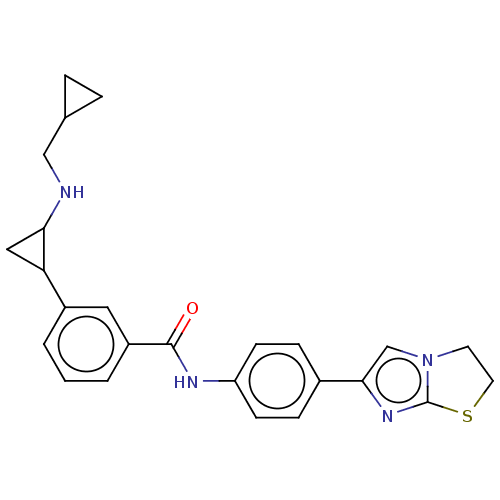
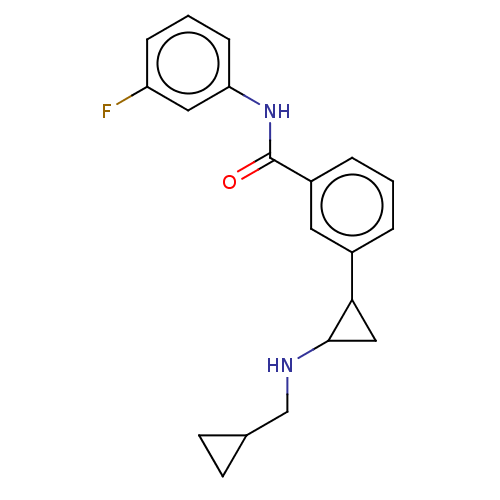
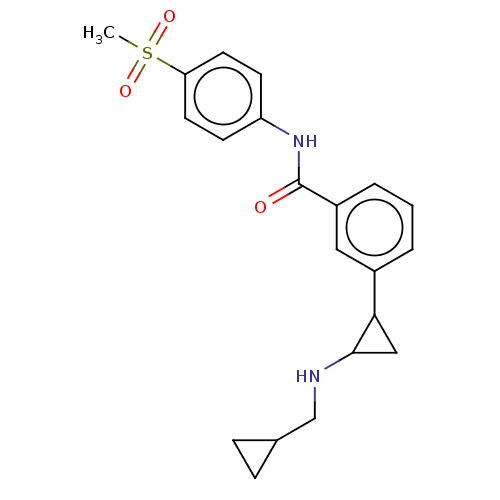
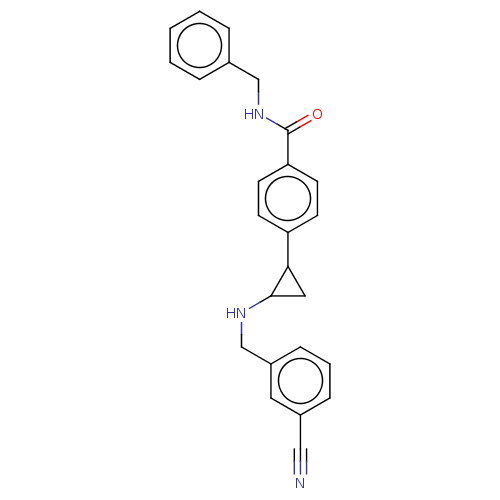
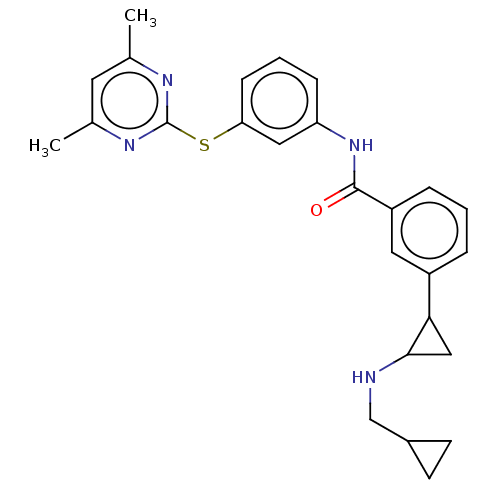
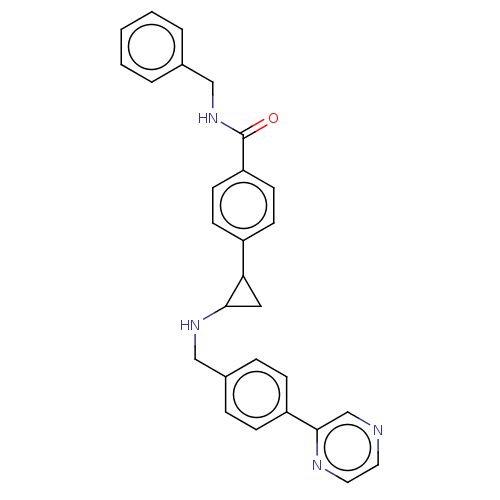
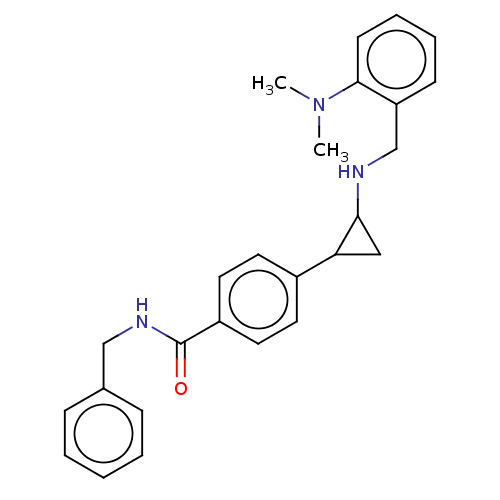
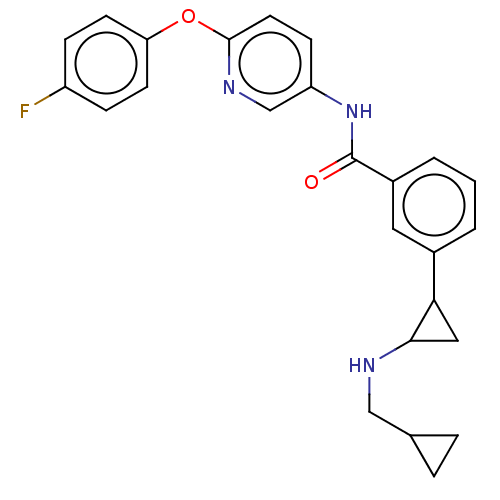
