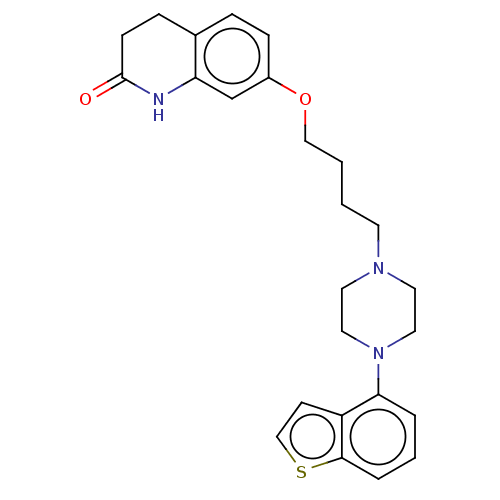
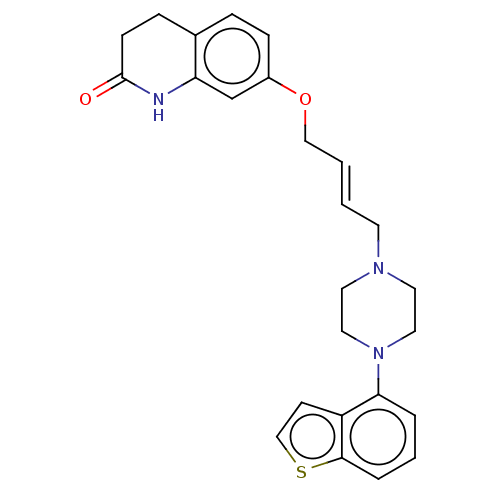
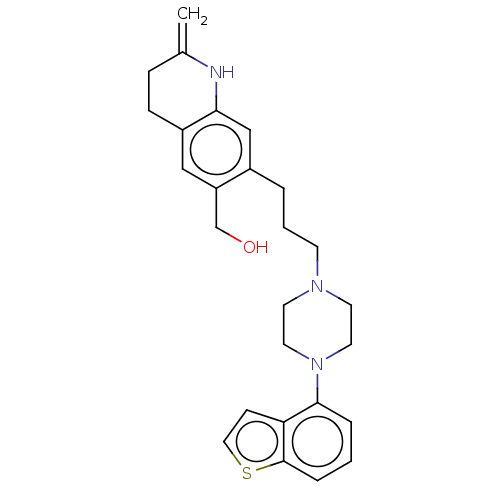
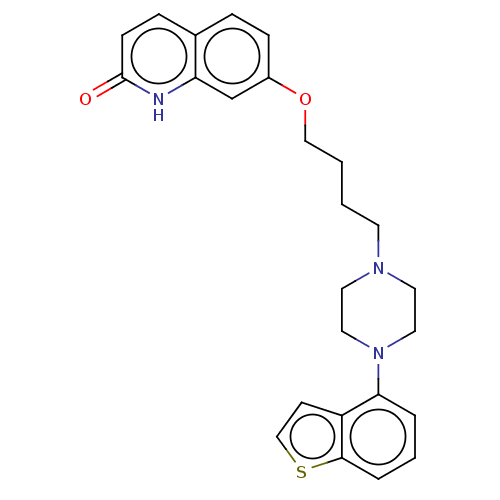
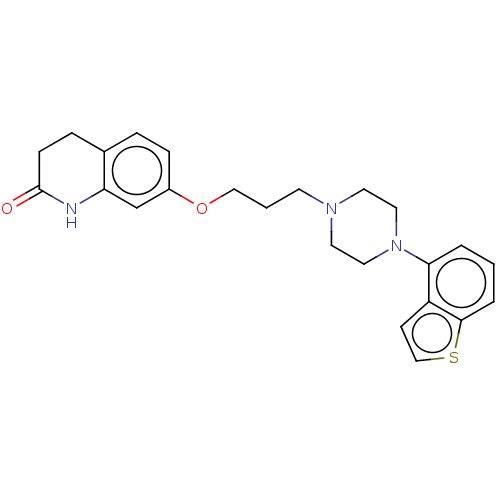
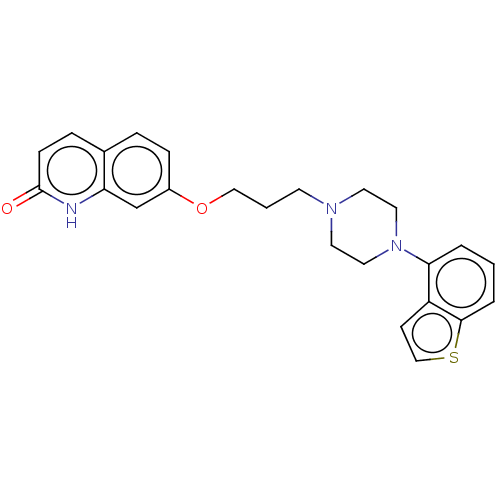
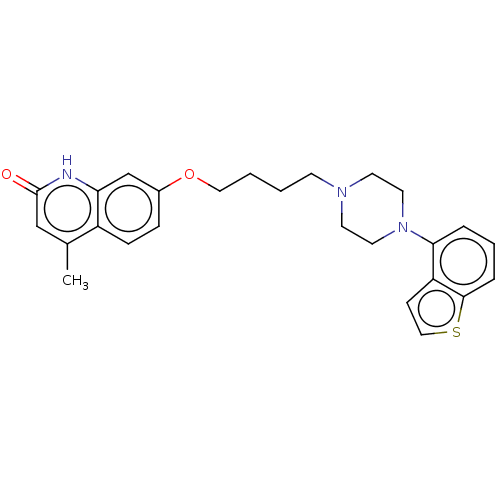
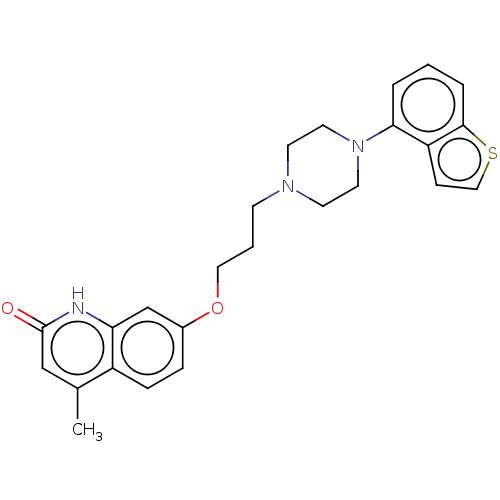
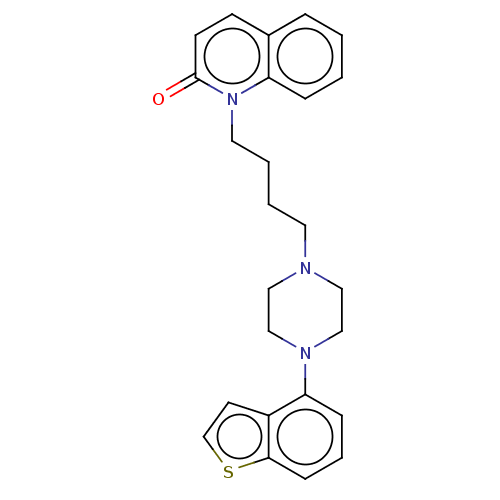
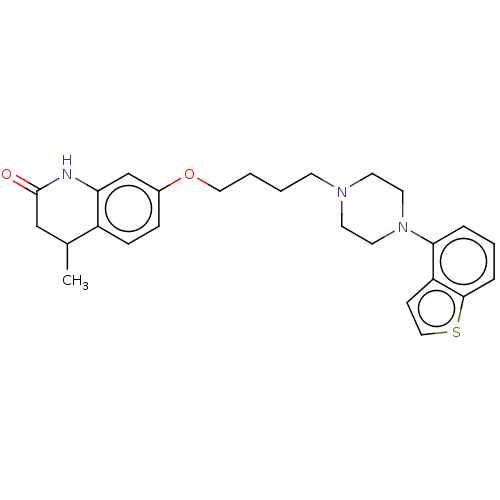
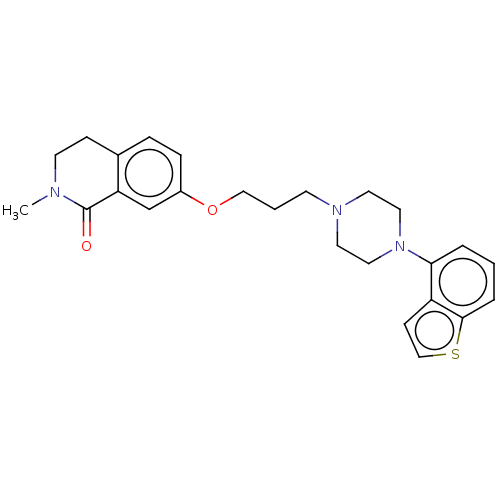
