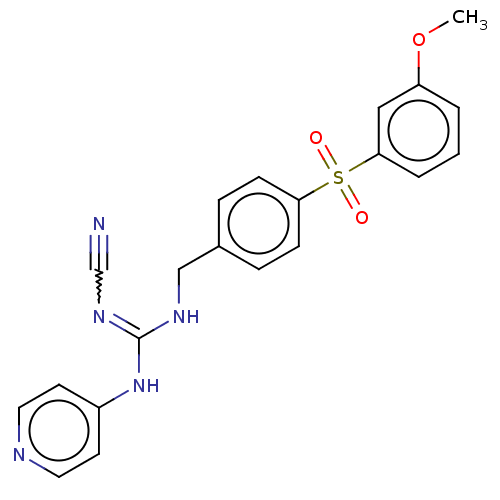
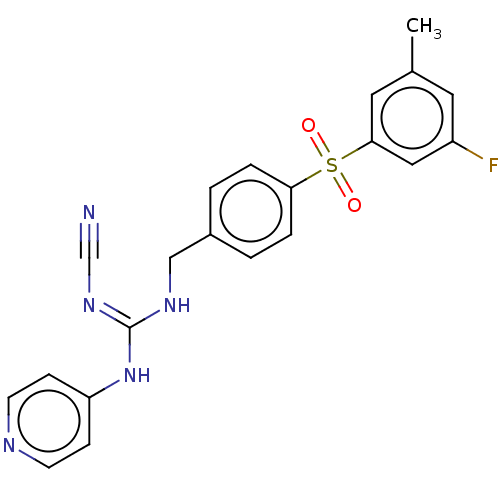
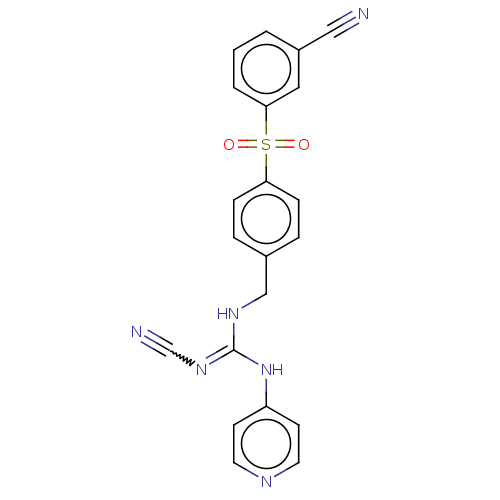
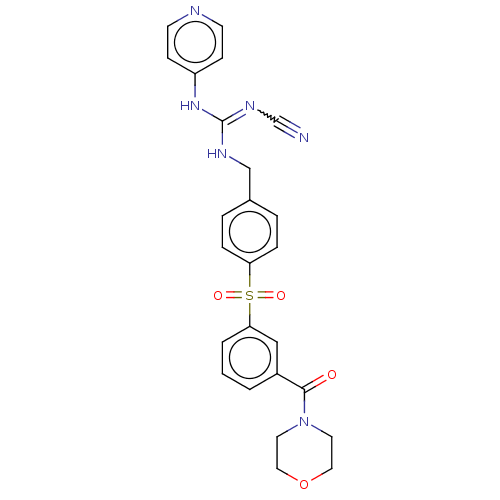
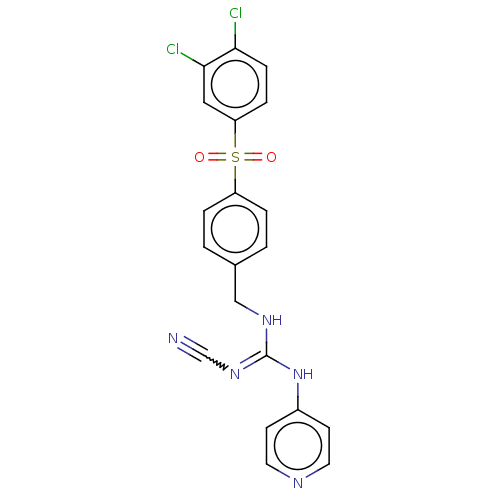
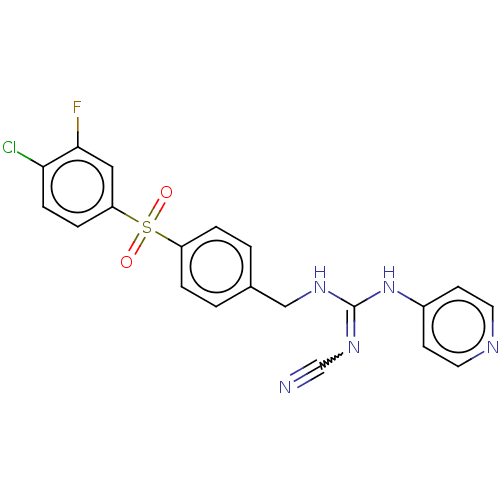
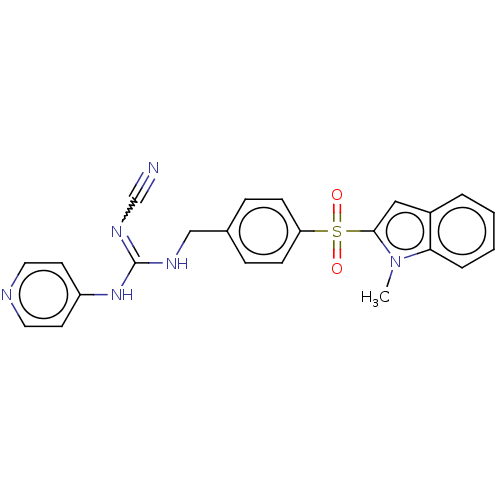
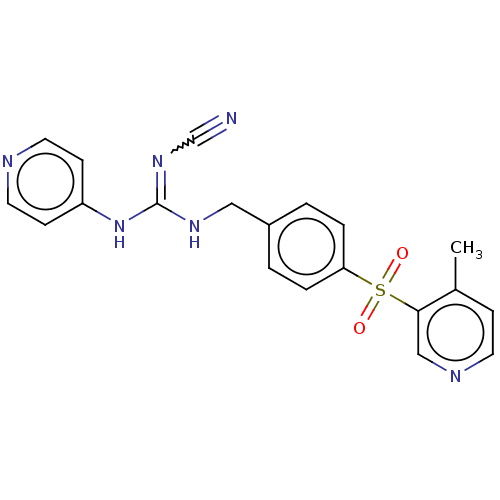
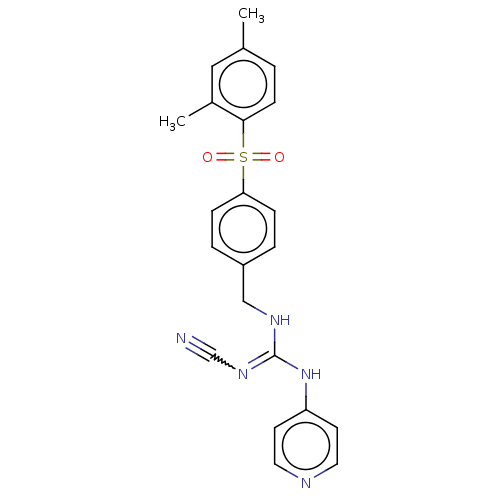
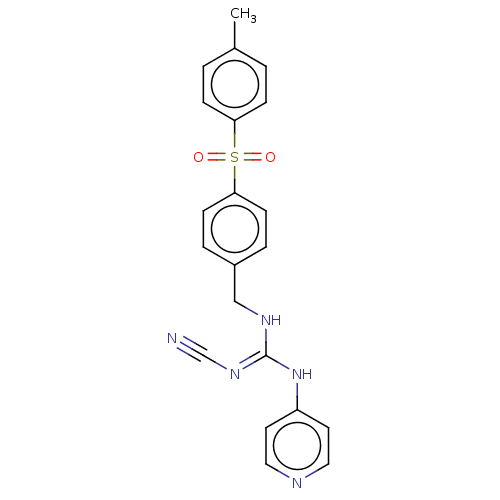
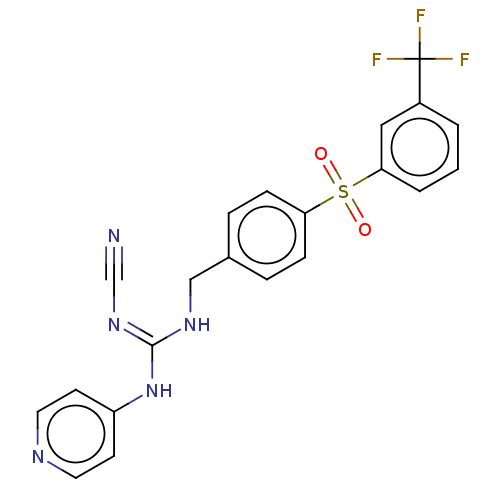
