Reaction Details  Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataCell Reactant:
Serine Hydroxymethyltransferase
Syringe Reactant:
BDBM286
Meas. Tech.:
Isothermal Titration Calorimetry
Entry Date.:
03/31/03
ΔG°:
-29.678±n/a (kJ/mole)
Temperature:
298.15±n/a (K)
ΔH° :
36.784±n/a (kJ/mole)
ΔHobs :
36.784±n/a (kJ/mole)
Corrected for ΔHioniz:
not known
ΔCp :
-2.5±n/a (kJ/mole)
Citation
 Hom, RK; Huang, T; Gailunas, AF; Wang, C; Mamo, S; Maras, B; Fang, LY; Barra, D; Tung, JS; Walker, DE; Schirch, V; Davis, D; Thorsett, ED; Jewett, NE; Moon, JB; John, V Thermodynamic analysis of the binding of the polyglutamate chain of 5-formyltetrahydropteroylpolyglutamates to serine hydroxymethyltransferase. Biochemistry 37:13536-42 (1998) [PubMed] Article
Hom, RK; Huang, T; Gailunas, AF; Wang, C; Mamo, S; Maras, B; Fang, LY; Barra, D; Tung, JS; Walker, DE; Schirch, V; Davis, D; Thorsett, ED; Jewett, NE; Moon, JB; John, V Thermodynamic analysis of the binding of the polyglutamate chain of 5-formyltetrahydropteroylpolyglutamates to serine hydroxymethyltransferase. Biochemistry 37:13536-42 (1998) [PubMed] ArticleCell React
Source:
purified in the laboratory
Prep. Method:
SHMT, about 6 mg/mL, was
dialyzed overnight against 20 mM potassium phosphate
buffer at pH 7.0, containing 50 mM glycine and 1 mM DTT.
This enzyme was then diluted to the desired concentration
with the dialysis buffer.
Name:
Serine Hydroxymethyltransferase
Synonyms:
GLYC_RABIT | SHMT1
Type:
Enzyme
Mol. Mass.:
52984.86
Organism:
Oryctolagus cuniculus (rabbit)
Description:
P07511
Residue:
484
Sequence:
MATAVNGAPRDAALWSSHEQMLAQPLKDSDAEVYDIIKKESNRQRVGLELIASENFASRAVLEALGSCLNNKYSEGYPGQRYYGGTEHIDELETLCQKRALQAYGLDPQCWGVNVQPYSGSPANFAVYTALVEPHGRIMGLDLPDGGHLTHGFMTDKKKISATSIFFESMAYKVNPDTGYIDYDRLEENARLFHPKLIIAGTSCYSRNLDYGRLRKIADENGAYLMADMAHISGLVVAGVVPSPFEHCHVVTTTTHKTLRGCRAGMIFYRRGVRSVDPKTGKEILYNLESLINSAVFPGLQGGPHNHAIAGVAVALKQAMTPEFKEYQRQVVANCRALSAALVELGYKIVTGGSDNHLILVDLRSKGTDGGRAEKVLEACSIACNKNTCPGDKSALRPSGLRLGTPALTSRGLLEKDFQKVAHFIHRGIELTVQIQDDTGPRATLKEFKEKLAGDEKHQRAVRALRQEVESFAALFPLPGLPGF
Syringe React
Source:
B. Schircks
Prep. Method:
A concentrated solution of 5-CHO-H4PteGlun
was passed through a 1 cm x 10 cm Bio-Gel
P-2 column equilibrated with dialysis buffer. The concentration
of the solution was determined from its absorbance at
288 nm using an extinction coefficient of 31 500 M -1 cm -1
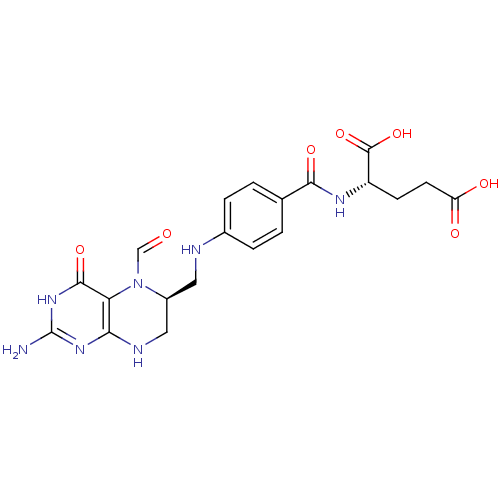
Name:
BDBM286
Synonyms:
(2S)-2-{[4-({[(6S)-2-amino-5-formyl-4-oxo-1,4,5,6,7,8-hexahydropteridin-6-yl]methyl}amino)phenyl]formamido}pentanedioic acid | 5-CHO-H4PteGlu1 | Folinic Acid | LEUCOVORIN CALCIUM | LEVOLEUCOVORIN CALCIUM | Leucovorin | Levoleucovorin
Type:
Small organic molecule
Emp. Form.:
C20H23N7O7
Mol. Mass.:
473.4393
SMILES:
Nc1nc2NC[C@H](CNc3ccc(cc3)C(=O)N[C@@H](CCC(O)=O)C(O)=O)N(C=O)c2c(=O)[nH]1