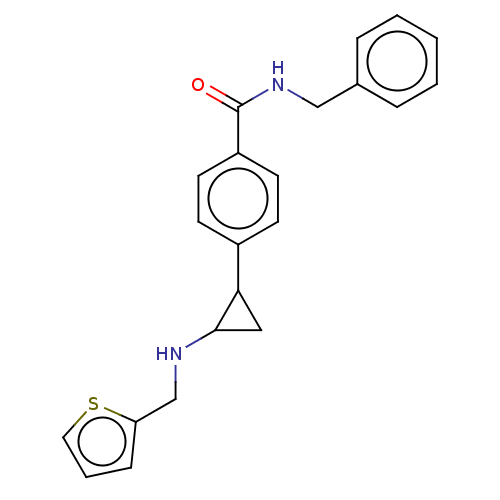
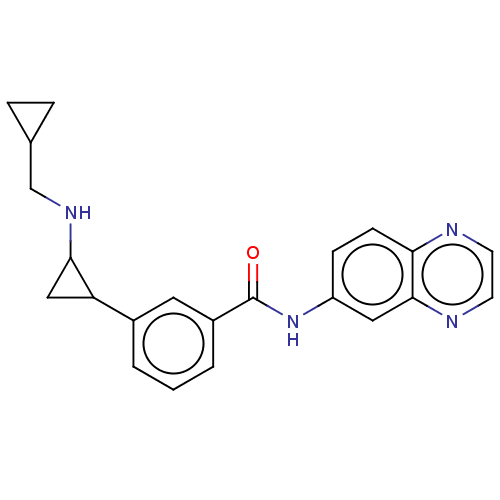
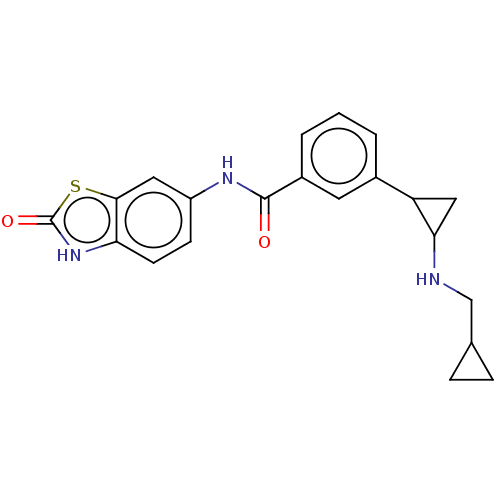
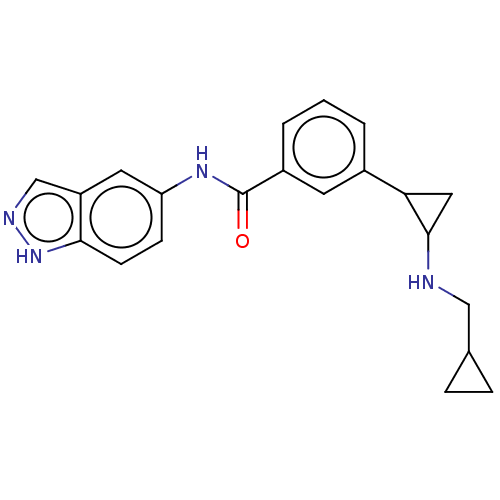
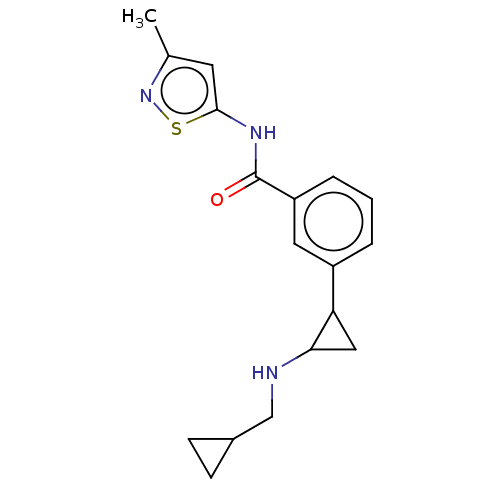
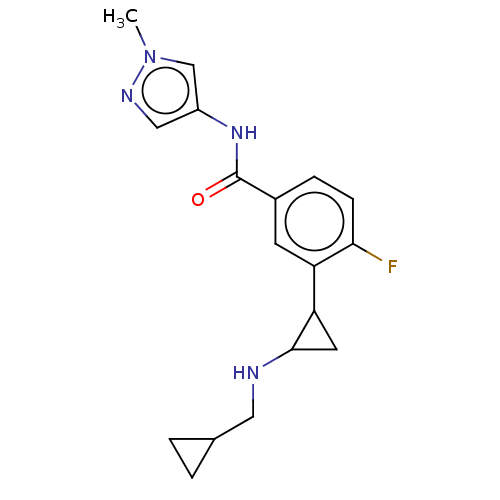
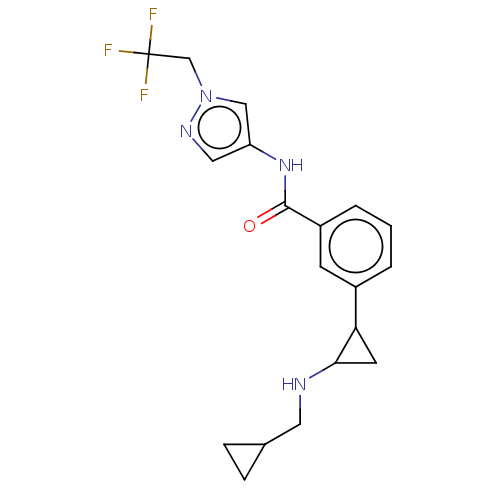
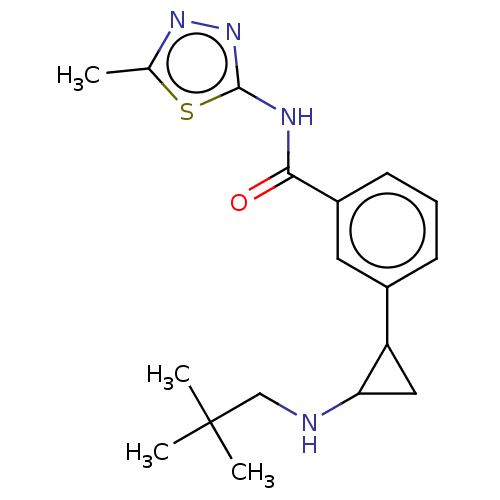
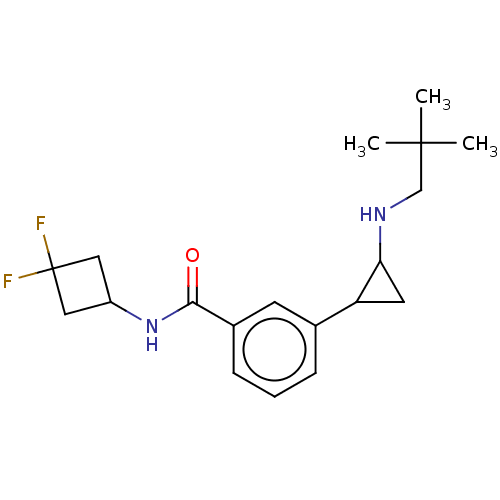
Report error Found 801 Enz. Inhib. hit(s) with all data for entry = 1749
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A test compound dissolved in 2.5% DMSO was added by 4 μL to 3 μL reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing...More data for this Ligand-Target Pair