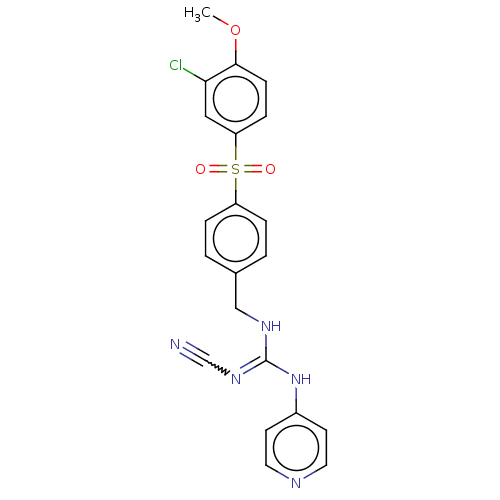
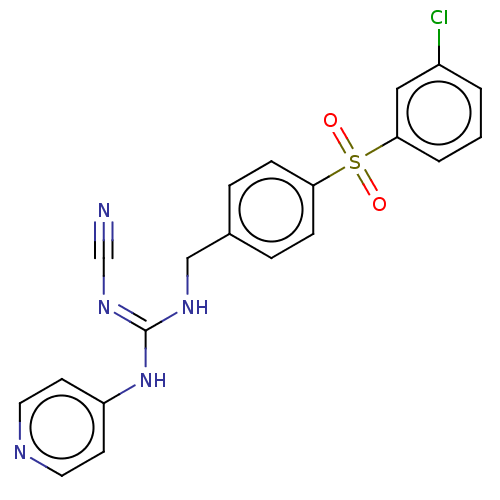
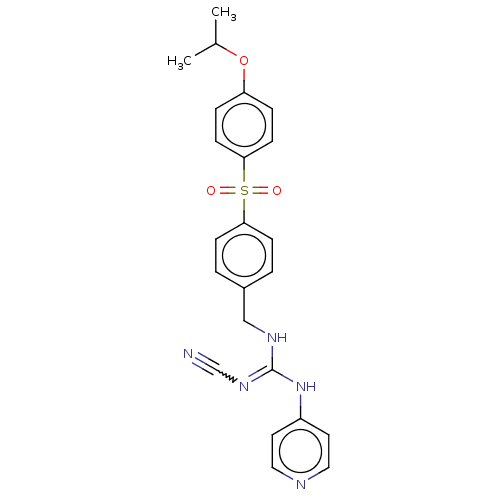
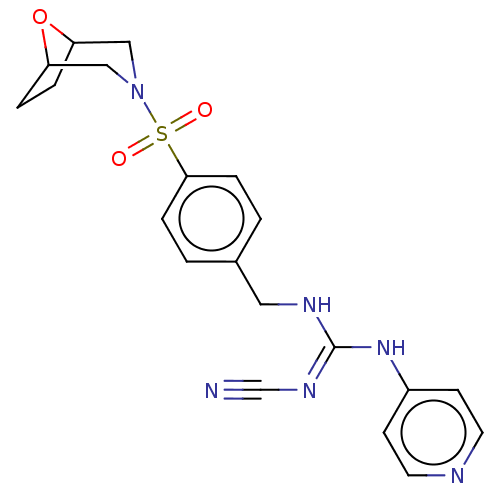
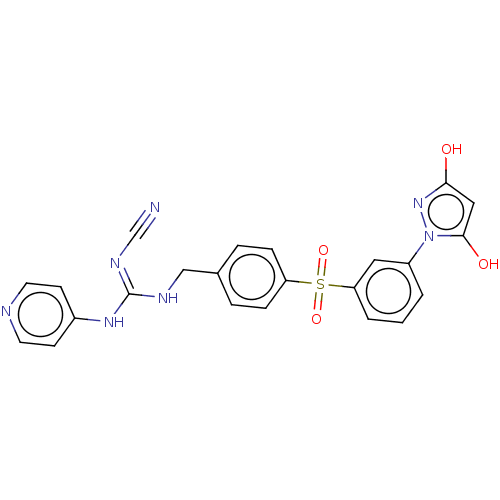
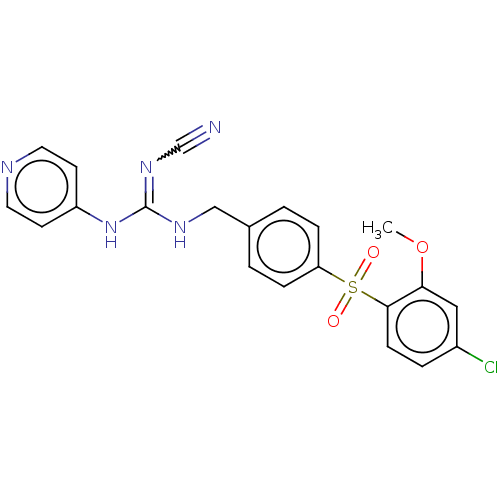
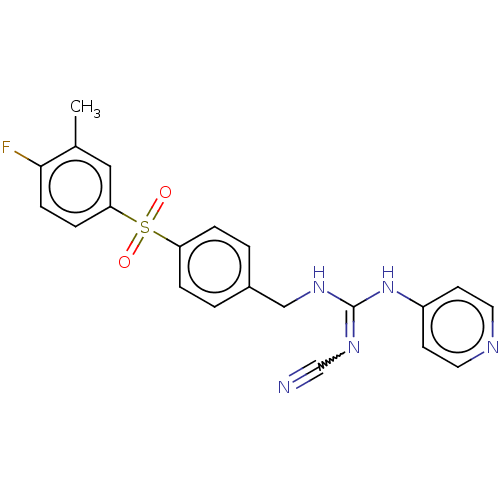
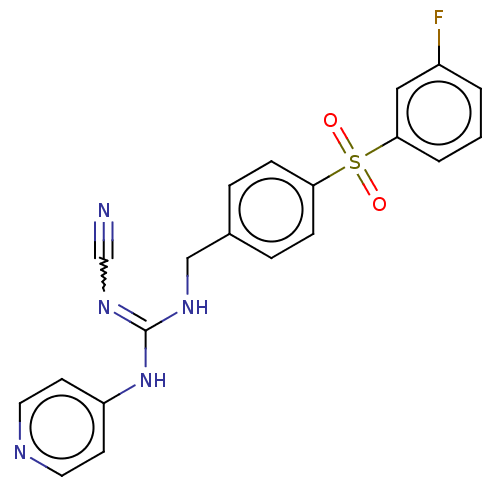
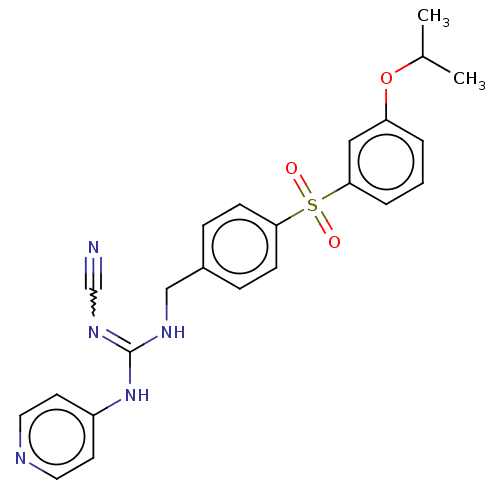
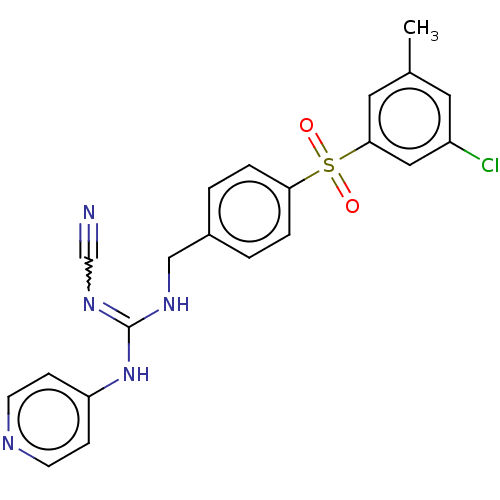