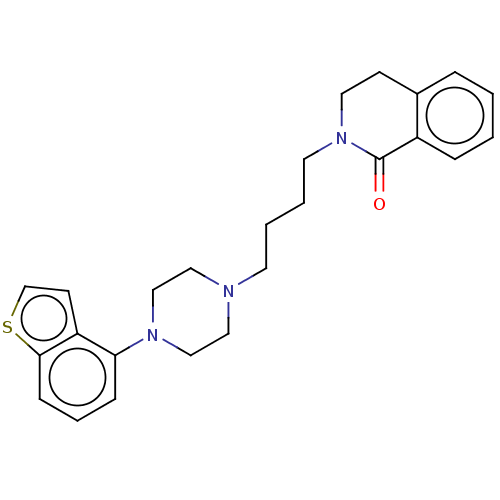
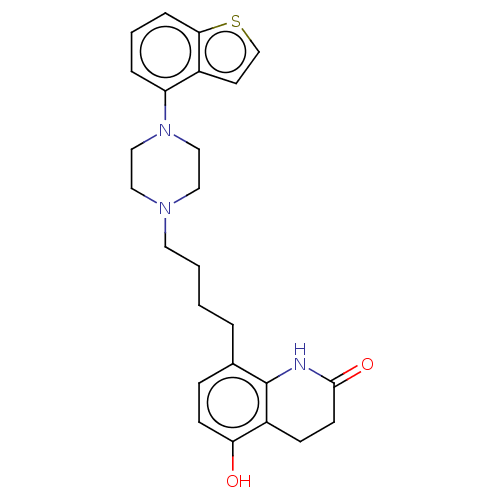
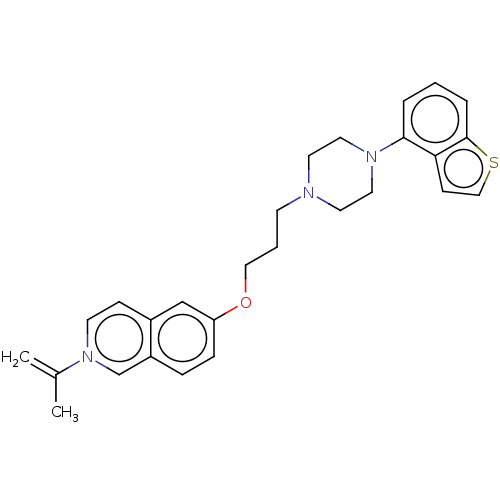
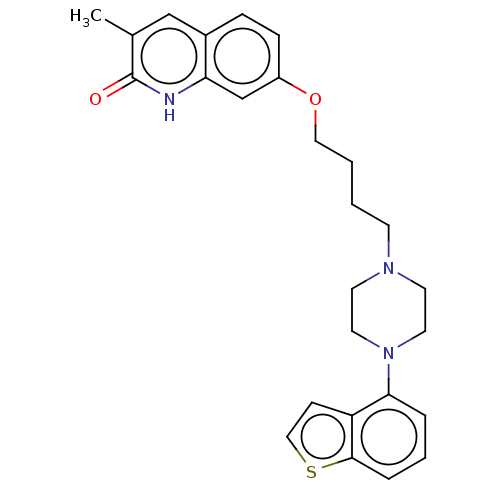
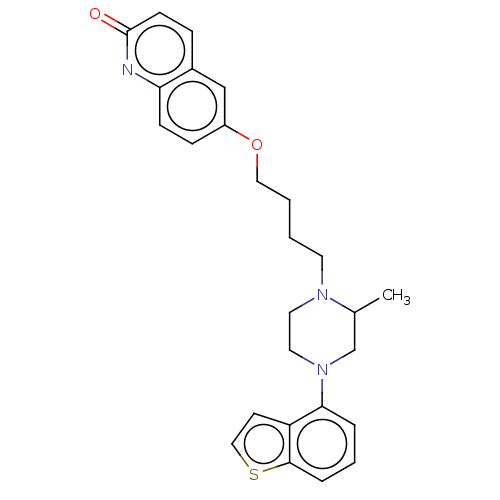
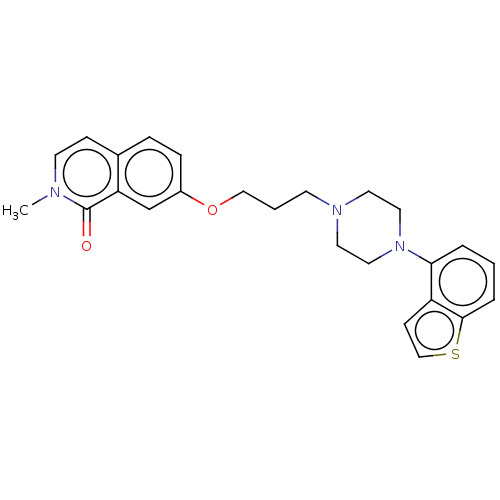
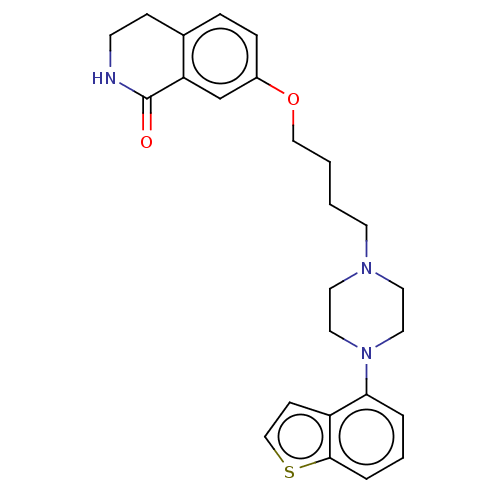
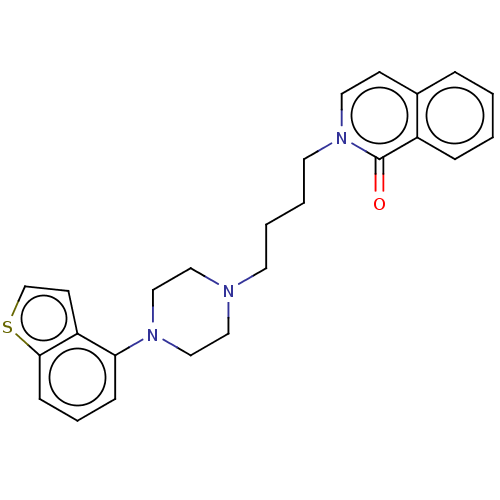
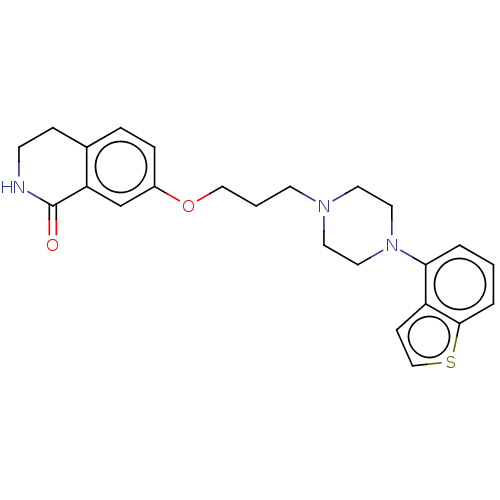
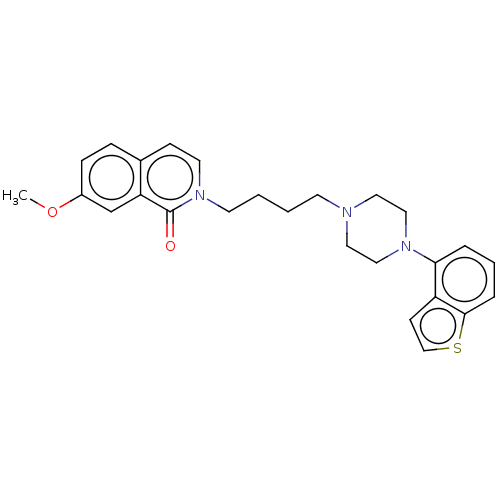
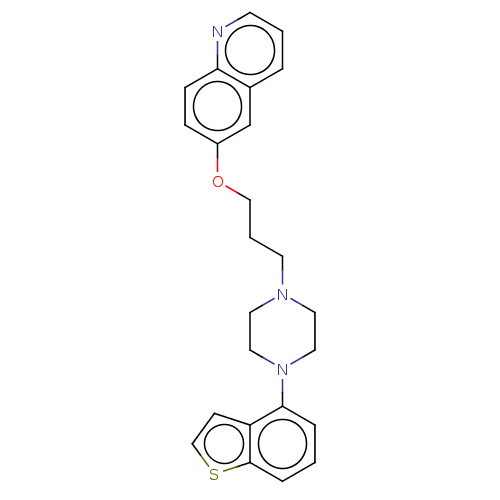
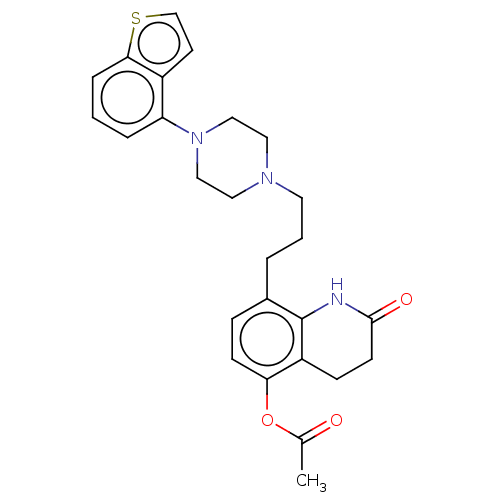
