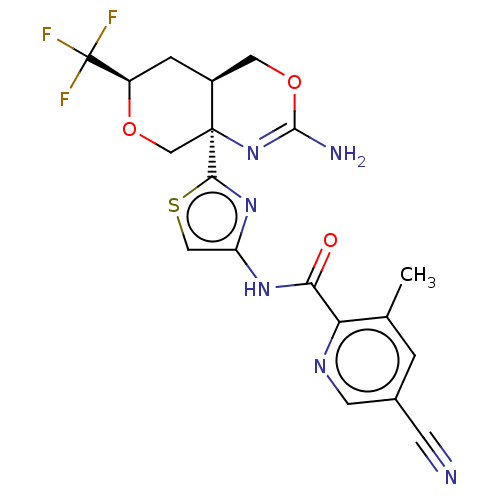
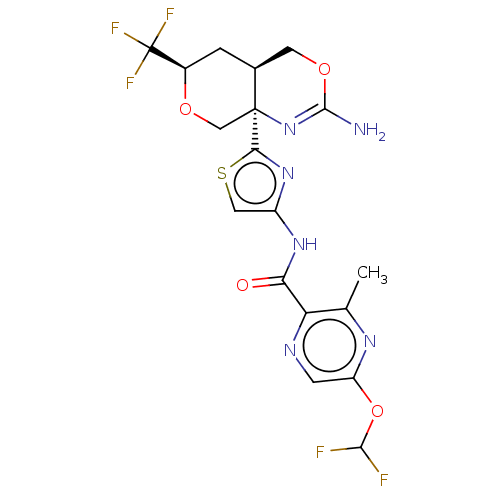
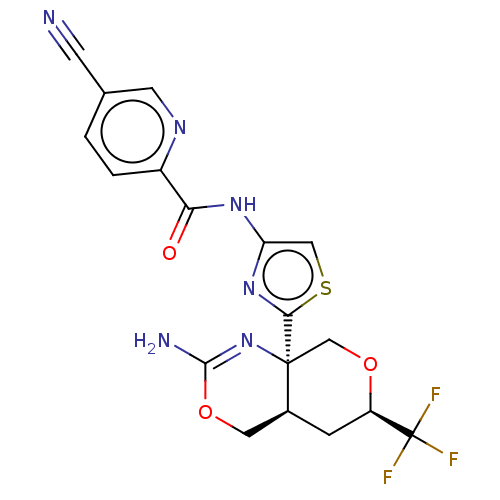
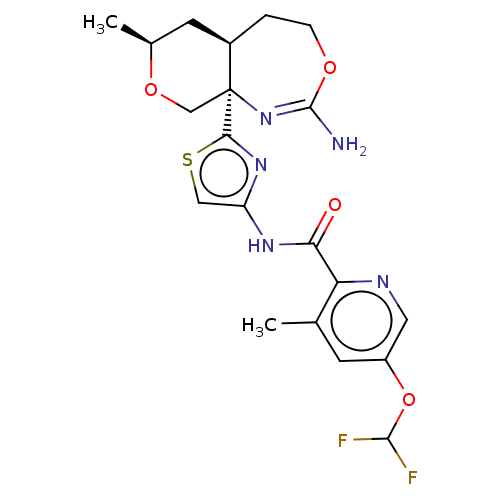
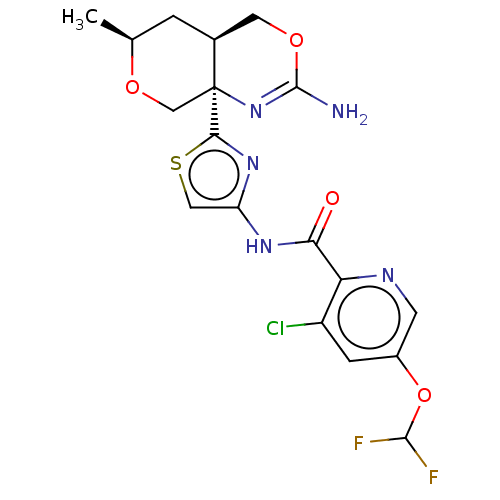
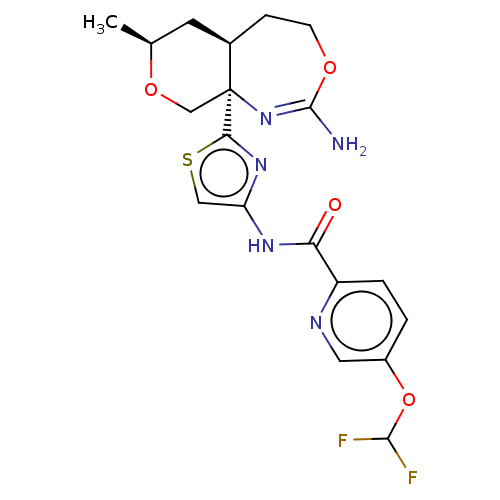
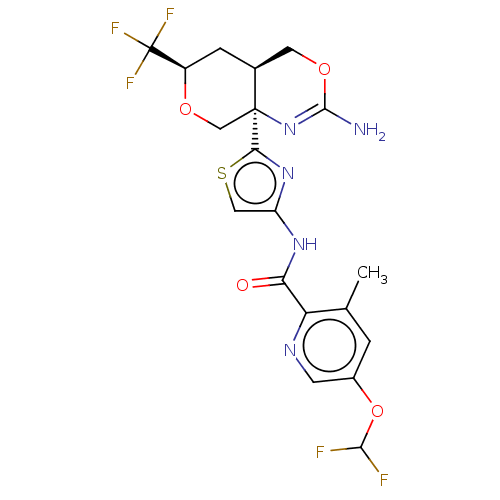
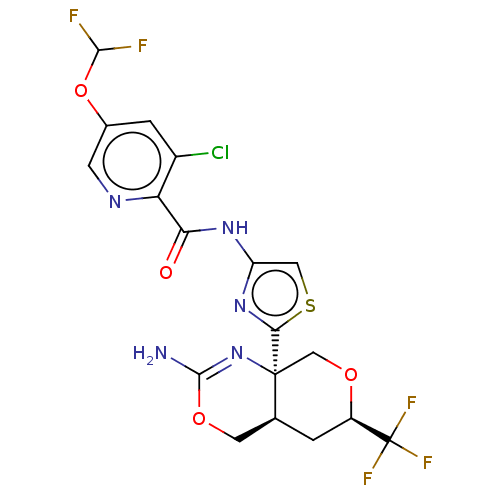
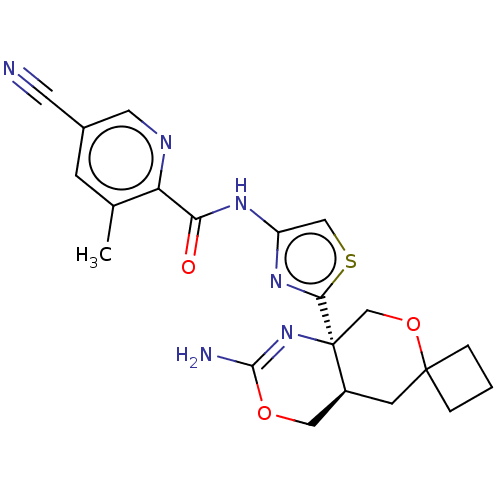
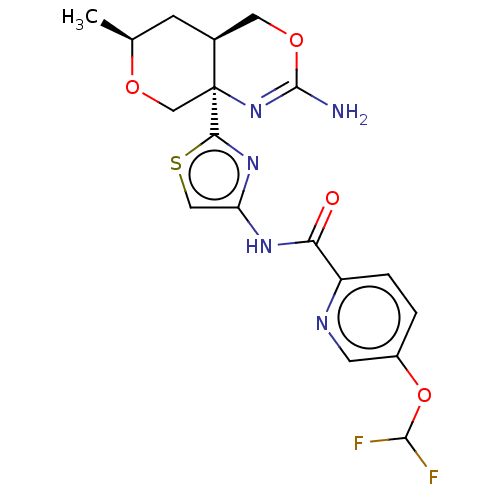
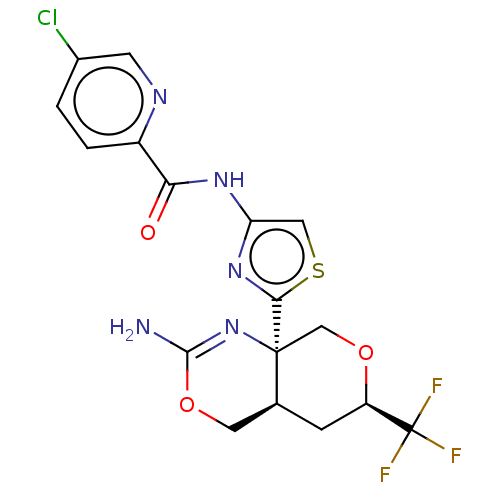
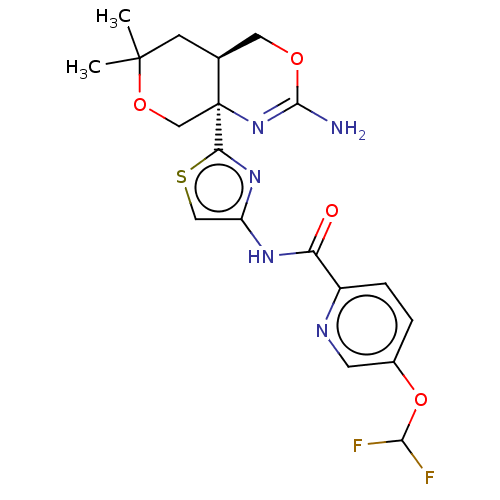
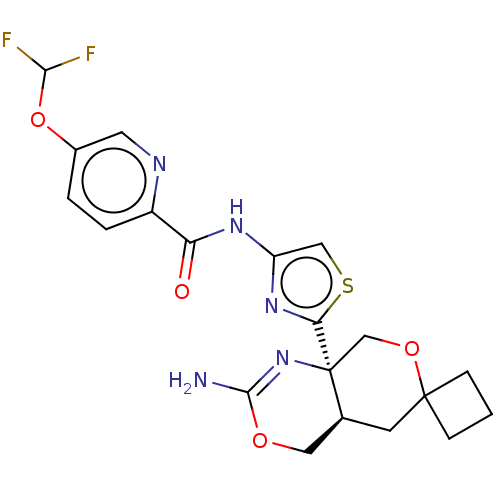
Report error Found 28 Enz. Inhib. hit(s) with all data for entry = 1825
Affinity DataIC50: 5nMAssay Description:The BACE1 binding assay measured beta-site amyloid precursor protein-cleaving enzyme (BACE) binding as a decrease in the counts of radioligand bound ...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:The BACE1 binding assay measured beta-site amyloid precursor protein-cleaving enzyme (BACE) binding as a decrease in the counts of radioligand bound ...More data for this Ligand-Target Pair
Affinity DataIC50: 27nMAssay Description:The BACE1 binding assay measured beta-site amyloid precursor protein-cleaving enzyme (BACE) binding as a decrease in the counts of radioligand bound ...More data for this Ligand-Target Pair
Affinity DataIC50: 71nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 85nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 112nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 123nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 124nMAssay Description:The BACE1 binding assay measured beta-site amyloid precursor protein-cleaving enzyme (BACE) binding as a decrease in the counts of radioligand bound ...More data for this Ligand-Target Pair
Affinity DataIC50: 146nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 155nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 162nMAssay Description:The BACE1 binding assay measured beta-site amyloid precursor protein-cleaving enzyme (BACE) binding as a decrease in the counts of radioligand bound ...More data for this Ligand-Target Pair
Affinity DataIC50: 184nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 185nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 214nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 270nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 310nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 370nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 371nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 395nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 475nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 561nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 610nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 860nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 2.26E+3nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 6.97E+3nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 1.08E+4nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+4nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair
Affinity DataIC50: 1.67E+4nMAssay Description:A synthetic APP substrate that can be cleaved by beta-secretase having N-terminal biotin and made fluorescent by the covalent attachment of Oregon Gr...More data for this Ligand-Target Pair