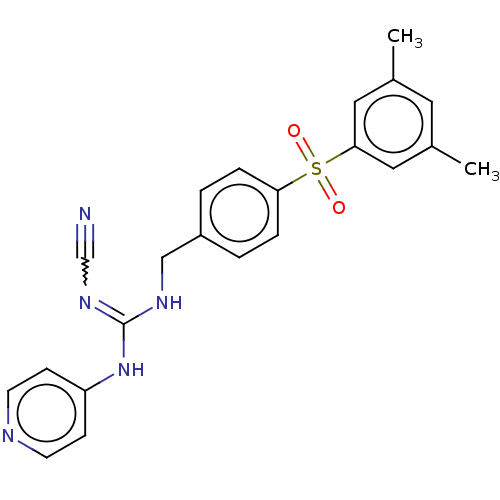
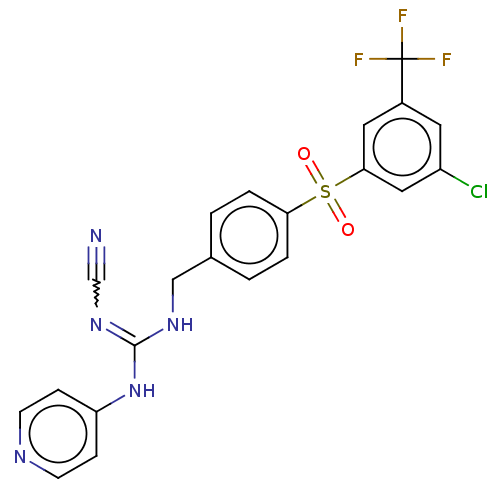
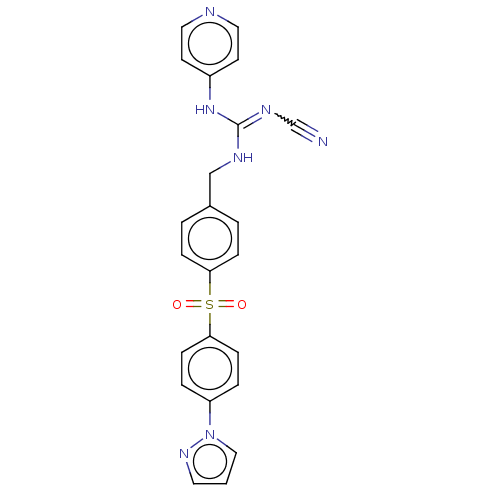
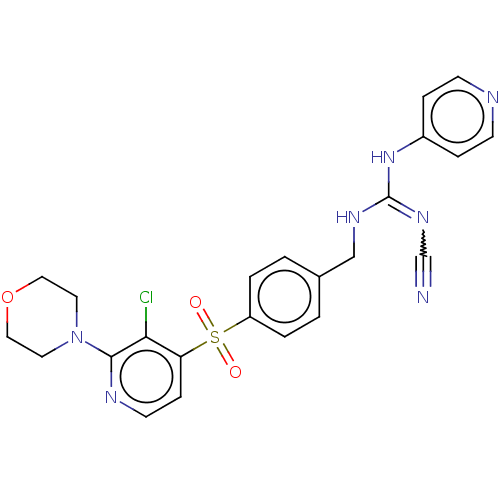
Report error Found 112 Enz. Inhib. hit(s) with all data for entry = 122
Affinity DataIC50: 0.800nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.70nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.70nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.70nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.70nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.40nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.90nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 2.90nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3.10nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3.30nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3.30nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3.30nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3.30nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3.40nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair
Affinity DataIC50: 3.40nMpH: 7.5 T: 2°CAssay Description:The NAMPT enzymatic reactions were carried out in Buffer A (50 mM Hepes pH 7.5, 50 mM NaCl, 5 mM MgCl2, and 1 mM THP) in 96-well V-bottom plates. The...More data for this Ligand-Target Pair