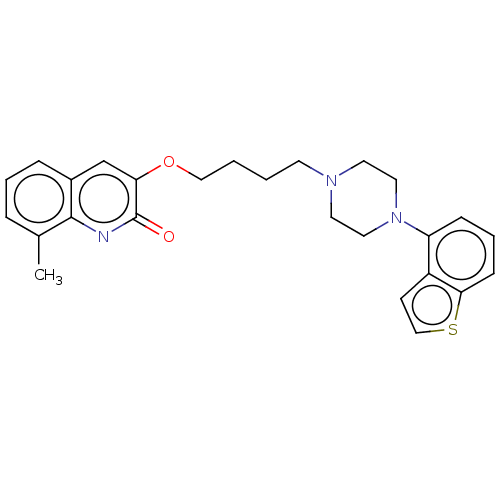
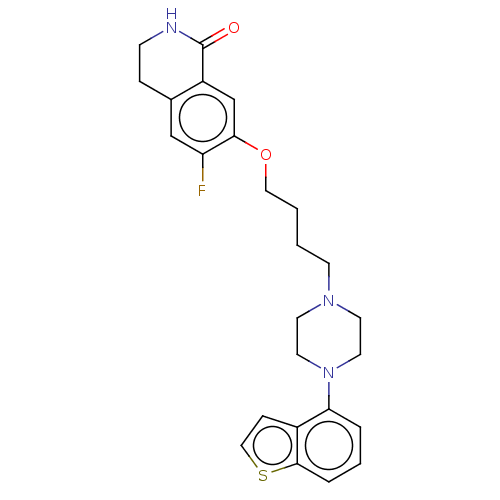
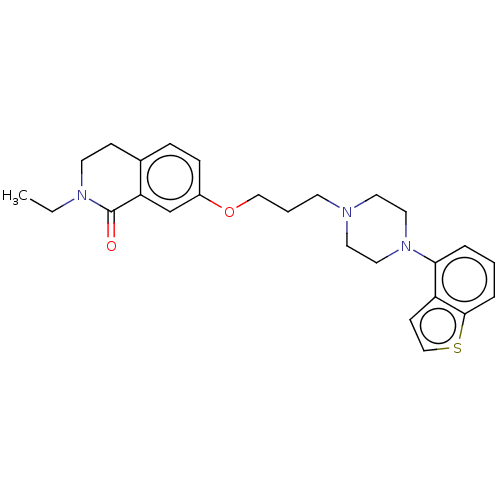
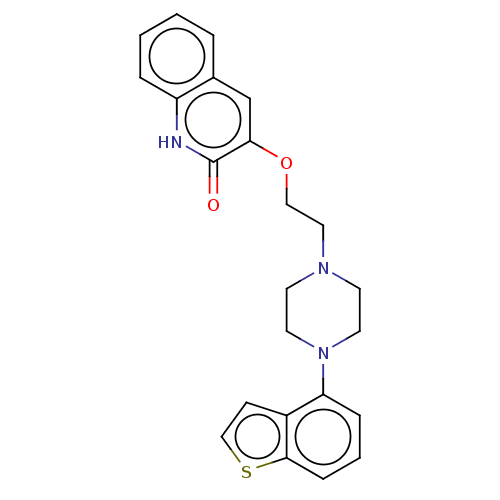
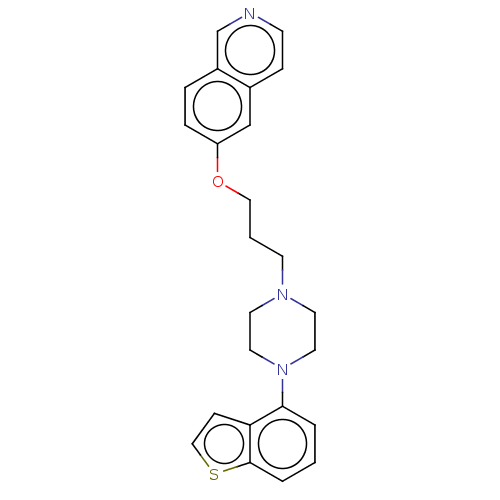
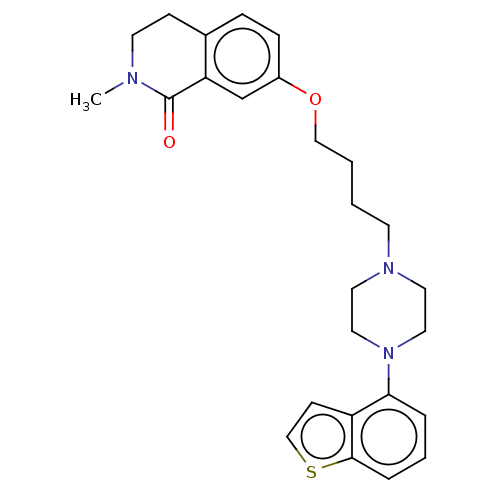
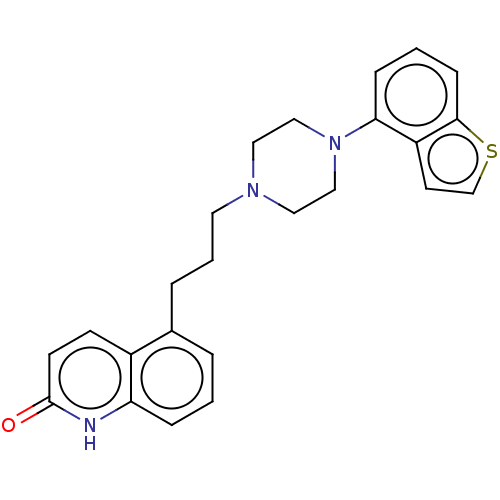
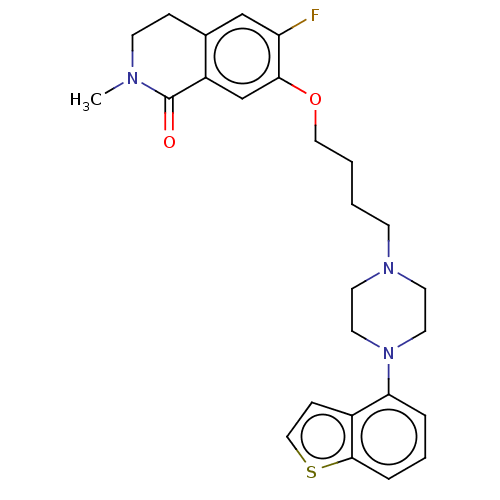
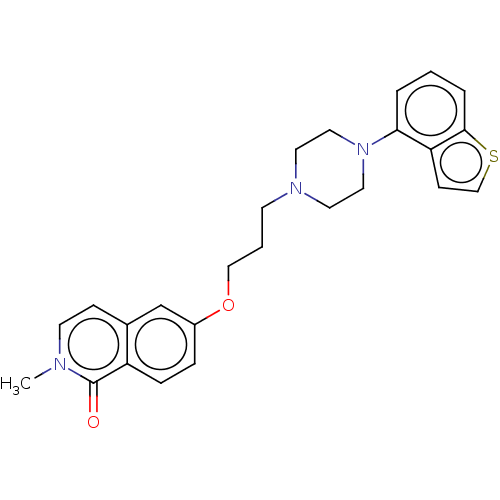
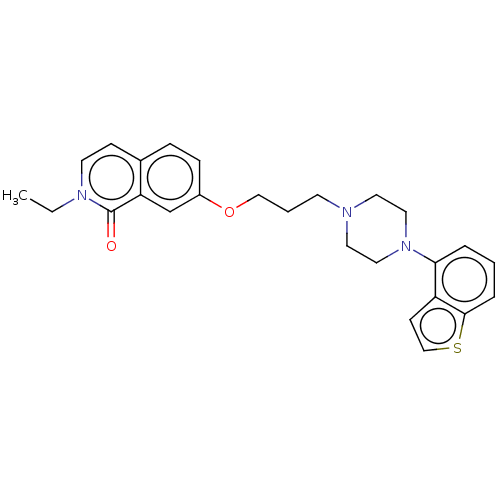
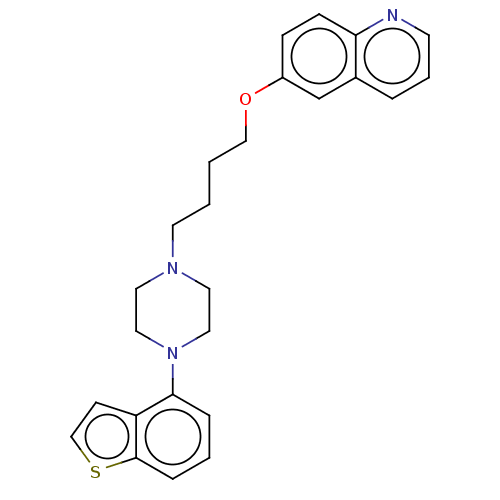
Report error Found 122 Enz. Inhib. hit(s) with all data for entry = 9191
Affinity DataKi: 0.100nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.100nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.200nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.200nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.200nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.400nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.400nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.5nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.5nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.5nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.600nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair
Affinity DataKi: 0.600nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.700nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.700nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.700nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.60nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.60nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.70nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.70nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair
Affinity DataKi: 1.90nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 1.90nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Dopamine D2: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-raclopride (final concentration 1 to 2 nM)...More data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair
Affinity DataKi: 2.10nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair
Affinity DataKi: 2.10nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair
Affinity DataKi: 2.10nMAssay Description:5-HT2A: The binding assay was performed using 40 μl of the membrane specimen, 20 μl of [3H]-Ketanserin (final concentration 1 to 3 nM), 20 ...More data for this Ligand-Target Pair